DUX4 Antibodies
Background
The DUX4 gene encodes a dual-homologous-box transcription factor, which is mainly present in human and other mammalian germ cells and during the early embryonic development stage. This protein can activate the expression of multiple downstream genes and plays a crucial regulatory role in the activation of the zygotic genome, promoting the transition of the embryo from maternal regulation to self-gene expression. In certain limb or cardiac tissues, DUX4 exhibits transient low-level expression, but its function has not yet been fully elucidated. It was first identified by Gabrielle Kardon et al. through the candidate cloning strategy in 1994, and this gene was subsequently confirmed to be directly related to facioscapulohumeral muscular dystrophy. Its abnormal expression - especially the shortening of the D4Z4 microsatellite repeat sequence in the 12q31 chromosomal region leading to ectopic activation in skeletal muscle - constitutes the main pathogenic mechanism of this disease. The continuous study of the DUX4 regulatory network and downstream target genes not only deepens our understanding of the regulatory mechanism of early embryonic development but also provides a key molecular basis for targeted treatment of facioscapulohumeral muscular dystrophy.
Structure of DUX4
The protein encoded by DUX4 has a molecular weight of approximately 47 kDa. There are differences in the molecular weight and sequence of this protein among different species.
| Species | Human | Mouse | Macaque | Bovine | Shrew |
| Molecular Weight (kDa) | 47 | 50 | 46.5 | 47.2 | 50.5 |
| Primary Structural Differences | The double homologous box structure is conserved, and the C-terminal region is rich in hydrophobic residues | The C-terminal sequence is significantly different from that of humans | High homology with humans | The sequence is long and there are inserted fragments | The homologous box structure has significant variations |
The DUX4 protein consists of approximately 424 amino acids. Its N-terminal region contains two tandem homologous domains that mediate sequence-specific DNA binding. The C-terminal region of the protein contains a transcriptional activation domain, which is responsible for recruiting transcriptional co-activators. This protein is transiently expressed in normal tissues only in the testis, thymus, and preimplantation embryos, but is abnormally persistently expressed in the skeletal muscles of patients with facioscapulohumeral muscular dystrophy. The DNA binding domain of DUX4 is composed of a helix-ring-helix-turn-helix structure, and the second homologous box is similar to the structure of the PAX3/7 family of transcription factors. This protein mainly interacts with histone acetyltransferases such as p300/CBP through its hydrophobic region at the C-terminal, initiating the transcription of downstream genes.
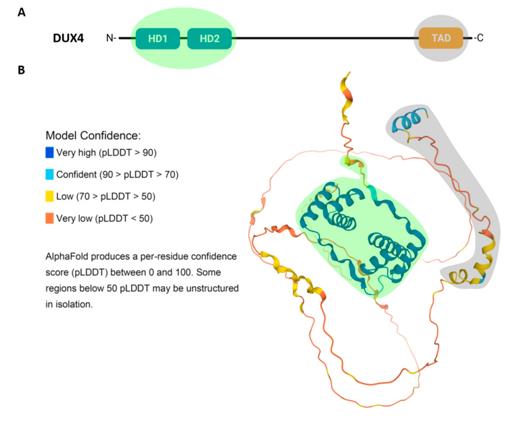 Fig. 1 Structure and Predicted 3D Conformation of the DUX4 Protein.1
Fig. 1 Structure and Predicted 3D Conformation of the DUX4 Protein.1
Key structural properties of DUX4:
- The N terminus contains two tandem homology domains, which mediate DNA sequence specific recognition
- The central area is rich in proline and serves as the flexible hinge zone
- The C-terminal is the transcriptional activation domain, containing hydrophobic heptapeptide repeats
- The DNA binding domain adopts a helix-turn-helix-ring-helix folding pattern
- The second homologous domain is highly similar in structure to the PAX protein
- The activation region recruits co-activators such as p300/CBP through hydrophobic interaction
Functions of DUX4
The main function of DUX4 is to act as a transcription factor to activate the expression of downstream genes. However, it also plays multiple regulatory roles in different physiological and pathological conditions.
| Function | Description |
| Zygote genome activation | DUX4 is transiently expressed in the 2-cell stage of the embryo, activating the transcription of the zygote genome and initiating the embryonic autonomous development program. |
| Reproductive cell expression | It is expressed at a low level in testicular germ cells, possibly participating in gametogenesis or maintaining the homeostasis of germ cells. |
| Duchenne/Becker muscular dystrophy pathogenesis | It is abnormally expressed in skeletal muscles, activating a large number of downstream genes, leading to cytotoxicity, oxidative stress and cell apoptosis. |
| Revertendo transposon regulation | DUX4 can activate the endogenous retroviral elements of the HERV-L family, affecting the genome structure and expression regulation. |
| Stem cell pluripotency regulation | It is transiently expressed in the early stage of induced pluripotent stem cells, assisting somatic cells in reprogramming to a pluripotent state. |
The downstream target genes of DUX4 are extensive, including various genes related to embryonic development among different species, genes specific to germ cells, and genes related to apoptosis. Its pathogenic expression shows high toxicity, activating immune-related pathways and interfering with myogenic differentiation.
Applications of DUX4 and DUX4 Antibody in Literature
1. Mocciaro, Emanuele, et al. "DUX4 role in normal physiology and in FSHD muscular dystrophy." Cells 10.12 (2021): 3322. https://doi.org/10.3390/cells10123322
The article indicates that the transcription factor DUX4 plays a crucial role in early embryonic development. Abnormal expression of DUX4 can lead to various diseases such as cancer, viral infections, and facioscapulohumeral muscular dystrophy (FSHD). Understanding the regulatory mechanism of DUX4 is of great significance for studying embryonic development and developing therapies for related diseases.
2. Duranti, Elisa, and Chiara Villa. "Influence of DUX4 expression in facioscapulohumeral muscular dystrophy and possible treatments." International Journal of Molecular Sciences 24.11 (2023): 9503. https://doi.org/10.3390/ijms24119503
The article indicates that facioscapulohumeral muscular dystrophy (FSHD) is mainly caused by abnormal expression of the transcription factor DUX4. DUX4 is wrongly activated in muscle cells, interfering with the muscle formation pathway and causing cytotoxicity. This article reviews its pathogenic mechanism and explores emerging therapeutic strategies targeting DUX4 expression.
3. Bakewell, Jack, Anthony V. Moorman, and Sarra L. Ryan. "DUX4-rearranged B-ALL: deciphering a biological and clinical conundrum: ACUTE LYMPHOBLASTIC LEUKEMIA." Leukemia 39.12 (2025): 2835-2847. https://doi.org/10.1038/s41375-025-02758-5
The article indicates that DUX4 gene rearrangement (DUX4-r) is a unique subtype of B-cell acute lymphoblastic leukemia, accounting for 5-10%. It leads to abnormal protein expression and hinders B-cell development. Recent sequencing and immunophenotyping techniques have improved its detection rate, which is beneficial for improving diagnosis, treatment and clinical prognosis.
4. Belayew, Alexandra, Alberto L. Rosa, and Peter S. Zammit. "DUX4 at 25: how it emerged from "junk DNA" to become the cause of facioscapulohumeral muscular dystrophy." Skeletal Muscle 15.1 (2025): 24. https://doi.org/10.1186/s13395-025-00388-0
The article indicates that the abnormal expression of the transcription factor DUX4 is the key pathogenic mechanism leading to facioscapulohumeral muscular dystrophy (FSHD). It is usually epigenetically suppressed, but is wrongly activated in FSHD, causing muscle toxicity. This article reviews the 25-year research history of DUX4 from a "pseudo-gene" to a core therapeutic target.
5. Sidlauskaite, Eva, et al. "DUX4 expression in FSHD muscles: Focus on its mRNA regulation." Journal of Personalized Medicine 10.3 (2020): 73. https://doi.org/10.3390/jpm10030073
The article indicates that the core cause of facioscapulohumeral muscular dystrophy (FSHD) is the epigenetic alteration of the D4Z4 sequence on chromosome 4, which leads to abnormal expression of the DUX4 gene. This paper focuses on the extremely low level of expression of DUX4 in skeletal muscles and its regulatory mechanism, and reviews how it causes muscle cell dysfunction.
Creative Biolabs: DUX4 Antibodies for Research
Creative Biolabs specializes in the production of high-quality DUX4 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom DUX4 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our DUX4 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Mocciaro, Emanuele, et al. "DUX4 role in normal physiology and in FSHD muscular dystrophy." Cells 10.12 (2021): 3322. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/cells10123322
Anti-DUX4 antibodies
Products List
 Loading...
Loading...
Hot products 
-
Mouse Anti-FN1 Monoclonal Antibody (D6) (CBMAB-1240CQ)

