AGO2 Antibodies
Background
AGO2 is a key protein of the RNA-induced silencing complex (RISC), mainly present in the cytoplasm of eukaryotic cells. This protein recognizes and cleaves complementary target mRNAs by binding to small molecule RNAs (such as miRNAs or siRNAs), thereby regulating gene expression at the post-transcriptional level and participating in various biological processes such as cell differentiation, development, and antiviral defense. Since its first discovery in fruit flies in 1998, AGO2 has become a research hotspot due to its core role in the RNA interference (RNAi) pathway. The resolution of its three-dimensional structure has greatly advanced our understanding of the RNA silencing mechanism, nucleic acid-protein interactions, and gene regulatory networks, and provided an important theoretical basis for the development of related therapeutic strategies.
Structure of AGO2
The AGO2 (Argonaute 2) gene encodes a protein with a molecular weight of approximately 97 kDa. The precise molecular weight varies slightly among different species due to sequence differences. This protein contains multiple domains, with the core being the PAZ and PIWI domains. Among them, the PIWI domain has an activity similar to ribonuclease H and is crucial for achieving mRNA cleavage of the target. The AGO2 protein can bind to small molecule RNAs (such as miRNA or siRNA), and is recognized and anchored by the PAZ domain at the 3' end, while being fixed by the MID domain at the 5' end. Thus, it precisely guides the entire RNA-induced silencing complex (RISC) to the complementary mRNA sequence, ultimately completing the regulatory function of gene silencing. This structural feature makes it an indispensable catalytic core in the RNA interference pathway.
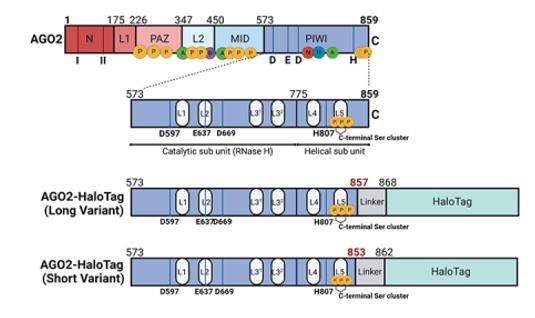 Fig. 1 Schematic to show known functionally important domains of AGO2.1
Fig. 1 Schematic to show known functionally important domains of AGO2.1
Key structural properties of AGO2:
- Catalytic core formed by PAZ and PIWI dual domains
- PIWI RNase H cutting activity structure domain
- MID structure domain is responsible for the small RNA 5 'end fixed and recognition
- The channels composed of aromatic amino acid residues are used to precisely position the target mRNA
- Conservative histidine and aspartic acid residue catalytic triplets
Functions of AGO2
The core function of the AGO2 protein is to execute gene silencing in the RNA interference pathway. The specific functional roles are as follows:
| Function | Description |
| Gene Silencing | As the catalytic core of the RNA-induced silencing complex, it precisely down-regulates gene expression at the post-transcriptional level by either cleaving the target mRNA or inhibiting its translation. |
| Antiviral Defense | In various biological mechanisms, interference and degradation of viral RNA occur, which constitutes an important part of the innate immunity. |
| Developmental Regulation | It participates in regulating key biological processes such as embryonic development, cell differentiation, and tissue homeostasis through the miRNA pathway. |
| Maintenance of Genome Stability | Silencing transposable elements and abnormal transcripts to protect the integrity of the genome and epigenetic regulation. |
| Metabolism and Signal Regulation | By regulating the abundance of specific mRNAs, it is involved in a wide range of signaling networks such as cellular metabolism, apoptosis, and stress responses. |
The mechanism of AGO2's action is closely related to the type of guide RNA it binds to: when binding to siRNA, it mainly mediates the direct cleavage of the target mRNA; while when binding to miRNA, it achieves more precise regulation mainly by inhibiting translation or promoting the destabilization of the mRNA, which demonstrates its multifunctionality and core position in the gene expression network.
Applications of AGO2 and AGO2 Antibody in Literature
1. Li, Jiong, et al. "YTHDF1 promotes mRNA degradation via YTHDF1‐AGO2 interaction and phase separation." Cell proliferation 55.1 (2022): e13157. https://doi.org/10.1111/cpr.13157
This study reveals that YTHDF1 recruits AGO2 through liquid-phase separation and promotes the formation of P-body to degrade mRNA. The deletion of YTHDF1 will change the P-body phase, resulting in delayed RNA aggregation and degradation. This provides a new perspective for post-transcriptional regulation of mRNA.
2. Shah, Kunal M., et al. "C-terminal tagging impairs AGO2 function." RNA biology 22.1 (2025): 1-24. https://doi.org/10.1080/15476286.2025.2534028
In this study, C-terminal haloTag-labeled AGO2 was constructed for the first time using CRISPaint technology. It was found that its binding to TNRC6A was weakened, and it significantly impaired RNA cutting, gene silencing and nuclear localization functions, indicating that C-terminal labeling affects the normal function of AGO2.
3. Huang, Hsiang-Chi, et al. "Nuclear AGO2 supports influenza A virus replication through type-I interferon regulation." Nucleic acids research 53.7 (2025): gkaf268. https://doi.org/10.1093/nar/gkaf268
Research has found that the influenza virus activates p53 to promote the accumulation of AGO2 protein in the cell nucleus, thereby silencing genes related to the interferon pathway and helping the virus evade the immune system. Inhibiting the nuclear translocation of p53 can enhance the antiviral response.
4. Liu, TingYu M., et al. "Dysregulation of AGO2-miRNA dynamics underlies the AGO2-associated Lessel–Kreienkamp syndrome." Nucleic Acids Research 53.19 (2025): gkaf1002. https://doi.org/10.1093/nar/gkaf1002
Research has found that mutations in the AGO2 gene associated with neurodevelopmental disorders (such as L192P) can alter its function, leading to delayed release and incorrect targeting of target RNA, disrupting the miRNA regulatory network within neurons, and thereby triggering pathological changes.
5. Javaid, Nasir, et al. "SFTSV NSs interacts with AGO2 to regulate the RNAi pathway for viral replication." Journal of Virology 99.4 (2025): e02205-24. https://doi.org/10.1128/jvi.02205-24
Research has found that the non-structural protein NSs of SFTS virus competitively inhibits the function of siRNA by forming a ternary complex with AGO2 and DICER in the host RNAi pathway, thereby evading antiviral RNA interference and promoting viral replication and pathogenicity.
Creative Biolabs: AGO2 Antibodies for Research
Creative Biolabs specializes in the production of high-quality AGO2 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom AGO2 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our AGO2 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Shah, Kunal M., et al. "C-terminal tagging impairs AGO2 function." RNA biology 22.1 (2025): 1-24. https://doi.org/10.1080/15476286.2025.2534028
Anti-AGO2 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-CFL1 Recombinant Antibody (CBFYC-1771) (CBMAB-C1833-FY)

