DDIT3 Antibodies
Background
DDIT3 is a stress-inducible protein that functions as a transcription factor. It is mainly highly expressed in cells when they are responding to endoplasmic reticulum stress. This protein regulates downstream gene expression by forming heterodimers with other members of the C/EBP family, thereby mediating growth arrest and cell apoptosis during the unfolded protein response. The expression regulation balance of DDIT3 is essential for embryonic development and the normal differentiation of various tissues and organs. When its function is dysregulated, it can promote the occurrence and development of diabetes, neurodegenerative diseases, and various cancers. Initially identified by David Ron and others from Chinese hamster ovary cells in 1988, DDIT3 was named GADD153 due to its strong induction characteristics under DNA damage and hypoxic conditions. Over the past few decades, the research on the regulatory network and pathological mechanism of this gene has continued to deepen, laying an important foundation for understanding the intracellular endoplasmic reticulum stress response and related disease intervention strategies.
Structure of DDIT3
DDIT3 is an intranuclear protein with a molecular weight of approximately 19 kDa. There are slight variations among different species due to differences in amino acid sequences.
| Species | Human | Mouse | Rat | Hamster | Pig |
| Molecular Weight (kDa) | 19.0 | 18.8 | 18.9 | 19.1 | 19.0 |
| Primary Structural Differences | Highly conservative | Individual site substitution | There is a slight variation at the C end | Similar to mice | Close to human origin |
The DDIT3 protein contains 169 amino acids and its structure includes a characteristic leucine zipper domain. This domain forms an heterodimer with members of the C/EBP family and mediates specific DNA binding. The N-terminal of the protein is a transcriptional activation domain, rich in acidic amino acids; the C-terminal is the basic region and the zipper region, responsible for dimerization and DNA binding. DDIT3 is expressed at extremely low levels under normal conditions. When endoplasmic reticulum stress occurs, its expression rapidly increases. It inhibits the transcription of genes regulated by C/EBP, causing cell cycle arrest or apoptosis. Phosphorylation modification is crucial for its transcriptional activity. Multiple sites of modification can enhance protein stability and regulate its nuclear translocation.
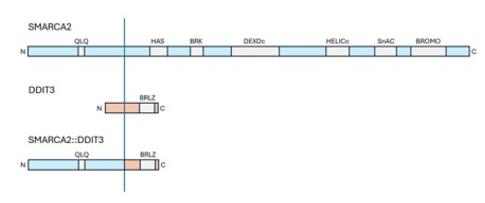 Fig. 1 Diagram of the predicted fusion protein SMARCA2::DDIT3.1
Fig. 1 Diagram of the predicted fusion protein SMARCA2::DDIT3.1
Key structural properties of DDIT3:
- Characteristic leucine zipper domain
- C-terminal basic DNA-binding domain
- N-terminal acidic transcriptional activation domain
- Dimerization interface mediates hetero-complex formation with C/EBP proteins
Functions of DDIT3
The core function of DDIT3 is to regulate cell fate under endoplasmic reticulum stress. Additionally, it is involved in various physiological processes such as adipocyte differentiation, hematopoietic regulation, and inflammatory responses.
| Function | Description |
| Apoptosis induction | DDIT3 activates pro-apoptotic genes and inhibits the expression of anti-apoptotic molecules, mediating cell death through continuous endoplasmic reticulum stress. |
| Growth arrest | By blocking cell cycle-related genes regulated by C/EBP, it causes the cell cycle to stall at the G1 phase. |
| Stress signal integration | Integrating signals from three pathways: PERK, ATF6, and IRE1, serving as the core transcriptional effector of the unfolded protein response. |
| Metabolic regulation | Inhibiting osteoblast and adipocyte differentiation, regulating the expression of genes related to liver gluconeogenesis during nutrient deficiency. |
| Embryonic development | Participating in cell differentiation and tissue remodeling during placental and neural tube development. |
The expression of DDIT3 remains at an extremely low level under normal physiological conditions, but is rapidly induced and upregulated during endoplasmic reticulum stress. Its transcriptional activation depends on the direct interaction of ATF4 and ATF6, and its protein activity is precisely regulated by phosphorylation modifications.
Applications of DDIT3 and DDIT3 Antibody in Literature
1. Shah, Ahmed, et al. "Novel SMARCA2:: DDIT3 Fusion in Primary Subcutaneous Myxoid Liposarcoma due to an Unusual Unbalanced Chromosomal Translocation." Genes, Chromosomes and Cancer 64.10 (2025): e70089. https://doi.org/10.1002/gcc.70089
This article reports a case of a 26-year-old male with a myxoid liposarcoma. The tissue morphology is typical, but there is a novel SMARCA2::DDIT3 gene fusion. The study found that this fusion retains the key functional domains of DDIT3, further confirming that the classic pathological morphology can guide molecular testing to reveal new variations.
2. Du, Mengbao, et al. "C/EBPα-p30 confers AML cell susceptibility to the terminal unfolded protein response and resistance to Venetoclax by activating DDIT3 transcription." Journal of Experimental & Clinical Cancer Research 43.1 (2024): 79. https://doi.org/10.1186/s13046-024-02975-3
The study found that the imbalance in the ratio of the two subtypes of C/EBPα (p42/p30) affects the endoplasmic reticulum stress response in AML by regulating DDIT3. Patients with a low p42/p30 ratio are resistant to BCL2 inhibitors when used alone, but this resistance can be overcome by combining with endoplasmic reticulum stress inducers.
3. Ranji, Parmida, et al. "Deciphering the role of FUS:: DDIT3 expression and tumor microenvironment in myxoid liposarcoma development." Journal of Translational Medicine 22.1 (2024): 389. https://doi.org/10.1186/s12967-024-05211-w
In this study, using a decellularized sarcoma model, it was found that the microenvironment of myxoid liposarcoma (MLS) jointly regulates the tumor phenotype with the FUS::DDIT3 fusion gene, and this fusion gene is a key factor determining its preadipocyte differentiation.
4. Berastegui, Nerea, et al. "The transcription factor DDIT3 is a potential driver of dyserythropoiesis in myelodysplastic syndromes." Nature Communications 13.1 (2022): 7619. https://doi.org/10.1038/s41467-022-35192-7
Studies have confirmed that the transcription factor DDIT3 is specifically highly expressed in hematopoietic stem cells of MDS patients and is a key factor driving the failure of red blood cell production. Targeted inhibition of DDIT3 can restore red blood cell differentiation, suggesting that it could serve as a new therapeutic target for MDS.
5. Jauhiainen, Alexandra, et al. "Distinct cytoplasmic and nuclear functions of the stress induced protein DDIT3/CHOP/GADD153." PloS one 7.4 (2012): e33208. https://doi.org/10.1371/journal.pone.0033208
The study found that the transcription factor DDIT3 regulates target genes through differential cellular localization (cytoplasmic or nuclear). Cytoplasmic DDIT3 mainly inhibits cell migration, while nuclear DDIT3 induces cell cycle arrest. Its mechanism of action depends on heterodimerization with different transcription factors.
Creative Biolabs: DDIT3 Antibodies for Research
Creative Biolabs specializes in the production of high-quality DDIT3 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom DDIT3 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our DDIT3 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Shah, Ahmed, et al. "Novel SMARCA2:: DDIT3 Fusion in Primary Subcutaneous Myxoid Liposarcoma due to an Unusual Unbalanced Chromosomal Translocation." Genes, Chromosomes and Cancer 64.10 (2025): e70089. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1002/gcc.70089
Anti-DDIT3 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-GFAP Recombinant Antibody (20) (CBMAB-G2914-LY)

