AMD1 Antibodies
Background
The AMD1 gene encodes S-adenosylmethionine decarboxylase, which is a key enzyme in the polyamine synthesis pathway, catalyzing the production of spermidine and spermine. This gene is highly expressed in various tumors and affects cell proliferation, differentiation, and stem cell maintenance by regulating polyamine metabolism. Its expression is regulated by promoter methylation, gene amplification, and transcription factors such as Sox10, and in some fungi, it requires A-to-I RNA editing to acquire function. AMD1 variations are associated with childhood obesity, suggesting its potential role in metabolic regulation. As a core gene in polyamine metabolism, AMD1 has become an important target for tumor treatment and metabolic disease research.
Structure of AMD1
The protein molecule encoded by the AMD1 gene has a molecular weight of approximately 38 kDa. The size of its monomer varies slightly among different species due to differences in amino acid sequences.
| Species | Human | Mouse | Rat | Yeast | Arabidopsis |
| Molecular Weight (kDa) | 38.3 | 38.0 | 38.1 | 40.2 | 36.8 |
| Primary Structural Differences | Contains PEST degradation signal | High homology with humans | Highly conserved catalytic domain | Presence of pre-mRNA splicing precursor | Plant-specific regulatory sequence |
This enzyme is synthesized in the body as a precursor protein and requires self-splicing activation to form the mature α/β chain heterodimer. Its active center contains a propionyl group, which is formed by post-translational processing of serine residues and is not dependent on pyridoxal phosphate. This coenzyme binds to the substrate S-adenosylmethionine and initiates the decarboxylation reaction. The half-life of the AMD1 protein is regulated by the feedback of polyamine concentration, and an increase in intracellular polyamine levels can accelerate its degradation, forming a precise metabolic regulatory circuit.
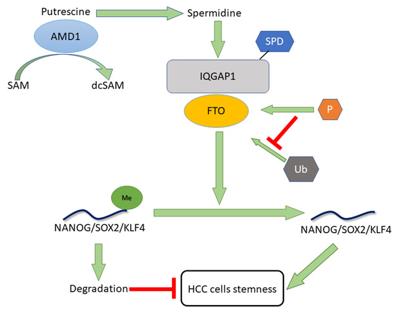 Fig. 1 Mechanisms by which AMD1 promotes hepatocellular carcinoma cell stemness.1
Fig. 1 Mechanisms by which AMD1 promotes hepatocellular carcinoma cell stemness.1
Key structural characteristics of the AMD1 protein:
- The proenzyme precursor needs to undergo self-cleavage to form an active heterodimer
- The active center contains a propionyl group, which is derived from serine
- The two subunits jointly form a substrate binding pocket
- The C-terminal PEST sequence mediates concentration-dependent degradation by polyamines
Functions of AMD1
The S-adenosylmethionine decarboxylase encoded by AMD1 is a rate-limiting enzyme in polyamine synthesis, and its function exhibits diverse metabolic regulatory capabilities.
| Function | Description |
| Polyamine synthesis | Catalyzes the decarboxylation of SAM to generate dcSAM, providing an amine propyl donor for spermidine and spermine synthesis. |
| Cell proliferation regulation | Influences nucleic acid and protein synthesis through polyamine levels, regulating the cell cycle progression. |
| Maintenance of tumor stem cells | Upregulates the expression of stemness genes such as SOX2, enhancing the self-renewal ability of tumor cells. |
| Gene expression reprogramming | Mediates the hydroxybutyrylation modification of eIF5A, activating the translation of specific mRNAs. |
| Metabolic sensing regulation | Protein stability is inhibited by polyamine feedback, constituting the intracellular metabolic sensing mechanism. |
This enzyme's catalytic reaction requires the participation of a propionyl group, and its activity is regulated at multiple levels including transcription, translation, and protein degradation. It is expressed at higher levels in rapidly proliferating cells.
Applications of Myoglobin and Myoglobin Antibody in Literature
1. Bian, Xinyu, et al. "AMD1 upregulates hepatocellular carcinoma cells stemness by FTO mediated mRNA demethylation." Clinical and translational medicine 11.3 (2021): e352. https://doi.org/10.1002/ctm2.352
The article indicates that high expression of AMD1 in hepatocellular carcinoma predicts a poor prognosis. It promotes the binding of IQGAP1 to FTO by upregulating the level of polyamines, enhances the stability and mRNA demethylation ability of FTO, thereby inducing the expression of stem cell genes and enhancing the tumor stemness, making it a potential therapeutic target.
2. Liao, Ruocen, et al. "AMD1 promotes breast cancer aggressiveness via a spermidine-eIF5A hypusination-TCF4 axis." Breast Cancer Research 26.1 (2024): 70. https://doi.org/10.1186/s13058-024-01825-6
The article indicates that in basal-like breast cancer, the overexpression of AMD1 is achieved through amplification, hypomethylation and Sox10 transcriptional activation. It promotes the production of spermidine, enhances the hydroxybutyrylation of eIF5A, activates TCF4 translation, and drives tumor progression, suggesting a poor prognosis and serving as a potential therapeutic target.
3. Cao, Shulin, et al. "RNA editing of the AMD1 gene is important for ascus maturation and ascospore discharge in Fusarium graminearum." Scientific reports 7.1 (2017): 4617. https://doi.org/10.1038/s41598-017-04960-7
The article indicates that the AMD1 gene of Fusarium graminearum corrects the stop codon through A-to-I RNA editing and encodes a MFS domain protein. This protein is located on the ascospore membrane and maintains the integrity of the ascospore wall. The editing event is crucial for the release of sexual ascospores.
4. Piloto, Ana Margarida, et al. "Plastic antibodies tailored on quantum dots for an optical detection of myoglobin down to the femtomolar range." Scientific reports 8.1 (2018): 4944. https://doi.org/10.1371/journal.pone.0033162
The article indicates that the first study on childhood obesity in India has found that the rs2796749 variation of the AMD1 gene in the homocysteine metabolism pathway is significantly associated with obesity, and is also related to body fat percentage and plasma leptin levels, serving as an early indicator of childhood obesity.
5. Hollensted, Mette, et al. "Common variants in LEPR, IL6, AMD1, and NAMPT do not associate with risk of juvenile and childhood obesity in Danes: a case–control study." BMC medical genetics 16.1 (2015): 105. https://doi.org/10.1186/s12881-015-0253-3
The article indicates that the Danish children study failed to verify the association between the four genetic variations (AMD1, etc.) found in Indian children and obesity. This suggests that this association may have racial specificity and needs to be further verified in different populations.
Creative Biolabs: AMD1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality AMD1 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom AMD1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our AMD1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Bian, Xinyu, et al. "AMD1 upregulates hepatocellular carcinoma cells stemness by FTO mediated mRNA demethylation." Clinical and translational medicine 11.3 (2021): e352. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1002/ctm2.352
Anti-AMD1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ENO2 Recombinant Antibody (85F11) (CBMAB-0276CQ)