-
Mouse Anti-CD83 Recombinant Antibody (HB15) (CBMAB-C1765-CQ)

-
Mouse Anti-FTH1 Recombinant Antibody (CBXF-1896) (CBMAB-F3426-CQ)

-
Rat Anti-CD34 Recombinant Antibody (MEC 14.7) (CBMAB-C10196-LY)

-
Mouse Anti-FLI1 Recombinant Antibody (CBXF-0733) (CBMAB-F0435-CQ)

-
Mouse Anti-ARHGAP5 Recombinant Antibody (54/P190-B) (CBMAB-P0070-YC)

-
Mouse Anti-ACKR3 Recombinant Antibody (V2-261265) (CBMAB-C1023-LY)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-0790) (CBMAB-0793-YY)

-
Mouse Anti-CD59 Recombinant Antibody (CBXC-2097) (CBMAB-C4421-CQ)

-
Mouse Anti-2C TCR Recombinant Antibody (V2-1556) (CBMAB-0951-LY)

-
Mouse Anti-AMACR Recombinant Antibody (CB34A) (CBMAB-CA034LY)

-
Mouse Anti-BCL6 Recombinant Antibody (CBYY-0435) (CBMAB-0437-YY)

-
Mouse Anti-CAT Recombinant Antibody (724810) (CBMAB-C8431-LY)

-
Mouse Anti-ACTB Recombinant Antibody (V2-179553) (CBMAB-A0870-YC)

-
Rat Anti-ADGRE4 Recombinant Antibody (V2-160163) (CBMAB-F0011-CQ)

-
Mouse Anti-CD8 Recombinant Antibody (C1083) (CBMAB-C1083-LY)

-
Mouse Anti-CD2AP Recombinant Antibody (BR083) (CBMAB-BR083LY)

-
Mouse Anti-ACLY Recombinant Antibody (V2-179314) (CBMAB-A0610-YC)

-
Mouse Anti-CTCF Recombinant Antibody (CBFYC-2371) (CBMAB-C2443-FY)

-
Mouse Anti-ADIPOR1 Recombinant Antibody (V2-179982) (CBMAB-A1368-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