-
Mouse Anti-ADAM29 Recombinant Antibody (V2-179787) (CBMAB-A1149-YC)

-
Mouse Anti-AKT1 (Phosphorylated S473) Recombinant Antibody (V2-505430) (PTM-CBMAB-0067LY)

-
Mouse Anti-BAD (Phospho-Ser136) Recombinant Antibody (CBYY-0138) (CBMAB-0139-YY)

-
Mouse Anti-FN1 Monoclonal Antibody (71) (CBMAB-1241CQ)

-
Mouse Anti-AOC3 Recombinant Antibody (CBYY-0014) (CBMAB-0014-YY)

-
Mouse Anti-ATP1B1 Recombinant Antibody (E4) (CBMAB-0463-LY)

-
Rabbit Anti-CAMK2A Recombinant Antibody (BA0032) (CBMAB-0137CQ)

-
Mouse Anti-FeLV g27 Recombinant Antibody (1) (CBMAB-V208-1714-FY)

-
Mouse Anti-APP Recombinant Antibody (5C2A1) (CBMAB-A3314-YC)

-
Mouse Anti-BRD3 Recombinant Antibody (CBYY-0801) (CBMAB-0804-YY)

-
Mouse Anti-FLT1 Recombinant Antibody (11) (CBMAB-V0154-LY)

-
Mouse Anti-CD24 Recombinant Antibody (ALB9) (CBMAB-0176CQ)

-
Mouse Anti-APOH Recombinant Antibody (4D9A4) (CBMAB-A3249-YC)

-
Mouse Anti-CARD11 Recombinant Antibody (CBFYC-0811) (CBMAB-C0866-FY)

-
Mouse Anti-APP Recombinant Antibody (DE2B4) (CBMAB-1122-CN)

-
Mouse Anti-AQP2 Recombinant Antibody (G-3) (CBMAB-A3359-YC)

-
Mouse Anti-AMIGO2 Recombinant Antibody (CBYY-C0756) (CBMAB-C2192-YY)

-
Mouse Anti-ARID3A Antibody (A4) (CBMAB-0128-YC)

-
Mouse Anti-dsRNA Recombinant Antibody (2) (CBMAB-D1807-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






