TPM2 Antibodies
Background
The TPM2 gene encodes the protomyosin β chain and is mainly present in the skeletal muscle and myocardial cells of vertebrates. This gene product participates in the formation of the filamentous structure of muscle fibers and controls the calcium ion response during muscle contraction by regulating the conformational changes of the troponin complex. Patients with congenital myasthenia gravis often carry TPM2 gene mutations, which affect their function of maintaining normal muscle fiber assembly and stability. This gene was first located in the 1970s, and the three-dimensional structure of the protein it encodes was analyzed by crystallography in the 1990s, laying a crucial foundation for understanding the function of the myosin family. Research on the TPM2 gene and its protein products continuously deepens people's understanding of muscle development, contraction mechanisms, and the molecular basis of related genetic diseases.
Structure of TPM2
β-protomyosin encoded by the TPM2 gene is a protein with a molecular weight of approximately 36 kDa. Its molecular weight varies slightly among different species, mainly due to the conservation of amino acid sequences and specific variants.
| Species | Human | Mouse | Rat | Zebrafish |
| Molecular Weight (kDa) | 36.1 | 35.8 | 35.9 | 34.7 |
| Primary Structural Differences | Typical α -helical coiled structure | The core structure is highly conservative | High similarity to human sequence | High homology and conservative function |
The TPM2 protein is mainly composed of α -helices forming its core coiled-helix secondary structure. This structure forms a stable double-stranded superhelix through heptapeptide repeat sequences, which is the basis for its function. The specific amino acid sequences at its N-terminal and C-terminal determine its binding specificity to actin filaments, thereby precisely regulating the location of troponin complexes in striated and smooth muscles to respond to calcium ion signals and achieve on-off control of muscle contraction.
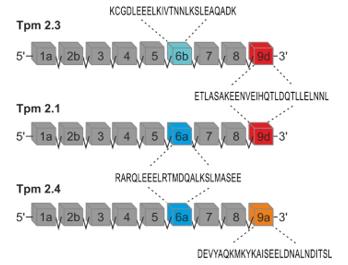 Fig. 1 A diagram illustrating the exon composition of the TPM2 gene.1
Fig. 1 A diagram illustrating the exon composition of the TPM2 gene.1
Key structural properties of TPM2:
- Stable α-helical coiled structure
- Continuous hydrophobic core
- Conservative repetitive binding sites
- Conformational regulatory region
Functions of TPM2
The β -protomyosin encoded by the TPM2 gene mainly functions to regulate the contraction of striated and smooth muscles. Meanwhile, it is also involved in a variety of cellular physiological processes, including cytoskeletal stability and signal transduction.
| Function | Description |
| Muscle contraction regulation | As a core component of the troponin-protomyoprotein complex, it slides on actin filaments and responds to calcium ion signals to control the contraction and relaxation of striated muscles. |
| Filament stability was maintained | By closely binding with actin filaments, it enhances the mechanical stability of the cytoskeleton and maintains the regular structure of the muscle segments. |
| Non-muscle cell functions | In non-muscular cells such as fibroblasts, it is involved in the dynamic regulation of the microfilament skeleton that participates in processes such as cell morphology maintenance, migration, and cytoplasmic division. |
| Association with genetic diseases | Loss of function or missense mutation is closely associated with congenital myopathy (e.g., rod-shaped myopathy) and skeletal muscle developmental disorders such as distal joint flexion. |
The synergistic effect with troponin enables TPM2 to respond rapidly to changes in intracellular calcium ion concentration. Its regulatory mode has the characteristic of an "allosteric switch", ensuring the accuracy and reliability of muscle contraction.
Applications of TPM2 and TPM2 Antibody in Literature
1. Wu, Zonglong, et al. "TPM2 attenuates progression of prostate cancer by blocking PDLIM7-mediated nuclear translocation of YAP1." Cell & Bioscience 13.1 (2023): 39. https://doi.org/10.1186/s13578-023-00993-w
This article found that TPM2 is lowly expressed in prostate cancer. It competitively binds to PDLIM7 through the PDZ motif, inhibiting the nuclear translocation and activity of YAP1, thereby delaying tumor progression and providing a new target for the treatment of castration-resistant prostate cancer.
2. Meng, Ling-Bing, et al. "TPM2 as a potential predictive biomarker for atherosclerosis." Aging (Albany NY) 11.17 (2019): 6960. https://doi.org/10.18632/aging.102231
This study, through bioinformatics analysis and animal model verification, found that TPM2 is lowly expressed in atherosclerosis, and its function is related to smooth muscle cells and the cytoskeleton, suggesting that TPM2 can serve as a potential diagnostic and therapeutic biomarker.
3. Siatkowska, Małgorzata, et al. "Tropomyosin isoforms encoded by TPM2 control the actin-bundling activity of fascin-1." Biological Research 58.1 (2025): 60. https://doi.org/10.1186/s40659-025-00640-3
The article indicates that the cytoplasmic isomers of TPM2 can directly bind to Fascin-1, inhibiting its binding and bundling ability with F-actin, thereby regulating the composition of actin bundles. This mechanism is helpful in suppressing the metastatic phenotype of cancer cells.
4. Meng, Ling-bing, et al. "A potential target for clinical atherosclerosis: a novel insight derived from TPM2." Aging and disease 13.2 (2022): 373. https://doi.org/10.14336/AD.2021.0926
The article indicates that clinical and bioinformatics analyses show that the expression of TPM2 is decreased in atherosclerotic tissues and is negatively correlated with the thickness of the vascular wall, suggesting that it can serve as an independent protective factor and potential diagnostic marker for this disease.
5. Śliwinska, Małgorzata, et al. "Mutations Q93H and E97K in TPM2 disrupt Ca-dependent regulation of actin filaments." International Journal of Molecular Sciences 22.8 (2021): 4036. https://doi.org/10.3390/ijms22084036
The article indicates that the Q93H and E97K mutations of the TPM2 gene respectively reduce and enhance the affinity of their encoded proteins for actin and ATPase activity, which leads to low/high contraction manifestations of muscle fibers and thereby explains the molecular mechanism of related congenital myopathy.
Creative Biolabs: TPM2 Antibodies for Research
Creative Biolabs specializes in the production of high-quality TPM2 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom TPM2 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our TPM2 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Siatkowska, Małgorzata, et al. "Tropomyosin isoforms encoded by TPM2 control the actin-bundling activity of fascin-1." Biological Research 58.1 (2025): 60. https://doi.org/10.1186/s40659-025-00640-3
Anti-TPM2 antibodies
Products List
 Loading...
Loading...
Hot products 
-
Mouse Anti-BCL2L1 Recombinant Antibody (H5) (CBMAB-1025CQ)

