GATM Antibodies
Background
The glycine amidinotransferase encoded by the GATM gene is a mitochondrial matrix enzyme, mainly distributed in kidney and liver tissues. This enzyme catalyzes the key rate-limiting step in creatine biosynthesis by transferring the amidine group of arginine to glycine to form ornithine and creatine precursor guanidinoacetic acid, thereby maintaining cellular energy metabolic homeostasis. The regulation of creatine levels in mammals is highly dependent on the activity of GATM, and its dysfunction is closely related to creatine deficiency syndrome and various metabolic diseases. This gene was first located and cloned in 1990. The tertiary structure of the protein it encodes was analyzed by X-ray crystallography in the early 21st century, providing a classic model for understanding the catalytic mechanism of metabolic enzymes. The highly conserved catalytic domain of GATM and its allosteric regulation mechanism continue to provide an important theoretical basis for the research of metabolic disease targets.
Structure of GATM
The glycine amidinotransferase encoded by the GATM gene is a mitochondrial matrix enzyme with a molecular weight of approximately 48 kDa. There are slight differences in its molecular weight among different species, mainly due to the length changes of the N-terminal mitochondrial targeting sequence.
| Species | Human | Mouse | Rat | Bovine |
| Molecular Weight (kDa) | 48 | 47.5 | 47.8 | 48.2 |
| Primary Structural Differences | Containing about 424 amino acids, n-terminal mitochondrial signal peptide has 39 amino acids | Signal peptide lengths are slightly different and the catalytic core is highly conserved | High homology with human sequence | Sequence is highly conserved structure of catalytic domain |
This enzyme functions in the form of a homodimer, with each monomer containing a mature peptide of approximately 385 amino acids. Its tertiary structure exhibits a typical double-domain folding: a large N-terminal domain is responsible for substrate binding and catalysis, containing highly conserved active centers; A small C-end domain participates in structural stability. The key arginine residues in the active center are responsible for recognizing and binding to the substrate glycine, while the glutamic acid residues play a crucial role in catalysis, jointly completing the amidine transfer reaction. This structure ensures its rate-limiting enzyme function in the creatine synthesis pathway.
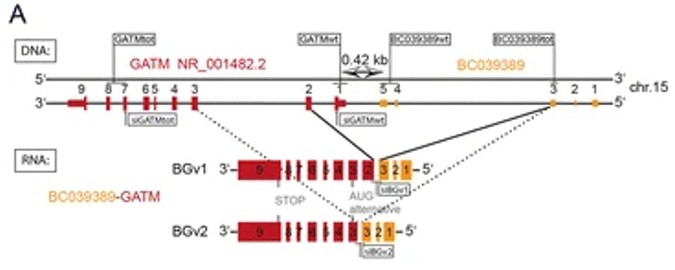 Fig. 1 DNA: Genomic structure of BC039389 and GATM on the DNA (−) strand.1
Fig. 1 DNA: Genomic structure of BC039389 and GATM on the DNA (−) strand.1
Key structural properties of GATM:
- Typical double-domain folding
- Highly conserved active centers
- Key catalytic residues
- Homomorphic dimer interface
Functions of GATM
The core function of the glycine amidinotransferase encoded by the GATM gene is to catalyze the rate-limiting step in creatine biosynthesis. Meanwhile, it is also involved in a variety of related physiological and pathological processes.
| Function | Description |
| Creatine synthesis | The transfer of the amidine group of arginine to glycine in the mitochondria to generate ornithine and guanidinoacetic acid (the direct precursor of creatine) is the key and sole step in the de novo synthesis of creatine in the body. |
| Regulation of energy metabolism | By controlling the synthesis of creatine and its phosphorylated form (phosphocreatine), it maintains the energy buffering and rapid ATP regeneration system of cells, especially high-energy-consuming muscles and nerve tissues. |
| One-carbon metabolism association | This reaction consumes arginine and produces ornithine, indirectly linking the urea cycle and the one-carbon metabolism pathway, and affecting the overall nitrogen metabolism balance. |
| Disease association | GATM gene mutations are the main cause of primary creatine deficiency syndrome, leading to delayed neurodevelopment and muscle weakness. Its abnormal expression is also associated with chronic kidney disease and cardiovascular diseases. |
| Drug development targets | Due to its rate-limiting enzyme status, GATM is a potential drug target for regulating creatine levels in the body and treating related metabolic diseases. |
Unlike the co-regulation of hemoglobin, the activity of GATM is mainly influenced by the availability of substrates (glycine and arginine), feedback inhibition of end products, and hormonal regulation at the transcriptional level, demonstrating its characteristic as a key control point in metabolic pathways.
Applications of GATM and GATM Antibody in Literature
1. Liu, Mengyuan, et al. "The association of GATM polymorphism with statin-induced myopathy: a systematic review and meta-analysis." European Journal of Clinical Pharmacology 77.3 (2021): 349-357. https://doi.org/10.1007/s00228-020-03019-3
The article indicates that loci such as rs9806699 of the GATM gene can reduce the risk of statin-related myopathy, especially for patients with mild symptoms. This gene polymorphism may serve as a biomarker for predicting the risk of myopathy, which is helpful for the safety assessment of clinical medication.
2. Forst, Anna-Lena, et al. "Distinct mitochondrial pathologies caused by mutations of the proximal tubular enzymes EHHADH and GATM." Frontiers in physiology 12 (2021): 715485. https://doi.org/10.3389/fphys.2021.715485
The article indicates that the mechanisms of mitochondrial dysfunction in the proximal renal tubules are diverse. Mutations in the GATM gene do not affect ATP synthesis, but they lead to mitochondrial protein aggregation and inflammatory activation, causing renal fibrosis and functional failure. It is a unique type of mitochondrial nephropathy.
3. Portales‐Castillo, Ignacio, et al. "Reduced guanidinoacetate in plasma of patients with autosomal dominant Fanconi syndrome due to heterozygous P341L GATM variant and study of organoids towards treatment." JIMD reports 65.5 (2024): 341-353. https://doi.org/10.1002/jmd2.12442
The article indicates that the GATM P341L gene mutation leads to the decrease of GAA levels in the plasma and urine of patients with GATM-FS. Creatine supplementation can significantly increase the plasma creatine concentration and down-regulate the expression of AGAT in the kidneys.
4. Yu, Li, et al. "Reprogramming alternative macrophage polarization by GATM-mediated endogenous creatine synthesis: A potential target for HDM-induced asthma treatment." Frontiers in Immunology 13 (2022): 937331. https://doi.org/10.3389/fimmu.2022.937331
The article indicates that GATM is a key factor for the polarization of macrophages to M2 type (substitution activation), and its expression is regulated by STAT6. The newly created synthetic pathway involved by this enzyme has a potential role in the occurrence of Th2-related inflammatory diseases such as asthma.
5. Pflueger, Dorothee, et al. "Functional characterization of BC039389-GATM and KLK4-KRSP1 chimeric read-through transcripts which are up-regulated in renal cell cancer." BMC genomics 16.1 (2015): 247. https://doi.org/10.1186/s12864-015-1446-z
In this study, multiple transmission-reading RNA chimeras were discovered in renal cell carcinoma through RNA-SEq. Among them, the expressions of BC039389-GATM and KLK4-KRSP1 were upregulated, and the latter was associated with poor prognosis of patients. Experiments show that these chimeras can antagonize the role of parental genes in cancer mutations, suggesting that they have regulatory functions.
Creative Biolabs: GATM Antibodies for Research
Creative Biolabs specializes in the production of high-quality GATM antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom GATM Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our GATM antibodies, custom preparations, or technical support, contact us at email.
Reference
- Yang, Jinshou, et al. "Epigenetic reprogramming-induced guanidinoacetic acid synthesis promotes pancreatic cancer metastasis and transcription-activating histone modifications." Journal of Experimental & Clinical Cancer Research 42.1 (2023): 155. https://doi.org/10.1186/s13046-023-02698-x
Anti-GATM antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-CFL1 (Phospho-Ser3) Recombinant Antibody (CBFYC-1770) (CBMAB-C1832-FY)

