Vimentin Antibodies
Background
Vimentin is a medium fibrin mainly present in mesenchymal cells of vertebrates. As a core component of the cytoskeleton, it maintains cell morphology, enhances mechanical stability, and participates in key processes such as intracellular material transport and signal transduction by forming a dynamic fiber network. This protein promotes cell migration through structural reorganization in physiological activities such as wound healing and embryonic development, while its abnormal expression is closely related to pathological processes such as cancer metastasis. In 1979, vimentin was first identified by the Elias Lazarides team. As the first medium fibrin to be resolved, the study of its α -helical rod-shaped domain and variable terminal domain provided a model for revealing the mechanism of cytoskeleton assembly, not only promoting the development of cell mechanics and cancer biology. It has become a classic model for the study of protein self-assembly and kinetics.
Structure of Vimentin
Vimentin is a medium fibrin with a molecular weight of approximately 57 kDa, and its precise molecular weight varies slightly among different species. As a major member of type III intermediate fibers, its molecular structure exhibits typical rod-shaped dimer characteristics.
| Species | Human | Mice | Rats | Bovine | Chicken |
| Molecular Weight (kDa) | 56.5 | 57.2 | 56.8 | 57.1 | 56.3 |
| Primary Structural Differences | Conserved central α-helical rod domain | C-terminal tail sequence variation | Differences in N-terminal header modifications | Different phosphorylation sites | To model a special expressed in embryonic development |
Under an electron microscope, vimentin presents a typical reticular fiber morphology. Its N-terminal head domain contains multiple serine phosphorylation sites, and these modification sites affect the fiber assembly state by altering the surface charge of the protein. The stability of the central rod-shaped domain stems from hydrophobic interactions and salt bridge networks, while the C-terminal tail domain prevents excessive fiber cross-linking through steric hindrance effects. This unique "rod-connection-rod" structural model endows the vimentin fiber network with both mechanical strength and dynamic plasticity.
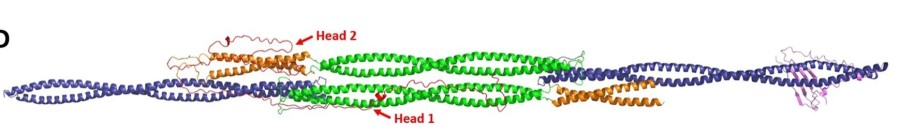 Fig. 1 Vimentin tetramer models. 1
Fig. 1 Vimentin tetramer models. 1
Key structural properties of vimentin:
- Highly conservative α -helical rod-shaped domain
- Dynamic end domain
- Four-level structure assembly mechanism
- Phosphorylation regulatory site
Functions of Vimentin
Vimentin, as a key component of the cytoskeleton, not only maintains the structural integrity of cells but also participates in a variety of important physiological and pathological processes.
| Function | Description |
| Mechanical support | Form a three-dimensional reticular structure to maintain cell morphology and enhance tissue tensile resistance. |
| Regulation of cell migration | Promote the movement of mesenchymal cells through dynamic recombination, and participate in wound healing and embryonic development. |
| Organelle localization | Anchor important organelles such as the nucleus and mitochondria to maintain their spatial distribution. |
| Signal transduction | As a scaffold protein, it integrates multiple signaling pathways (such as TGF-β and Wnt pathways). |
| Stress protection | Stabilize cell structure under conditions such as oxidative stress and prevent mechanical damage. |
| Promotion of tumor metastasis | Overexpression during epithelial-mesenchymal transition (EMT) enhances the invasiveness of cancer cells. |
| Regulation of inflammatory response | Regulate the secretion of inflammatory factors and the migration of immune cells. |
The functional performance of vimentin depends on its unique dynamic assembly characteristics: it forms a stable network in the resting state and rapidly depolymerizes and recombines during cell migration or stress. This "solid-liquid" phase transition characteristic enables it to respond rapidly to changes in the micro-environment. When acting in synergy with actin microtubules, vimentin fibers exhibit distinct strain strengthening properties, with their elastic modulus increasing tenfold within the range of 10-100% deformation. This characteristic is particularly important for tissues subjected to mechanical forces, such as vascular endothelium and muscle.
Applications of Vimentin and Vimentin Antibody in Literature
1. Yu, Yvonne Ting-Chun, et al. "Surface vimentin is critical for the cell entry of SARS-CoV." Journal of biomedical science 23 (2016): 1-10. https://doi.org/10.1186/s12929-016-0234-7
Research has found that vimentin interacts directly with the spike protein of SARS-CoV and participates in viral invasion. Antibody neutralization and gene knockout experiments confirmed that targeting vimentin can effectively inhibit viral binding and endocytosis, suggesting its potential as a drug target against SARS-CoV.
2. Dejaco, Christian, et al. "Diagnostic value of antibodies against a modified citrullinated vimentin in rheumatoid arthritis." Arthritis research & therapy 8 (2006): 1-6. https://doi.org/10.1186/ar2008
Anti-citrullinated vimentin antibody (anti-MCV) is a specific marker for rheumatoid arthritis (RA). Compared with anti-CCP2, the sensitivity of anti-MCV detection was similar (69.5% vs 70.1%), but anti-CCP2 was superior in the high specificity range (98.7% vs 90.8%), and both were applicable to the diagnosis of RA.
3. Jørgensen, Mathias Lindh, et al. "An anti vimentin antibody promotes tube formation." Scientific Reports 7.1 (2017): 3576. https://doi.org/10.1038/s41598-017-03799-2
Research has found that anti-vimentin antibodies can significantly promote endothelial cell tube formation in vitro (increasing by 21% within 5 hours) by targeting the coil 2 domain of vimentin, without affecting cell migration or proliferation. This is the first time that the regulatory role of vimentin in angiogenesis under normoxic conditions has been confirmed.
4. Mak, Tim N., and Holger Brüggemann. "Vimentin in bacterial infections." Cells 5.2 (2016): 18.https://doi.org/10.3390/cells5020018
Research has found that vimentin, as a type III intermediate fibrin, plays a dual role in bacterial infections: it not only serves as a surface-binding receptor for pathogens to promote invasion but also participates in intracellular innate immune signal transduction. Its function can be targeted and regulated by bacterial virulence factors.
5. Lahat, Guy, et al. "Vimentin is a novel anti-cancer therapeutic target; insights from in vitro and in vivo mice xenograft studies." PloS one 5.4 (2010): e10105. https://doi.org/10.1371/journal.pone.0010105
Research has found that the natural compound Withaferin-A significantly inhibits the growth and metastasis of soft tissue sarcomas by inducing vimentin degradation and caspase-dependent apoptosis, suggesting that targeting vimentin is a new strategy for the treatment of mesenchymal tumors.
Creative Biolabs: Vimentin Antibodies for Research
Creative Biolabs specializes in the production of high-quality vimentin antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom Vimentin Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our vimentin antibodies, custom preparations, or technical support, please contact us.
Reference
- Vermeire, Pieter-Jan, et al. "Molecular structure of soluble vimentin tetramers." Scientific Reports 13.1 (2023): 8841. https://doi.org/10.1038/s41598-023-34814-4
Anti-Vimentin antibodies
 Loading...
Loading...
Hot products 
-
Rabbit Anti-ALK (Phosphorylated Y1278) Recombinant Antibody (D59G10) (PTM-CBMAB-0035YC)