-
Rabbit Anti-B2M Recombinant Antibody (CBYY-0059) (CBMAB-0059-YY)

-
Mouse Anti-CORO1A Recombinant Antibody (4G10) (V2LY-1206-LY806)

-
Rat Anti-ADGRE4 Recombinant Antibody (V2-160163) (CBMAB-F0011-CQ)

-
Mouse Anti-ATG5 Recombinant Antibody (9H197) (CBMAB-A3945-YC)

-
Mouse Anti-ALB Recombinant Antibody (V2-363290) (CBMAB-S0173-CQ)

-
Mouse Anti-AQP2 Recombinant Antibody (E-2) (CBMAB-A3358-YC)

-
Mouse Anti-BACE1 Recombinant Antibody (CBLNB-121) (CBMAB-1180-CN)

-
Rat Anti-ADAM10 Recombinant Antibody (V2-179741) (CBMAB-A1103-YC)

-
Mouse Anti-AKT1 Recombinant Antibody (V2-180546) (CBMAB-A2070-YC)

-
Mouse Anti-CD24 Recombinant Antibody (HIS50) (CBMAB-C10123-LY)

-
Mouse Anti-CAT Recombinant Antibody (724810) (CBMAB-C8431-LY)

-
Mouse Anti-CCT6A/B Recombinant Antibody (CBXC-0168) (CBMAB-C5570-CQ)

-
Mouse Anti-ACTN4 Recombinant Antibody (V2-6075) (CBMAB-0020CQ)

-
Mouse Anti-ALDOA Recombinant Antibody (A2) (CBMAB-A2316-YC)

-
Mouse Anti-CDKL5 Recombinant Antibody (CBFYC-1629) (CBMAB-C1689-FY)

-
Mouse Anti-CCN2 Recombinant Antibody (CBFYC-2383) (CBMAB-C2456-FY)

-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Mouse Anti-ADGRE5 Recombinant Antibody (V2-360335) (CBMAB-C2088-CQ)

-
Rabbit Anti-BAD (Phospho-Ser136) Recombinant Antibody (CAP219) (CBMAB-AP536LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






