BMPR1A Antibodies
Background
The BMPR1A gene encodes the bone morphogenetic protein receptor 1A, which is a transmembrane serine/threonine kinase receptor that plays a crucial role in cell signal transduction. This protein is mainly present on the cell surface of various tissues and activates the SMAD protein-dependent signaling pathway by specifically binding to bone morphogenetic proteins, thereby regulating important physiological processes such as cell proliferation, differentiation, and apoptosis. During embryonic development, the signal transduction mediated by BMPR1A is particularly crucial for achieving bone formation, neural tube closure, and the normal development of internal organs. Studies have shown that mutations in the BMPR1A gene are closely related to various genetic diseases such as juvenile polyposis syndrome, and the abnormal function of this gene may lead to the formation of gastrointestinal polyps and an increased susceptibility to malignant tumors. As an important member of the TGF-β superfamily receptor, the structural and functional studies of BMPR1A have deepened our understanding of developmental biology and disease occurrence mechanisms, and have provided a theoretical basis for clinical diagnosis and targeted treatment of related diseases.
Structure of BMPR1A
The BMPR1A gene encodes receptor IA for bone morphogenetic protein, with a molecular weight of approximately 55 kDa. There are sequence differences among different species, but the structure is highly conserved. The intracellular region of this receptor has serine/threonine kinase activity and transmits signals by phosphorylating SMAD proteins. The receptor is composed of a signal peptide, an extracellular ligand-binding domain, a transmembrane region, and an intracellular kinase domain. The extracellular region contains 10 cysteine residues that form a conserved disulfide bond structure, maintaining ligand recognition specificity. The intracellular kinase domain contains a characteristic glycine-rich loop and a conserved catalytic core sequence. The mutation of the arginine residue at position 229 leads to juvenile polyposis, and the phosphorylation of threonine at position 387 is a key switch for signal activation. After binding to the ligand, this receptor forms an heterotrimer and regulates downstream signal cascades through the GS domain.
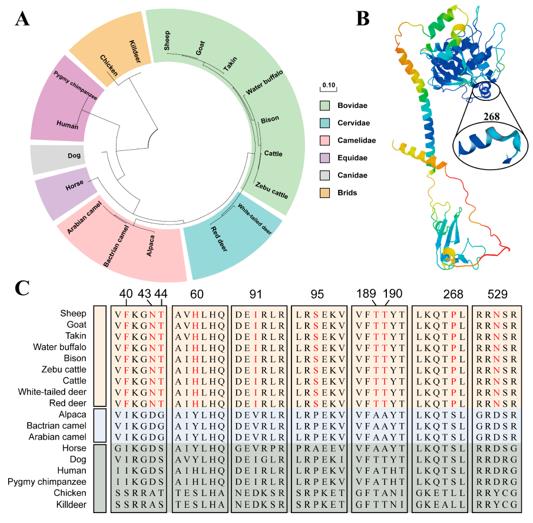 Fig. 1 Evolution, Structure, and Horn-Specific Sites of BMPR1A.1
Fig. 1 Evolution, Structure, and Horn-Specific Sites of BMPR1A.1
Key structural properties of BMPR1A:
- The extracellular region contains 10 conserved cysteines forming disulfide bonds
- The transmembrane region is a single transmembrane structure
- The intracellular region has a serine/threonine kinase domain
- The GS domain contains the characteristic sequence SGSGS, which regulates the phosphorylation of downstream SMAD proteins
- The kinase activity depends on the ATP binding site, and the 277th lysine is crucial
Functions of BMPR1A
The BMPR1A gene encodes the bone morphogenetic protein receptor IA, which plays multiple roles in cellular signal transduction:
| Function | Description |
| Bone development | Mediates BMP signaling to regulate osteoblast differentiation and bone formation. |
| Ectopic pattern formation | Participates in the establishment of the embryonic body axis and organogenesis in the early stage of the embryo. |
| Cell proliferation regulation | Regulates the cell cycle and cell proliferation through the SMAD pathway. |
| Cell differentiation induction | Regulates the fate determination of various cell types. |
| Tumor suppression | Maintains the homeostasis of intestinal stem cells, and mutations can cause juvenile polyposis. |
This receptor forms an heterotrimeric complex by recognizing BMP ligands. The signal intensity depends on the phosphorylation status of the intracellular GS domain. Unlike activin receptors, BMPR1A mainly phosphorylates SMAD1/5/8 proteins.
Applications of BMPR1A and BMPR1A Antibody in Literature
1. Zhang, Guoqing, et al. "Expression, polymorphism, and potential functional sites of the BMPR1A gene in the sheep horn." Genes 15.3 (2024): 376. https://doi.org/10.3390/genes15030376
This study reveals that the BMPR1A gene is closely related to the horn type of sheep: its expression is significantly higher in normal horned individuals than in short-horned sheep. Moreover, 43 SNPs and 10 ASE loci were found to potentially affect the size and presence of horns, providing molecular markers for horn type breeding.
2. Papadopulos, M. E., J. P. Plazzer, and F. A. Macrae. "Genotype–phenotype correlation of BMPR1a disease causing variants in juvenile polyposis syndrome." Hereditary Cancer in Clinical Practice 21.1 (2023): 12. https://doi.org/10.1186/s13053-023-00255-3
The study found that the pathogenic mutations of BMPR1a mainly cause colorectal polyps and malignant tumors, while gastric lesions are rare. It is recommended that mutation carriers only need to monitor colorectal conditions and do not require routine gastric screening, providing new basis for clinical management.
3. Zhang, Dali, et al. "Targeting local lymphatics to ameliorate heterotopic ossification via FGFR3-BMPR1a pathway." Nature communications 12.1 (2021): 4391. https://doi.org/10.1038/s41467-021-24643-2
The study found that FGFR3 regulates the formation of lymphatic endothelial cells through the BMPR1a signal, and affects post-traumatic ectopic ossification. Col2+ cells can differentiate into lymphatic endothelial cells. Activating FGFR3 to promote lymphatic vessel formation can inhibit ectopic bone formation, providing a new target for treatment.
4. Hochgerner, Mathias, et al. "BMPR1a is required for the optimal TGFβ1-dependent CD207+ langerhans cell differentiation and limits skin inflammation through CD11c+ cells." Journal of Investigative Dermatology 142.9 (2022): 2446-2454. https://doi.org/10.1016/j.jid.2022.02.014
The study found that TGFβ1 induces the differentiation of Langerhans cells through the BMPR1a signal and upregulates the expression of CD207. The absence of BMPR1a affects the development of LCs, but in psoriasis, it can inhibit the activation and migration of LCs, alleviate skin inflammation, and reveal its dual role in immune homeostasis.
5. Jiang, Bo, et al. "Lysosomal protein transmembrane 5 promotes lung-specific metastasis by regulating BMPR1A lysosomal degradation." Nature communications 13.1 (2022): 4141. https://doi.org/10.1038/s41467-022-31783-6
The study found that LAPTM5 recruits WWP2 to degrade BMPR1A, blocking the pulmonary BMP signal and promoting the stem cell characteristics of renal cancer and pulmonary-specific metastasis. LAPTM5 can serve as a predictor of lung metastasis and a therapeutic target.
Creative Biolabs: BMPR1A Antibodies for Research
Creative Biolabs specializes in the production of high-quality BMPR1A antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom BMPR1A Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our BMPR1A antibodies, custom preparations, or technical support, contact us at email.
Reference
- Zhang, Guoqing, et al. "Expression, polymorphism, and potential functional sites of the BMPR1A gene in the sheep horn." Genes 15.3 (2024): 376. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/genes15030376
Anti-BMPR1A antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-Acetyl-α-Tubulin (Lys40) Recombinant Antibody (V2-623485) (CBMAB-CP2897-LY)