-
Rabbit Anti-AKT2 (Phosphorylated S474) Recombinant Antibody (V2-556130) (PTM-CBMAB-0605LY)

-
Mouse Anti-CCNH Recombinant Antibody (CBFYC-1054) (CBMAB-C1111-FY)

-
Mouse Anti-GLP1R Recombinant Antibody (4F3) (CBMAB-G0521-LY)

-
Mouse Anti-AKR1B1 Antibody (V2-2449) (CBMAB-1001CQ)

-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Mouse Anti-FOXL1 Recombinant Antibody (CBXF-0845) (CBMAB-F0462-CQ)

-
Mouse Anti-FYN Recombinant Antibody (10) (CBMAB-S6332-CQ)

-
Mouse Anti-ELAVL4 Recombinant Antibody (6B9) (CBMAB-1132-YC)

-
Mouse Anti-CD59 Recombinant Antibody (CBXC-2097) (CBMAB-C4421-CQ)

-
Mouse Anti-ARG1 Recombinant Antibody (CBYCL-103) (CBMAB-L0004-YC)

-
Mouse Anti-Acetyl-α-Tubulin (Lys40) Recombinant Antibody (V2-623485) (CBMAB-CP2897-LY)

-
Mouse Anti-AMOT Recombinant Antibody (CBYC-A564) (CBMAB-A2552-YC)

-
Mouse Anti-ATM Recombinant Antibody (2C1) (CBMAB-A3970-YC)

-
Mouse Anti-FN1 Monoclonal Antibody (D6) (CBMAB-1240CQ)

-
Mouse Anti-GIPC2 Recombinant Antibody (10) (CBMAB-G0476-LY)

-
Mouse Anti-FLT1 Recombinant Antibody (11) (CBMAB-V0154-LY)

-
Mouse Anti-BIRC7 Recombinant Antibody (88C570) (CBMAB-L0261-YJ)

-
Mouse Anti-B2M Recombinant Antibody (CBYY-0050) (CBMAB-0050-YY)

-
Mouse Anti-CCT6A/B Recombinant Antibody (CBXC-0168) (CBMAB-C5570-CQ)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






