OPRM1 Antibodies
Background
The OPRM1 gene encodes the μ-opioid receptor, which is a G protein-coupled receptor mainly distributed in the central nervous system and peripheral tissues. This receptor participates in regulating various physiological processes such as pain perception, stress response, and reward mechanisms after binding to endogenous opioid peptides and exogenous opioid drugs (such as morphine). It plays a key role in mediating analgesic effects and the formation of addiction, making it a key target in pain treatment and drug dependence research. In 1993, a research team successfully cloned this gene for the first time. Subsequently, through functional analysis and structural biology studies, the molecular mechanism of its interaction with ligands was gradually revealed. In-depth research on the polymorphisms of the OPRM1 gene not only helps explain the differences in pain sensitivity and the efficacy of opioid drugs among individuals, but also provides an important genetic basis for the formulation of personalized pain management and addiction prevention strategies.
Structure of OPRM1
The μ-opioid receptor protein encoded by the OPRM1 gene has a molecular weight of approximately 44-45 kDa. This value is relatively conserved across different species, but there are also some variations.
| Species | Human | Mouse | Rat | Pig | Monkey |
| Molecular Weight (kDa) | 44.8 | 44.7 | 44.8 | 45.0 | 44.8 |
| Primary Structural Differences | Contains 4 N-glycosylation sites, with cysteine bridges in the extracellular loop | Homologous to the human sequence at 95%, with a slight difference in the C-terminal intracellular region | Highly conserved transmembrane domain, with the same ligand binding site | Different N-terminal glycosylation pattern, affecting the receptor maturation process | Non-human primate model, with the sequence most similar to the human source |
The μ-opioid receptor encoded by the OPRM1 gene consists of approximately 400 amino acids and forms a typical seven-transmembrane structure. This receptor belongs to the G-protein-coupled receptor family. Its protein structure is composed of the extracellular N-terminal, seven transmembrane α-helices, three extracellular loops, three intracellular loops, and the intracellular C-terminal. The extracellular region contains multiple glycosylation sites, which are involved in the folding and membrane localization of the receptor; the transmembrane domains form the ligand-binding pocket, which recognizes endogenous opioid peptides and exogenous opioid drugs. The third intracellular loop is the key region for G-protein coupling, responsible for signal transduction; the C-terminal intracellular region contains phosphorylation sites, which are involved in receptor desensitization and endocytosis regulation. This exquisite structural design enables the μ-opioid receptor to precisely mediate the analgesic effect and reward response.
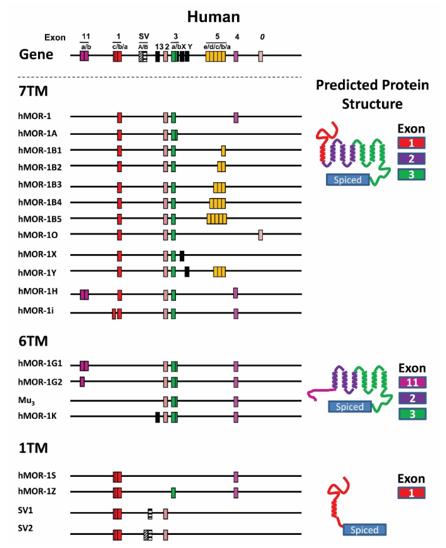 Fig. 1 Schematic of the human OPRM1 gene structure and alternative splicing.1
Fig. 1 Schematic of the human OPRM1 gene structure and alternative splicing.1
Key structural properties of OPRM1:
- A typical seven-transmembrane structure
- Exocytic N-terminal glycosylation modification
- A ligand-binding pocket located in the transmembrane domain
- An intracellular loop that couples with G proteins and mediates signal transduction
Functions of OPRM1
The main function of the OPRM1 gene is to mediate the analgesic and rewarding effects of opioid substances. However, this receptor is also involved in regulating various physiological processes, including stress responses, emotional regulation, and immune functions.
| Function | Description |
| Analgesic effect | After binding to μ-opioid receptors, opioids inhibit the transmission of pain signals and produce analgesic effects. |
| Reward regulation | Activating the dopaminergic pathway, mediating feelings of pleasure and rewarding effects, and participating in the formation of addiction. |
| Respiratory inhibition | Activating receptors in the brainstem respiratory center can lead to a decrease in respiratory rate, which is a major side effect of opioids. |
| Gastrointestinal function | Inhibiting gastrointestinal peristalsis, causing constipation, which is a common adverse reaction of opioids. |
| Immune regulation | Expressing on the surface of immune cells, participating in the regulation of inflammatory responses and immune functions. |
The binding of the μ-opioid receptor encoded by the OPRM1 gene to its ligand exhibits typical saturation kinetic characteristics. The binding curve is a rectangular hyperbola, which is different from the linear binding pattern of ligand-gated ion channels. This reflects the signal amplification effect produced after the receptor is coupled to G proteins.
Applications of OPRM1 and OPRM1 Antibody in Literature
1. Liu, Shan, et al. "Alternative pre-mRNA splicing of the mu opioid receptor gene, OPRM1: insight into complex mu opioid actions." Biomolecules 11.10 (2021): 1525. https://doi.org/10.3390/biom11101525
This review summarizes the three types of variants generated by alternative splicing of the OPRM1 gene, which play a significant role in the pharmacology of opioids. Targeting these variants may lead to the development of new, potent analgesics with fewer side effects.
2. Qian, Cheng, et al. "Intercellular communication atlas reveals Oprm1 as a neuroprotective factor for retinal ganglion cells." Nature communications 15.1 (2024): 2206. https://doi.org/10.1038/s41467-024-46428-z
This study, through single-cell RNA sequencing, revealed that highly surviving retinal ganglion cells have more ligand-receptor interactions with adjacent cells. Among them, the μ-opioid receptor (Oprm1) has neuroprotective effects, and activating it can improve the visual function of mice.
3. Haupt, Larisa M., et al. "The effects of OPRM1 118A> G on methadone response in pain management in advanced cancer at end of life." Scientific Reports 14.1 (2024): 3411. https://doi.org/10.1038/s41598-024-54009-9
This study investigated the effect of the OPRM1 gene A118G polymorphism on the treatment of cancer pain with methadone. No significant correlation was found between this variation and the drug dosage or pain score. However, significant racial differences were observed: carriers of this variant in Chinese patients required a higher dose of opioids.
4. Hsu, Pei-Shan, et al. "OPRM1 A118G polymorphism modulating motor pathway for pain adaptability in women with primary dysmenorrhea." Frontiers in Neuroscience 17 (2023): 1179851. https://doi.org/10.3389/fnins.2023.1179851
The study found that the A118G polymorphism of the OPRM1 gene affects the white matter structure of patients with primary dysmenorrhea. Individuals carrying the G allele showed structural changes in brain regions such as the corpus callosum during menstruation, and these changes were associated with the pattern of pain perception differently from the AA genotype.
5. Boroń, Agnieszka, et al. "OPRM1 gene polymorphism in women with alcohol use disorder." International Journal of Molecular Sciences 25.5 (2024): 3067. https://doi.org/10.3390/ijms25053067
This study is the first to reveal that the rs1074287 polymorphism of the OPRM1 gene is associated with the openness personality trait of female alcohol-dependent individuals. There are significant differences in the openness levels between different genotypes and the control group, indicating that personality traits may play an important role in the addiction mechanism.
Creative Biolabs: OPRM1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality OPRM1 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom OPRM1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our OPRM1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Liu, Shan, et al. "Alternative pre-mRNA splicing of the mu opioid receptor gene, OPRM1: insight into complex mu opioid actions." Biomolecules 11.10 (2021): 1525. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/biom11101525
Anti-OPRM1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-AGO2 Recombinant Antibody (V2-634169) (CBMAB-AP203LY)

