PRCP Antibodies
Background
The prolyl carboxypeptidase encoded by the PRCP gene is a lysosomal serine protease mainly found in tissues such as endothelial cells, kidneys, and lungs. This enzyme specifically hydrolyzes the peptide bond at the C-terminal proline, participating in the regulation of the metabolism of active peptides such as angiotensin 1-7 and bradykinin, thereby influencing blood pressure regulation and inflammatory responses. Studies have found that abnormal function of PRCP is associated with various pathological conditions such as cardiovascular diseases, metabolic syndrome, and sleep apnea, making it an important target for drug development.
Structure of PRCP
The PRCP gene encodes a prolyl carboxypeptidase with a molecular weight of approximately 55 kDa, and there are slight differences among different species. This enzyme belongs to the S28 family of serine proteases, and its activity depends on the conformation of the catalytic triad Ser-Asp-His.
| Species | Human | Mouse | Rat | Monkey |
| Molecular Weight (kDa) | 55.8 | 54.2 | 54.1 | 55.6 |
| Primary Structural Differences | Contains signal peptide and proline-rich region | Highly conserved catalytic domain | 89% homology to humans | Complete sequence of active site is identical |
PRCP is composed of approximately 496 amino acids and includes an N-terminal signal peptide, a proline-rich region, and an α/β hydrolase catalytic domain. Its three-dimensional structure presents a typical α/β fold, with the catalytic center located in the crevice between the two domains. The enzyme activity is dependent on the specific recognition of the C-terminal proline of the substrate, and achieves peptide bond hydrolysis by stabilizing the transition state.
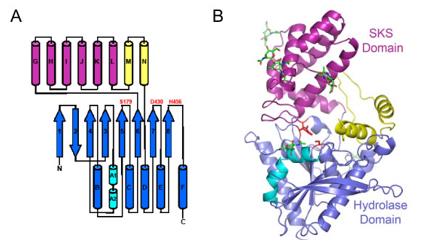 Fig. 1 The structure of PRCP.1
Fig. 1 The structure of PRCP.1
The core structural features of PRCP are as follows:
- Typical α/β hydrolase folding structure
- Catalytic triad composed of -Ser-Asp-His
- The C-terminal proline specifically recognizes the pocket (S1 site)
- Unique helical domains cover the active center and regulate the entry of substrates
- Disulfide bonds maintaining the stability of the tertiary structure
Functions of PRCP
The main function of PRCP is to regulate the metabolism of active peptides such as angiotensin 1-7 and bradykinin by hydrolyzing the C-terminal proline residue. This, in turn, participates in blood pressure regulation, inflammatory responses, and maintenance of vascular endothelial function.
| Function | Description |
| Blood pressure regulation | Converts angiotensin II into the vasodilator angiotensin 1-7, reducing blood pressure; simultaneously inactivates bradykinin and regulates vascular permeability. |
| Inflammation regulation | Regulates the expression of inflammatory factors and leukocyte adhesion by influencing the kininase-kinin system. |
| Metabolic involvement | Related to insulin sensitivity and fat metabolism, with abnormal expression in obesity and diabetes. |
| Cardiovascular protection | Regulates myocardial ischemia-reperfusion injury, exerting protective effects by influencing mitochondrial autophagy. |
| Endothelial function maintenance | Highly expressed in endothelial cells, maintaining vascular homeostasis and anticoagulant state. |
PRCP exhibits strict proline specificity in the hydrolysis of substrates, and this property enables it to play a unique bidirectional regulatory role in the renin-angiotensin system and the kinin system.
Applications of PRCP and PRCP Antibody in Literature
1. Soisson, Stephen M., et al. "Structural definition and substrate specificity of the S28 protease family: the crystal structure of human prolylcarboxypeptidase." BMC structural biology 10.1 (2010): 16. https://doi.org/10.1186/1472-6807-10-16
This article analyzes the crystal structure of human PRCP, revealing the structural basis for the differences in substrate specificity between the S28 protease family PRCP and DPP7, providing a basis for the design of selective PRCP modulators.
2. Ngo, My-Linh, et al. "Upregulation of prolylcarboxypeptidase (PRCP) in lipopolysaccharide (LPS) treated endothelium promotes inflammation." Journal of inflammation 6.1 (2009): 3. https://doi.org/10.1186/1476-9255-6-3
The study found that the upregulation of PRCP can promote the inflammatory response of endothelial cells induced by LPS. PRCP activates kininase, leading to continuous production of bradykinin, which may be a new mechanism for the persistence of inflammation.
3. Ye, Weilong, et al. "Decoding the hypoxia-exosome-immune triad in OSA: PRCP/UCHL1/BTG2-driven metabolic dysregulation revealed by interpretable machine learning." Frontiers in Immunology 16 (2025): 1587522. https://doi.org/10.3389/fimmu.2025.1587522
By integrating and analyzing the transcriptome data, the study identified PRCP, UCHL1 and BTG2 as key exosome-related biomarkers that regulate immune metabolic disorders in OSA, providing new targets for diagnosis and treatment.
4. Gittleman, Haley R., et al. "A cross-sectional study of KLKB1 and PRCP polymorphisms in patient samples with cardiovascular disease." Frontiers in medicine 3 (2016): 17. https://doi.org/10.3389/fmed.2016.00017
The study found that the polymorphism of the PRCP gene was slightly correlated with the history of coronary angioplasty; while the G allele of rs3733402 in the KLKB1 gene could significantly reduce the risk of coronary heart disease history, and this allele was associated with lower plasma kallikrein enzyme activity.
5. Hao, Panpan, et al. "Prolylcarboxypeptidase mitigates myocardial ischemia/reperfusion injury by stabilizing mitophagy." Frontiers in cell and developmental biology 8 (2020): 584933. https://doi.org/10.3389/fcell.2020.584933
Studies have shown that PRCP promotes mitochondrial autophagy during myocardial ischemia by generating Ang-(1-7) and BK-(1-9), and inhibits autophagy during reperfusion, thereby alleviating myocardial damage.
Creative Biolabs: PRCP Antibodies for Research
Creative Biolabs specializes in the production of high-quality PRCP antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom PRCP Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our PRCP antibodies, custom preparations, or technical support, contact us at email.
Reference
- Soisson, Stephen M., et al. "Structural definition and substrate specificity of the S28 protease family: the crystal structure of human prolylcarboxypeptidase." BMC structural biology 10.1 (2010): 16. Distributed under Open Access license CC BY 2.0, without modification. https://doi.org/10.1186/1472-6807-10-16
Anti-PRCP antibodies
 Loading...
Loading...
Hot products 
-
Rat Anti-EMCN Recombinant Antibody (28) (CBMAB-E0280-FY)