-
Rat Anti-ADGRE4 Recombinant Antibody (V2-160163) (CBMAB-F0011-CQ)

-
Mouse Anti-F11R Recombinant Antibody (402) (CBMAB-0026-WJ)

-
Mouse Anti-FOSB Recombinant Antibody (CBXF-3593) (CBMAB-F2522-CQ)

-
Rabbit Anti-B2M Recombinant Antibody (CBYY-0059) (CBMAB-0059-YY)

-
Mouse Anti-AHCYL1 Recombinant Antibody (V2-180270) (CBMAB-A1703-YC)

-
Mouse Anti-CD8 Recombinant Antibody (C1083) (CBMAB-C1083-LY)

-
Rat Anti-ABCC11 Recombinant Antibody (V2-179001) (CBMAB-A0236-YC)

-
Mouse Anti-BZLF1 Recombinant Antibody (BZ.1) (CBMAB-AP705LY)

-
Mouse Anti-ENO2 Recombinant Antibody (85F11) (CBMAB-0276CQ)

-
Mouse Anti-APOA1 Monoclonal Antibody (CBFYR0637) (CBMAB-R0637-FY)

-
Mouse Anti-DMD Recombinant Antibody (D1190) (CBMAB-D1190-YC)

-
Mouse Anti-ATP1B1 Recombinant Antibody (E4) (CBMAB-0463-LY)

-
Mouse Anti-ACLY Recombinant Antibody (V2-179314) (CBMAB-A0610-YC)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Mouse Anti-BANF1 Recombinant Antibody (3F10-4G12) (CBMAB-A0707-LY)

-
Mouse Anti-GFAP Recombinant Antibody (5) (CBMAB-G0346-LY)

-
Mouse Anti-BIRC3 Recombinant Antibody (16E63) (CBMAB-C3367-LY)

-
Mouse Anti-ALB Recombinant Antibody (V2-55272) (CBMAB-H0819-FY)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






