ACSL1 Antibodies
Background
The ACSL1 gene encodes a member of the long-chain fatty acyl-CoA synthetase family, which is mainly highly expressed in metabolically active tissues such as fat, muscle, and liver. This enzyme catalyzes the activation of long-chain fatty acids into fatty acyl-CoA, participating in fatty acid oxidation, triglyceride synthesis, and lipid signal regulation. Studies have shown that ACSL1 mediates lipid accumulation and resistance to ferroptosis in tumor metabolic reprogramming, regulates ceramide synthesis in insulin-resistant tissues, and is transcriptionally activated by C/EBPα during adipogenic differentiation. Abnormal expression of ACSL1 is closely related to the progression of lung cancer, peritoneal metastasis of ovarian cancer, and metabolic syndrome. Targeted inhibition of ACSL1 by Triacsin C can synergistically enhance the efficacy of chemotherapeutic drugs, providing a new strategy for the intervention of lipid metabolism-related diseases.
Structure of ACSL1
The ACSL1 protein has a molecular weight of approximately 78 kDa and is highly conserved among different species. This enzyme is located on the outer mitochondrial membrane and the endoplasmic reticulum. It catalyzes the formation of acyl-CoA from long-chain fatty acids and participates in lipid synthesis and β-oxidation.
| Species | Human | Mouse | Rat | Pig |
| Molecular Weight (kDa) | 78.2 | 78.1 | 78.2 | 78.3 |
| Primary Structural Differences | Contains AMP binding domain | 97% homology to human | Highly homologous to mouse | Differences in tissue expression profile |
ACSL1 is composed of 720 amino acids and includes an AMP-binding domain, a fatty acid acylation domain, and multiple phosphorylation sites. Its three-dimensional structure exhibits the typical fold pattern of acyl-CoA synthetase: the N-terminal core catalytic domain is responsible for ATP binding, and the C-terminal domain mediates substrate recognition. The active site of the enzyme captures fatty acids through the "induced fit" mechanism and catalyzes two-step reactions to generate fatty acyl-CoA. This enzyme is highly expressed in cardiac muscle and adipose tissue, and its activity is regulated by insulin and PPARs transcription.
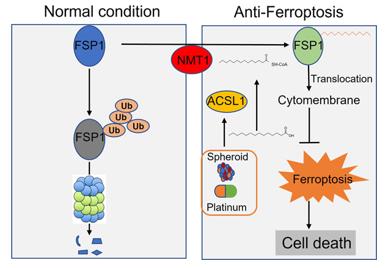 Fig. 1 ACSL1 involvement in anti-ferroptosis in ovarian cancer.1
Fig. 1 ACSL1 involvement in anti-ferroptosis in ovarian cancer.1
Key structural features of ACSL1:
- Contains an AMP binding domain and a fatty acid acylation domain
- Hydrophobic pocket recognizes long-chain fatty acid substrates
- ATP binding site mediates the catalytic two-step reaction
- Serine phosphorylation site regulates enzyme activity and localization
Functions of ACSL1
The core function of ACSL1 is to activate long-chain fatty acids and participate in energy metabolism and lipid synthesis. This enzyme regulates the level of acyl-CoA, thereby influencing various physiological processes.
| Function | Description |
| Fatty acid activation | Catalyzes the generation of acyl-CoA from long-chain fatty acids, providing substrates for β-oxidation and triglyceride synthesis. |
| Energy metabolism regulation | Promotes fatty acid oxidation in cardiac and skeletal muscles, maintaining energy homeostasis. |
| Lipid synthesis | Participates in triglyceride synthesis in adipose tissue, regulating fat storage. |
| Signal transduction | Mediates the participation of fatty acyl-CoA in protein phosphorylation modification, regulating inflammation and insulin signaling. |
| Tumor metabolic remodeling | Promotes lipid accumulation in cancer cells, enhancing iron death resistance and metastasis ability. |
ACSL1 shows substrate specificity for C16-C20 long-chain fatty acids, and its expression is regulated by transcription factors such as PPARs and SREBP.
Applications of ACSL1 and ACSL1 Antibody in Literature
1. Zhang, Qingyu, et al. "ACSL1-induced ferroptosis and platinum resistance in ovarian cancer by increasing FSP1 N-myristylation and stability." Cell death discovery 9.1 (2023): 83. https://doi.org/10.1038/s41420-023-01385-2
The article indicates that ACSL1 enhances the N-myristoylation modification of FSP1, inhibits its degradation and promotes membrane translocation, thereby reducing lipid oxidation levels, endowing ovarian cancer cells with anti-ferroptosis ability against iron, and promoting peritoneal metastasis.
2. Ma, Yunxia, et al. "The diagnostic value of ACSL1, ACSL4, and ACSL5 and the clinical potential of an ACSL inhibitor in non-small-cell lung cancer." Cancers 16.6 (2024): 1170. https://doi.org/10.3390/cancers16061170
The article indicates that ACSL1 is highly expressed in lung cancer cells and lung adenocarcinoma tissues, and can serve as a biomarker for lung adenocarcinoma. Its expression is related to cell differentiation, and the combination of ACSL inhibitor Triacsin C can overcome chemotherapy and EGFR-TKI resistance.
3. Roszczyc-Owsiejczuk, Kamila, et al. "shRNA-mediated down-regulation of Acsl1 reverses skeletal muscle insulin resistance in obese C57BL6/J mice." Plos one 19.8 (2024): e0307802. https://doi.org/10.1371/journal.pone.0307802
The article indicates that ACSL1 mediates muscle insulin resistance induced by high-fat diet. Downregulation of Acsl1 can reduce the accumulation of ceramide and glycerol diester, improve insulin sensitivity and glucose uptake, and its benefits outweigh the negative effects of reduced mitochondrial fatty acid metabolism.
4. Yang, Xiuqin, et al. "Transcriptional regulation associated with subcutaneous adipogenesis in porcine ACSL1 gene." Biomolecules 13.7 (2023): 1057. https://doi.org/10.3390/biom13071057
The article indicates that ACSL1 is regulated by C/EBPα and mediates its adipogenic differentiation function. The promoter double-linked SNP is unique to Min pigs and can reduce ACSL1 expression. The study reveals the transcriptional regulation, alternative splicing and molecular markers of pig ACSL1.
5. Szaluś-Jordanow, Olga, et al. "A primary multiple pleomorphic rhabdomyosarcoma of the heart in an adult dog." BMC Veterinary Research 19.1 (2023): 137. https://doi.org/10.3390/biom9100555
The article indicates that ACSL1 mediates the production of GM-CSF induced by TNFα. Inhibiting ACSL1 activity can block the phosphorylation of p38, ERK, and NF-κB, and significantly reduce the secretion of GM-CSF. ACSL1 is a potential new target for regulating tumor growth.
Creative Biolabs: ACSL1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality ACSL1 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom ACSL1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our ACSL1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Zhang, Qingyu, et al. "ACSL1-induced ferroptosis and platinum resistance in ovarian cancer by increasing FSP1 N-myristylation and stability." Cell death discovery 9.1 (2023): 83. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1038/s41420-023-01385-2
Anti-ACSL1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-BCL6 Recombinant Antibody (CBYY-0435) (CBMAB-0437-YY)