-
Rabbit Anti-CCL5 Recombinant Antibody (R0437) (CBMAB-R0437-CN)

-
Mouse Anti-ALPL Antibody (B4-78) (CBMAB-1009CQ)

-
Rat Anti-CCR2 Recombinant Antibody (475301) (CBMAB-C1338-LY)

-
Mouse Anti-FeLV g27 Recombinant Antibody (1) (CBMAB-V208-1714-FY)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Mouse Anti-ALB Recombinant Antibody (V2-180650) (CBMAB-A2186-YC)

-
Rabbit Anti-AKT2 (Phosphorylated S474) Recombinant Antibody (V2-556130) (PTM-CBMAB-0605LY)

-
Mouse Anti-CCNH Recombinant Antibody (CBFYC-1054) (CBMAB-C1111-FY)

-
Mouse Anti-DMD Recombinant Antibody (D1190) (CBMAB-D1190-YC)

-
Mouse Anti-ENO2 Recombinant Antibody (85F11) (CBMAB-0276CQ)

-
Mouse Anti-AHCYL1 Recombinant Antibody (V2-180270) (CBMAB-A1703-YC)

-
Mouse Anti-ATP1B1 Recombinant Antibody (E4) (CBMAB-0463-LY)

-
Mouse Anti-GIPC2 Recombinant Antibody (10) (CBMAB-G0476-LY)

-
Mouse Anti-APOH Recombinant Antibody (4D9A4) (CBMAB-A3249-YC)

-
Mouse Anti-BANF1 Recombinant Antibody (3F10-4G12) (CBMAB-A0707-LY)

-
Mouse Anti-FYN Recombinant Antibody (10) (CBMAB-S6332-CQ)

-
Mouse Anti-AKT1 Recombinant Antibody (V2-180546) (CBMAB-A2070-YC)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503416) (CBMAB-V208-1402-FY)

-
Mouse Anti-DDC Recombinant Antibody (8E8) (CBMAB-0992-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






