PIN1 Antibodies
Background
The PIN1 gene encodes a peptidyl-prolyl cis-trans isomerase, which regulates the conformation and function of various proteins by catalyzing the isomerization of specific phosphorylated serine/threonine-proline groups. Its functions mainly involve key biological processes such as cell cycle progression, signal transduction, and transcriptional regulation, and are crucial for maintaining normal cell proliferation and differentiation. This gene was first identified in 1996, and its unique isomerase mechanism provides an important example for understanding the temporal regulation of phosphorylation-dependent signaling pathways. Studies have shown that PIN1 is abnormally expressed in various diseases, especially in cancer and neurodegenerative diseases. Its dysfunction can drive pathological processes by affecting the conformation of key proteins such as p53 and Tau. Therefore, PIN1 has become an important object for disease molecular mechanism research and targeted drug development. The structural and functional studies have continuously deepened our understanding of the regulatory network of post-translational modification of proteins.
Structure of PIN1
The PIN1 gene encodes a peptidyl-prolyl cis-trans isomerase with a molecular weight of approximately 18 kDa. Its molecular weight is relatively conserved among different species, mainly due to the high sequence homology of the catalytic domain.
| Species | Human | Mouse | Rat | Fruit fly | Yeast |
| Molecular Weight (kDa) | 18.0 | 17.9 | 18.1 | 18.3 | 18.6 |
| Primary Structural Differences | Contains the WW domain and the PPI enzyme domain | Extremely similar to humans in origin | The catalytic core is highly conserved | Contains only a single PPI enzyme domain | The direct homolog of PIN1 |
The PIN1 protein is composed of 163 amino acids and its three-dimensional structure is shown as a compact spherical protein. Its functional core relies on two independent domains: the N-terminal WW domain is responsible for recognizing and binding to substrate proteins containing phosphorylated serine/threonine-proline (pSer/Thr-Pro) motifs; the C-terminal peptidyl-prolyl isomerase (PPIase) domain catalyzes the formation of cis-trans isomerization of the peptide bond preceding the proline residue in the substrate. These two domains are connected by a flexible hinge region and work together to alter the conformation and activity of various signaling proteins and cyclins, thereby playing a crucial regulatory role in cell proliferation, differentiation, and stress responses.
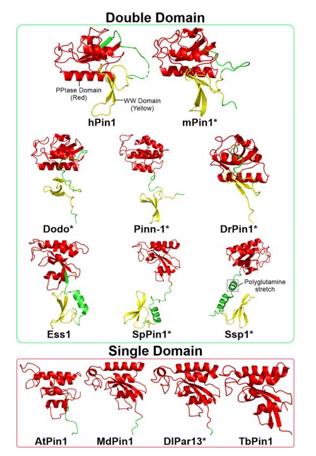 Fig. 1 Structures of Pin1 orthologs across various species.1
Fig. 1 Structures of Pin1 orthologs across various species.1
Key structural properties of PIN1:
- Two-domain modular architecture
- Specific pocket for substrate binding
- Hydrophobic environment of catalytic center
Functions of PIN1
The prolyl isomerase encoded by the PIN1 gene mainly functions to regulate cell signal transduction and cell cycle progression. However, it is also widely involved in various pathological and physiological processes, including cancer occurrence and neurodegenerative diseases.
| Function | Description |
| Protein conformational regulation | Specifically recognizes and binds to phosphorylated serine/threonine-proline (pSer/Thr-Pro) motifs, catalyzes the cis-trans isomerization of proline peptide bonds, thereby altering the spatial conformation and functional state of the substrate protein. |
| Cell Cycle Regulation | By altering cell cycle proteins (such as Cyclin D1) and phosphorylating CDK, it drives the cell to transition from the G2 phase to the M phase, which is a key regulatory factor for cell proliferation. |
| Signal Pathway Hub | It acts as a molecular switch in multiple important signaling pathways such as Wnt/β-catenin, NF-κB, and p53, regulating downstream gene transcription by altering the stability and subcellular localization of key signaling proteins. |
| Tumor Promotion | It is overexpressed in various cancers. Its isomerase activity can abnormally activate proto-oncogenes (such as c-Myc) and inactivate tumor suppressor genes (such as p53), thereby driving the proliferation, invasion and metastasis of tumor cells. |
| Neurotoxicity Regulation | In pathological processes such as Alzheimer's disease, PIN1 can bind to the over-phosphorylated Tau protein, influencing its conformation and aggregation tendency, thereby participating in the regulation of neurofibrillary tangle formation. |
Unlike many enzymes with a single and continuous activity, the functional activity of PIN1 exhibits a strict phosphorylation-dependent switch characteristic. This feature enables it to precisely respond to upstream kinase/phosphatase signals, achieving instantaneous and reversible regulation of the downstream signaling network at specific temporal and spatial nodes, thereby playing a crucial role in cell fate determination.
Applications of PIN1 and PIN1 Antibody in Literature
1. Lee, Yew Mun, et al. "The kingdom of the prolyl-isomerase Pin1: The structural and functional convergence and divergence of Pin1." Frontiers in Cell and Developmental Biology 10 (2022): 956071. https://doi.org/10.3389/fcell.2022.956071
It has been discovered for over 20 years that Pin1 plays a crucial and highly conserved role in various diseases such as cancer and neurodegenerative disorders. The development of specific drugs for Pin1 remains challenging, which is driving the study of the functions and structures of Pin1 homologues in different species and providing new ideas for targeted therapy.
2. Jeong, Jessica, et al. "Pin1-catalyzed conformation changes regulate protein ubiquitination and degradation." Cells 13.9 (2024): 731. https://doi.org/10.3390/cells13090731
The article indicates that PIN1 regulates the stability of substrate proteins through the isomerization of phosphorylated Ser/Thr-Pro motifs. It promotes the accumulation of oncogenic proteins in cancer and affects the clearance of pathogenic proteins in neurodegenerative diseases. Targeting the PIN1-ubiquitination pathway may provide new strategies for treatment.
3. Caligiuri, Isabella, et al. "The metabolic crosstalk between PIN1 and the tumour microenvironment." Seminars in Cancer Biology. Vol. 91. Academic Press, 2023. https://doi.org/10.1016/j.semcancer.2023.03.001
The article indicates that PIN1, as a phosphorylation-specific isomerase, regulates the stability of numerous oncogenic proteins and tumor suppressor proteins, thereby leading to the metabolic reprogramming of cancer cells (such as the Waddington effect) and their interaction with the tumor microenvironment, thereby driving the progression of cancer.
4. Liu, Chuanfeng, et al. "The mechanisms of Pin1 as targets for cancer therapy." Frontiers in Immunology 15 (2024): 1482088. https://doi.org/10.3389/fimmu.2024.1482088
The article indicates that PIN1 serves as an anti-cancer target, driving oncogenic signals through the isomerization of phosphorylated threonine-proline motifs. Its natural inhibitor can block the oncogenic activity of PIN1, providing a new strategy for reversing treatment resistance.
5. Li, Jingyi, et al. "Roles of peptidyl-prolyl isomerase Pin1 in disease pathogenesis." Theranostics 11.7 (2021): 3348. https://doi.org/10.7150/thno.45889
The article indicates that PIN1, as a peptidyl-prolyl isomerase, regulates key processes such as cell cycle, movement and apoptosis by catalyzing the cis-trans isomerization of proline in the substrate protein. Its dysfunction is closely related to the occurrence and development of various diseases.
Creative Biolabs: PIN1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality PIN1 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom PIN1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our PIN1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Lee, Yew Mun, et al. "The kingdom of the prolyl-isomerase Pin1: The structural and functional convergence and divergence of Pin1." Frontiers in Cell and Developmental Biology 10 (2022): 956071. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3389/fcell.2022.956071
Anti-PIN1 antibodies
Products List
 Loading...
Loading...
Hot products 
-
Mouse Anti-C4B Recombinant Antibody (CBYY-C2996) (CBMAB-C4439-YY)

