TREX1 Antibodies
Background
TREX1 is a 3'→5' exonuclease located in the cytoplasm and is mainly present in various tissue cells of mammals. The protein encoded by this gene can specifically degrade double-stranded DNA in the cytoplasm, maintaining genomic stability and preventing abnormal activation of autoimmune responses. When TREX1 function is lost, the accumulated DNA fragments within cells will trigger abnormal activation of the type I interferon pathway, leading to the occurrence of the autoimmune disease Aicardi-Goutieres syndrome. This gene was first clearly defined by researchers in 2006 for its relationship with human diseases. The discovery of its pathogenic mechanism not only deepened our understanding of the association between nucleic acid metabolism and immune homeostasis, but also provided an important molecular target for the diagnosis and treatment research of autoimmune diseases such as systemic lupus erythematosus.
Structure of TREX1
TREX1 is an exonuclease with a molecular weight of approximately 33.8 kDa. Its molecular weight is highly conserved among different mammals, which is in line with its key cellular functions.
| Species | Human | Mouse | Rat | Bovine |
| Molecular Weight (kDa) | 33.8 | 33.9 | 33.7 | 33.8 |
| Primary Structural Differences | 314 amino acids, N-terminal catalytic domain | Highly homologous, with consistent catalytic activity | The core functional areas are highly conservative | High similarity to human sequence |
This protein is composed of 314 amino acids and its three-dimensional structure contains a typical DNase I folding catalytic core. The center of this structure has a magnesium ion binding site, which is crucial for hydrolyzing the 3' end of double-stranded DNA. Its active site is composed of highly conserved acidic amino acid residues, which are responsible for coordinating metal ions and catalyzing the hydrolysis of phosphodiester bonds. This streamlined and efficient structural design makes it the main enzyme for degrading genome-long double-stranded DNA in the cytoplasm of mammalian cells.
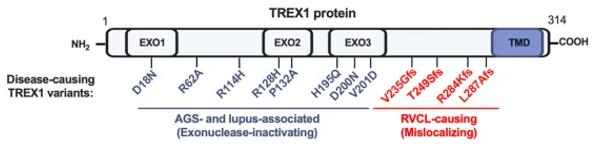 Fig. 1 Domain-Specific TREX1 Variants: Linking RVCL and Interferonopathies.1
Fig. 1 Domain-Specific TREX1 Variants: Linking RVCL and Interferonopathies.1
Key structural properties of TREX1:
- Typical DNase I folded domain
- Conserved Mg/Mn ion binding catalytic site
- 3' → 5' exonuclease activity center
- The dimerization interface is crucial for its full functionality
Functions of TREX1
The core function of the TREX1 gene is to degrade nucleic acids in the cytoplasm to maintain genomic stability. However, it is also widely involved in various pathophysiological processes, especially in the regulation of autoimmune responses.
| Function | Description |
| DNA clearance | Specifically degrade the DNA derived from retrotransposons in the cytoplasm and the DNA fragments leaked from the genome to prevent their abnormal accumulation. |
| Maintenance of immune homeostasis | By eliminating its nucleic acid to prevent cGAS - STING innate immune pathways activated by continuous abnormal, is the key of immune tolerance. |
| Genomic stability | Timely remove the erroneous or redundant nucleic acid components generated during DNA replication and repair to maintain the integrity of cellular genetic material. |
| Mechanism of disease occurrence | Losing its function sex mutation directly lead to Aicardi - Goutieres syndrome, familial chilblain lupus disease type I interferon. |
| Association with cancer development | Abnormal expression in some tumor cells may be involved in cancer progression by affecting genome stability and tumor microenvironment. |
The degradation of double-stranded DNA by TREX1 exhibits continuous and non-synergistic enzymatic kinetic characteristics, which is in line with its role positioning as a "scavenger" rather than a "regulator", ensuring the efficient removal of substrates at different concentrations.
Applications of TREX1 and TREX1 Antibody in Literature
1. Vanpouille-Box, Claire, et al. "DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity." Nature communications 8.1 (2017): 15618. https://doi.org/10.1038/ncomms15618
Studies have shown that Trex1 is induced by high doses (>12-18 Gy) during radiotherapy. It weakens tumor immunogenicity by degrading cytoplasmic DNA, inhibits the cGAS-STING pathway and interferon -β production, affects the activation of dendritic cells and the initiation of CD8⁺T cells, thereby weakening the distal anti-tumor effect. It is suggested that Trex1 can be used as a potential indicator for combined radiotherapy dose and immunotherapy.
2. Karasu, Mehmet E., et al. "Removal of TREX1 activity enhances CRISPR–Cas9-mediated homologous recombination." Nature Biotechnology 43.7 (2025): 1168-1176. https://doi.org/10.1038/s41587-024-02356-3
Research has found that the DNA exonuctase TREX1 is a key inhibitor of CRISPR-Cas9 homologous directed repair (HDR). It significantly reduces HDR efficiency by degrading single-stranded or linear double-stranded DNA templates. TREX1 is highly expressed in various cells and can serve as a negative biomarker for HDR efficiency. Knocking out TREX1 or using chemically protected DNA templates can effectively enhance the efficiency of HDR editing.
3. Chauvin, Samuel D., et al. "Inherited C-terminal TREX1 variants disrupt homology-directed repair to cause senescence and DNA damage phenotypes in Drosophila, mice, and humans." Nature communications 15.1 (2024): 4696. https://doi.org/10.1038/s41467-024-49066-7
Research has found that mutations at the C-terminal of the TREX1 gene can cause it to be truncated and wrongly located in the nucleus. This abnormal TREX1 can cause DNA damage and inhibit homologous recombination repair, thereby accelerating cellular aging and leading to retinal and small vessel lesions in the brain. This provides a mechanistic association between the DNA damage theory and aging-related vascular diseases.
4. Murayama, Takahiko, et al. "Targeting TREX1 induces innate immune response in drug-resistant small-cell lung cancer." Cancer Research Communications 4.9 (2024): 2399-2414. https://doi.org/10.1158/2767-9764.CRC-24-0360
Research has found that TREX1 is upregulated in chemotherapy-resistant small cell lung cancer. It reduces tumor immunogenicity by eliminating double-stranded DNA in the cytoplasm and inhibiting the cGAS-STING innate immune pathway. Targeted inhibition of TREX1 can activate the immune response and sensitize chemotherapy, providing a new strategy for the treatment of such "immune cold tumors".
5. Técher, Hervé, et al. "MRE11 and TREX1 control senescence by coordinating replication stress and interferon signaling." Nature Communications 15.1 (2024): 5423. https://doi.org/10.1038/s41467-024-49740-w
Studies have shown that in oncogene-induced cellular senescence, the cytoplasmic DNase TREX1 plays a key regulatory role. Overexpression of TREX1 can clear cytoplasmic DNA, thereby inhibiting Cgas-STING-mediated type I interferon response and preventing the aging process. This reveals a new mechanism by which the interferon response amplifies the DNA damage response through a positive feedback loop to promote cellular senescence.
Creative Biolabs: TREX1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality TREX1 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom TREX1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our TREX1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Chauvin, Samuel D., et al. "Inherited C-terminal TREX1 variants disrupt homology-directed repair to cause senescence and DNA damage phenotypes in Drosophila, mice, and humans." Nature communications 15.1 (2024): 4696. https://doi.org/10.1038/s41467-024-49066-7
Anti-TREX1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-EGR1 Recombinant Antibody (CBWJZ-100) (CBMAB-Z0289-WJ)