-
Mouse Anti-EGR1 Recombinant Antibody (CBWJZ-100) (CBMAB-Z0289-WJ)

-
Mouse Anti-AFDN Recombinant Antibody (V2-58751) (CBMAB-L0408-YJ)

-
Mouse Anti-BAD (Phospho-Ser136) Recombinant Antibody (CBYY-0138) (CBMAB-0139-YY)

-
Mouse Anti-AGK Recombinant Antibody (V2-258056) (CBMAB-M0989-FY)

-
Mouse Anti-ENO1 Recombinant Antibody (CBYC-A950) (CBMAB-A4388-YC)

-
Mouse Anti-ACTN4 Recombinant Antibody (V2-6075) (CBMAB-0020CQ)

-
Mouse Anti-C5AR1 Recombinant Antibody (R63) (CBMAB-C9553-LY)

-
Mouse Anti-dsRNA Recombinant Antibody (2) (CBMAB-D1807-YC)

-
Mouse Anti-CD1C Recombinant Antibody (L161) (CBMAB-C2173-CQ)

-
Mouse Anti-ALB Recombinant Antibody (V2-180650) (CBMAB-A2186-YC)

-
Rabbit Anti-CAMK2A Recombinant Antibody (BA0032) (CBMAB-0137CQ)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

-
Mouse Anti-GLP1R Recombinant Antibody (4F3) (CBMAB-G0521-LY)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7697) (CBMAB-1869CQ)

-
Mouse Anti-EIF4G1 Recombinant Antibody (2A9) (CBMAB-A2544-LY)

-
Mouse Anti-DISP2 Monoclonal Antibody (F66A4B1) (CBMAB-1112CQ)

-
Rat Anti-EMCN Recombinant Antibody (28) (CBMAB-E0280-FY)

-
Rat Anti-ABCC11 Recombinant Antibody (V2-179001) (CBMAB-A0236-YC)

-
Mouse Anti-CAT Recombinant Antibody (724810) (CBMAB-C8431-LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