-
Mouse Anti-ADRB2 Recombinant Antibody (V2-180026) (CBMAB-A1420-YC)

-
Mouse Anti-FPR2 Recombinant Antibody (1D6) (CBMAB-F2628-CQ)

-
Mouse Anti-ASB9 Recombinant Antibody (1D8) (CBMAB-A0529-LY)

-
Mouse Anti-AQP2 Recombinant Antibody (G-3) (CBMAB-A3359-YC)

-
Mouse Anti-GIPC2 Recombinant Antibody (10) (CBMAB-G0476-LY)

-
Mouse Anti-ENPP1 Recombinant Antibody (CBFYE-0159) (CBMAB-E0375-FY)

-
Mouse Anti-CD59 Recombinant Antibody (CBXC-2097) (CBMAB-C4421-CQ)

-
Mouse Anti-CD33 Recombinant Antibody (6C5/2) (CBMAB-C8126-LY)

-
Human Anti-SARS-CoV-2 Spike Recombinant Antibody (CR3022) (CBMAB-CR014LY)

-
Mouse Anti-DHFR Recombinant Antibody (D0821) (CBMAB-D0821-YC)

-
Mouse Anti-CD2AP Recombinant Antibody (BR083) (CBMAB-BR083LY)

-
Mouse Anti-CGAS Recombinant Antibody (CBFYM-0995) (CBMAB-M1146-FY)

-
Mouse Anti-ARHGAP5 Recombinant Antibody (54/P190-B) (CBMAB-P0070-YC)

-
Mouse Anti-CFL1 (Phospho-Ser3) Recombinant Antibody (CBFYC-1770) (CBMAB-C1832-FY)

-
Mouse Anti-CSPG4 Recombinant Antibody (CBFYM-1050) (CBMAB-M1203-FY)

-
Mouse Anti-CD63 Recombinant Antibody (CBXC-1200) (CBMAB-C1467-CQ)

-
Mouse Anti-ADAM29 Recombinant Antibody (V2-179787) (CBMAB-A1149-YC)

-
Mouse Anti-DES Monoclonal Antibody (440) (CBMAB-AP1857LY)

-
Mouse Anti-CFL1 Recombinant Antibody (CBFYC-1771) (CBMAB-C1833-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