-
Mouse Anti-CD46 Recombinant Antibody (CBFYC-0076) (CBMAB-C0085-FY)

-
Mouse Anti-AMH Recombinant Antibody (5/6) (CBMAB-A2527-YC)

-
Mouse Anti-HTLV-1 gp46 Recombinant Antibody (CBMW-H1006) (CBMAB-V208-1154-FY)

-
Mouse Anti-BIRC7 Recombinant Antibody (88C570) (CBMAB-L0261-YJ)

-
Mouse Anti-CCDC25 Recombinant Antibody (CBLC132-LY) (CBMAB-C9786-LY)

-
Mouse Anti-APP Recombinant Antibody (5C2A1) (CBMAB-A3314-YC)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-1728) (CBMAB-2077-YY)

-
Mouse Anti-ADGRE5 Recombinant Antibody (V2-360335) (CBMAB-C2088-CQ)

-
Mouse Anti-DMPK Recombinant Antibody (CBYCD-324) (CBMAB-D1200-YC)

-
Mouse Anti-APP Recombinant Antibody (DE2B4) (CBMAB-1122-CN)

-
Mouse Anti-2C TCR Recombinant Antibody (V2-1556) (CBMAB-0951-LY)

-
Mouse Anti-ADAM29 Recombinant Antibody (V2-179787) (CBMAB-A1149-YC)

-
Mouse Anti-ALPL Antibody (B4-78) (CBMAB-1009CQ)

-
Mouse Anti-BANF1 Recombinant Antibody (3F10-4G12) (CBMAB-A0707-LY)

-
Mouse Anti-CFL1 Recombinant Antibody (CBFYC-1771) (CBMAB-C1833-FY)

-
Mouse Anti-ATM Recombinant Antibody (2C1) (CBMAB-A3970-YC)

-
Mouse Anti-BIRC5 Recombinant Antibody (6E4) (CBMAB-CP2646-LY)

-
Mouse Anti-FLI1 Recombinant Antibody (CBXF-0733) (CBMAB-F0435-CQ)

-
Mouse Anti-ENO1 Recombinant Antibody (8G8) (CBMAB-E1329-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