-
Mouse Anti-APCS Recombinant Antibody (CBYC-A663) (CBMAB-A3054-YC)

-
Mouse Anti-BLK Recombinant Antibody (CBYY-0618) (CBMAB-0621-YY)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503416) (CBMAB-V208-1402-FY)

-
Mouse Anti-ESR1 Recombinant Antibody (Y31) (CBMAB-1208-YC)

-
Rabbit Anti-ALK (Phosphorylated Y1278) Recombinant Antibody (D59G10) (PTM-CBMAB-0035YC)

-
Mouse Anti-HTLV-1 gp46 Recombinant Antibody (CBMW-H1006) (CBMAB-V208-1154-FY)

-
Mouse Anti-ARHGDIA Recombinant Antibody (CBCNA-009) (CBMAB-R0415-CN)

-
Mouse Anti-CD8 Recombinant Antibody (C1083) (CBMAB-C1083-LY)

-
Mouse Anti-ARIH1 Recombinant Antibody (C-7) (CBMAB-A3563-YC)

-
Mouse Anti-ALOX5 Recombinant Antibody (33) (CBMAB-1890CQ)

-
Mouse Anti-CARD11 Recombinant Antibody (CBFYC-0811) (CBMAB-C0866-FY)

-
Mouse Anti-CCN2 Recombinant Antibody (CBFYC-2383) (CBMAB-C2456-FY)

-
Mouse Anti-CD33 Recombinant Antibody (6C5/2) (CBMAB-C8126-LY)

-
Rabbit Anti-CBL Recombinant Antibody (D4E10) (CBMAB-CP0149-LY)

-
Mouse Anti-FOSB Recombinant Antibody (CBXF-3593) (CBMAB-F2522-CQ)

-
Mouse Anti-CD59 Recombinant Antibody (CBXC-2097) (CBMAB-C4421-CQ)

-
Mouse Anti-GLP1R Recombinant Antibody (4F3) (CBMAB-G0521-LY)

-
Rabbit Anti-ATF4 Recombinant Antibody (D4B8) (CBMAB-A3872-YC)

-
Mouse Anti-BACE1 Recombinant Antibody (CBLNB-121) (CBMAB-1180-CN)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