-
Mouse Anti-AK4 Recombinant Antibody (V2-180419) (CBMAB-A1891-YC)

-
Mouse Anti-FLT1 Recombinant Antibody (11) (CBMAB-V0154-LY)

-
Mouse Anti-AKT1 Recombinant Antibody (V2-180546) (CBMAB-A2070-YC)

-
Rabbit Anti-CCL5 Recombinant Antibody (R0437) (CBMAB-R0437-CN)

-
Rat Anti-EPO Recombinant Antibody (16) (CBMAB-E1578-FY)

-
Rat Anti-ABCC11 Recombinant Antibody (V2-179001) (CBMAB-A0236-YC)

-
Mouse Anti-ARG1 Recombinant Antibody (CBYCL-103) (CBMAB-L0004-YC)

-
Mouse Anti-AMH Recombinant Antibody (5/6) (CBMAB-A2527-YC)

-
Mouse Anti-CHRNA9 Recombinant Antibody (8E4) (CBMAB-C9161-LY)

-
Mouse Anti-DMPK Recombinant Antibody (CBYCD-324) (CBMAB-D1200-YC)

-
Mouse Anti-CTCF Recombinant Antibody (CBFYC-2371) (CBMAB-C2443-FY)

-
Mouse Anti-dsDNA Recombinant Antibody (22) (CBMAB-AP1954LY)

-
Mouse Anti-BRD3 Recombinant Antibody (CBYY-0801) (CBMAB-0804-YY)

-
Mouse Anti-BACE1 Recombinant Antibody (61-3E7) (CBMAB-1183-CN)

-
Mouse Anti-APP Recombinant Antibody (DE2B4) (CBMAB-1122-CN)

-
Mouse Anti-ENO2 Recombinant Antibody (85F11) (CBMAB-0276CQ)

-
Mouse Anti-GGT1 Recombinant Antibody (1F9) (CBMAB-G3273-LY)

-
Mouse Anti-ATP1B3 Recombinant Antibody (1E9) (CBMAB-A4021-YC)

-
Mouse Anti-CD63 Recombinant Antibody (CBXC-1200) (CBMAB-C1467-CQ)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