-
Mouse Anti-CD33 Recombinant Antibody (P67.6) (CBMAB-C10189-LY)

-
Mouse Anti-CD59 Recombinant Antibody (CBXC-2097) (CBMAB-C4421-CQ)

-
Mouse Anti-ASTN1 Recombinant Antibody (H-9) (CBMAB-1154-CN)

-
Mouse Anti-BANF1 Recombinant Antibody (3F10-4G12) (CBMAB-A0707-LY)

-
Mouse Anti-CORO1A Recombinant Antibody (4G10) (V2LY-1206-LY806)

-
Mouse Anti-DLL4 Recombinant Antibody (D1090) (CBMAB-D1090-YC)

-
Mouse Anti-GFP Recombinant Antibody (28) (CBMAB-G3038-LY)

-
Mouse Anti-AMOT Recombinant Antibody (CBYC-A564) (CBMAB-A2552-YC)

-
Human Anti-SARS-CoV-2 Spike Recombinant Antibody (CBC05) (CBMAB-CR005LY)

-
Mouse Anti-ACKR3 Recombinant Antibody (V2-261265) (CBMAB-C1023-LY)

-
Mouse Anti-CASQ1 Recombinant Antibody (CBFYC-0863) (CBMAB-C0918-FY)

-
Mouse Anti-ABL2 Recombinant Antibody (V2-179121) (CBMAB-A0364-YC)

-
Mouse Anti-ALB Recombinant Antibody (V2-363290) (CBMAB-S0173-CQ)

-
Mouse Anti-AOC3 Recombinant Antibody (CBYY-0014) (CBMAB-0014-YY)

-
Mouse Anti-ADV Recombinant Antibody (V2-503423) (CBMAB-V208-1364-FY)

-
Mouse Anti-A2M Recombinant Antibody (V2-178822) (CBMAB-A0036-YC)

-
Rat Anti-(1-5)-α-L-Arabinan Recombinant Antibody (V2-501861) (CBMAB-XB0003-YC)

-
Rat Anti-FABP3 Recombinant Antibody (CBXF-2299) (CBMAB-F1612-CQ)

-
Mouse Anti-APOA1 Monoclonal Antibody (CBFYR0637) (CBMAB-R0637-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






