HELZ Antibodies
Background
The HELZ gene encodes a RNA helicase protein, which is widely present in various human tissue cells. The product of this gene participates in regulating post-transcriptional processing of genes and RNA metabolism by binding and unwinding double-stranded RNA, playing an important role in maintaining the normal RNA homeostasis of cells. Studies have shown that abnormal function of the HELZ gene is associated with thyroid hormone resistance syndrome, developmental disorders, and the occurrence of some tumors. Its protein structure contains characteristic helicase core domain and zinc finger domain, which work together to achieve the recognition and unwinding function of specific RNA substrates. The continuous research on this gene has deepened our understanding of the regulatory mechanism of RNA metabolism and provided potential targets for molecular diagnosis of related diseases.
Structure of HELZ
HELZ2 (also known as HELZ) is a large-molecular-weight multifunctional protein with a molecular weight of approximately 330 kDa. The human HELZ2 protein is composed of 2875 amino acids, and its primary structure contains multiple characteristic functional domains. These structural differences collectively determine its complex functions.
| Species | Human | Mouse | Zebrafish |
| Molecular Weight (kDa) | ~330 | ~328 | ~320 |
| Primary Structural Differences | Including the core domain of helicase, the CCCH-type zinc finger array, and the RING domain | Highly conserved structure, functional domains of similar to humans | With core helicase domain, but some auxiliary structure differences between domains |
The secondary structure of this protein is composed of multiple α helices and β sheets, forming a complex spatial configuration. The core region contains a typical DExH-box RNA helicase domain, which can hydrolyze ATP to unwind the RNA double strand; meanwhile, the densely arranged CCCH-type zinc finger domains at the N-terminus are responsible for specifically recognizing and binding to the target RNA molecules. The coordinated action of these two key domains in space enables it to precisely regulate the metabolism and processing of RNA.
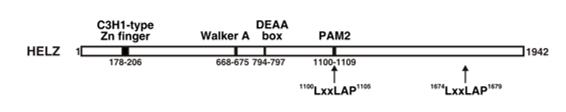 Fig. 1 Schematic representation of the predicted human HELZ protein architecture.1
Fig. 1 Schematic representation of the predicted human HELZ protein architecture.1
Key structural properties of HELZ:
- Containing the conservative DExH-box RNA helicase core domain
- Contains a tandemly repeated CCCH-type zinc finger motif for RNA binding
- Having the RING finger domain, it may be involved in protein interactions
- The overall structure of multiple domain of complex spatial configuration, control RNA metabolism and gene expression
Functions of HELZ
The protein encoded by the HELZ gene (HELZ2) is a multifunctional RNA helicase. Its core function is to participate in RNA metabolism and transcriptional regulation of gene expression, playing a crucial role especially in embryonic development and cell differentiation.
| Function | Description |
| RNA Unwinding and Metabolism | By utilizing its DExH-box helicase domain to hydrolyze ATP, it unwinds double-stranded RNA or RNA-DNA hybrids, and participates in the splicing, processing, and degradation of mRNA precursors. |
| Transcriptional Co-regulation | By interacting with various transcription factors such as nuclear receptors (such as thyroid hormone receptor, retinoic acid receptor), it regulates the transcriptional activity of downstream target genes. |
| Cell Differentiation Regulation | During the differentiation processes of various cell lineages such as adipocytes and neurons, the RNA stability of specific gene clusters is regulated to guide the determination of cell fate. |
| Embryo Development Support | It is crucial for the early embryonic development. Its absence would lead to severe developmental defects, highlighting its role in regulating the developmental-related gene network. |
| Tumor-related functions | In some cancers, they are expressed abnormally, possibly by altering the RNA metabolism of oncogenes or tumor suppressor genes, thereby affecting the proliferation and survival of tumor cells. |
Unlike the simple myoglobin, the function of the HELZ protein is highly dependent on its complex multi-domain structure, which enables it to integrate instructions from multiple signaling pathways, functioning like a "molecular command center" to precisely coordinate the complex biological processes at the RNA level.
Applications of HELZ and HELZ Antibody in Literature
1. Hanet, Aoife, et al. "HELZ directly interacts with CCR4–NOT and causes decay of bound mRNAs." Life science alliance 2.5 (2019). https://doi.org/10.26508/lsa.201900405
The article indicates that the eukaryotic SF1 helicase HELZ has been discovered to directly bind to the C-terminal domain of the CCR4-NOT deadenylation complex. It relies on this complex to mediate translation inhibition and mRNA degradation, and plays a role in the regulation of gene expression related to nervous system development.
2. Hasgall, Philippe A., et al. "The putative RNA helicase HELZ promotes cell proliferation, translation initiation and ribosomal protein S6 phosphorylation." PloS one 6.7 (2011): e22107. https://doi.org/10.1371/journal.pone.0022107
The research has found that although the HELZ protein contains degradation motifs similar to HIFα, its degradation is not regulated by oxygen. Overexpression of HELZ can activate overall protein translation, while downregulation of HELZ will inhibit translation initiation, lead to the disassembly of polyribosomes, and slow down cell proliferation.
3. Lu, Wei, et al. "FOLFOX treatment response prediction in metastatic or recurrent colorectal cancer patients via machine learning algorithms." Cancer Medicine 9.4 (2020): 1419-1429. https://doi.org/10.1002/cam4.2786
The study found that the HELZ gene was expressed at a lower level in colorectal cancer patients who were sensitive to FOLFOX chemotherapy. The machine learning model constructed based on differentially expressed genes can effectively predict chemotherapy sensitivity, and among them, the MLKL and CCDC124 genes are independent prognostic factors.
4. Falcone, Noemi, et al. "Identification of a De novo deletion by using A-CGH involving PLNAX2: An interesting candidate gene in psychomotor developmental delay." Medicina 58.4 (2022): 524. https://doi.org/10.3390/medicina58040524
The research found that by analyzing the genome of a child with delayed motor development, it was discovered that the child carried copy number variations involving multiple genes such as PLXNA2 and HELZ. These gene variations jointly affected the development of the nervous system and might be related to the patient's clinical manifestations.
5. Chen, Chi-Hua, et al. "Leveraging genome characteristics to improve gene discovery for putamen subcortical brain structure." Scientific reports 7.1 (2017): 15736. https://doi.org/10.1038/s41598-017-15705-x
The research found that by using the newly enhanced functional annotation analysis method, the researchers discovered 11 genetic loci in the genome-wide association study of the volume of the brain nucleus, including HELZ. This has enhanced the effectiveness in identifying genetic variations related to brain structure.
Creative Biolabs: HELZ Antibodies for Research
Creative Biolabs specializes in the production of high-quality HELZ antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom HELZ Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our HELZ antibodies, custom preparations, or technical support, contact us at email.
Reference
- Hasgall, Philippe A., et al. "The putative RNA helicase HELZ promotes cell proliferation, translation initiation and ribosomal protein S6 phosphorylation." PloS one 6.7 (2011): e22107. https://doi.org/10.1371/journal.pone.0022107
Anti-HELZ antibodies
 Loading...
Loading...
Hot products 
-
Rat Anti-C5AR1 Recombinant Antibody (8D6) (CBMAB-C9139-LY)