-
Mouse Anti-BBS2 Recombinant Antibody (CBYY-0253) (CBMAB-0254-YY)

-
Mouse Anti-ESR1 Recombinant Antibody (Y31) (CBMAB-1208-YC)

-
Rabbit Anti-ENO2 Recombinant Antibody (BA0013) (CBMAB-0272CQ)

-
Mouse Anti-ADGRL2 Recombinant Antibody (V2-58519) (CBMAB-L0166-YJ)

-
Mouse Anti-ALB Recombinant Antibody (V2-180650) (CBMAB-A2186-YC)

-
Mouse Anti-DLG1 Monolconal Antibody (4F3) (CBMAB-0225-CN)

-
Mouse Anti-BACE1 Recombinant Antibody (61-3E7) (CBMAB-1183-CN)

-
Mouse Anti-EMP3 Recombinant Antibody (CBFYE-0100) (CBMAB-E0207-FY)

-
Mouse Anti-BMI1 Recombinant Antibody (CBYC-P026) (CBMAB-P0108-YC)

-
Mouse Anti-GFAP Recombinant Antibody (20) (CBMAB-G2914-LY)

-
Mouse Anti-CD8 Recombinant Antibody (C1083) (CBMAB-C1083-LY)

-
Mouse Anti-ADIPOR2 Recombinant Antibody (V2-179983) (CBMAB-A1369-YC)

-
Mouse Anti-ADAM29 Recombinant Antibody (V2-179787) (CBMAB-A1149-YC)

-
Mouse Anti-ADAM12 Recombinant Antibody (V2-179752) (CBMAB-A1114-YC)

-
Mouse Anti-CD164 Recombinant Antibody (CBFYC-0077) (CBMAB-C0086-FY)

-
Mouse Anti-FYN Recombinant Antibody (10) (CBMAB-S6332-CQ)

-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Mouse Anti-EGR1 Recombinant Antibody (CBWJZ-100) (CBMAB-Z0289-WJ)

-
Mouse Anti-BSN Recombinant Antibody (219E1) (CBMAB-1228-CN)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