-
Mouse Anti-GFAP Recombinant Antibody (24) (CBMAB-G2927-LY)

-
Mouse Anti-EIF4G1 Recombinant Antibody (2A9) (CBMAB-A2544-LY)

-
Mouse Anti-APP Recombinant Antibody (5C2A1) (CBMAB-A3314-YC)

-
Mouse Anti-ENO2 Recombinant Antibody (H14) (CBMAB-E1341-FY)

-
Mouse Anti-F11R Recombinant Antibody (402) (CBMAB-0026-WJ)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503417) (CBMAB-V208-1369-FY)

-
Mouse Anti-GFAP Recombinant Antibody (5) (CBMAB-G0346-LY)

-
Rabbit Anti-CCL5 Recombinant Antibody (R0437) (CBMAB-R0437-CN)

-
Rat Anti-CD34 Recombinant Antibody (MEC 14.7) (CBMAB-C10196-LY)

-
Mouse Anti-CARTPT Recombinant Antibody (113612) (CBMAB-C2450-LY)

-
Mouse Anti-FAS2 Monoclonal Antibody (1D4) (CBMAB-0071-CN)

-
Mouse Anti-DISP2 Monoclonal Antibody (F66A4B1) (CBMAB-1112CQ)

-
Mouse Anti-CCND2 Recombinant Antibody (DCS-3) (CBMAB-G1318-LY)

-
Mouse Anti-FN1 Monoclonal Antibody (D6) (CBMAB-1240CQ)

-
Mouse Anti-ARG1 Recombinant Antibody (CBYCL-103) (CBMAB-L0004-YC)

-
Mouse Anti-CD2AP Recombinant Antibody (BR083) (CBMAB-BR083LY)

-
Mouse Anti-AKT1 (Phosphorylated S473) Recombinant Antibody (V2-505430) (PTM-CBMAB-0067LY)

-
Mouse Anti-C5b-9 Recombinant Antibody (aE11) (CBMAB-AO138LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






