IgD Antibodies
Background
IgD is a membrane-bound antibody mainly distributed on the surface of B lymphocytes, composed of two heavy chains (δ chains) and two light chains. Its unique hinge region structure is easily hydrolyzed by proteases, resulting in a relatively short half-life of free IgD. As a key component of the B-cell receptor (BCR), IgD participates in the initiation of the immune response by recognizing specific antigens and plays an important role in activating primary B cells and regulating immune tolerance. This protein was first isolated and identified in human myeloma proteins in 1975. However, due to its extremely low content in serum (less than 1% of total immunoglobulins), the research progress has been relatively slow. Recent studies have found that IgD can not only participate in mucosal immunity and inflammatory regulation through interactions with basophils, mast cells, etc., but also its abnormal expression is closely related to autoimmune diseases such as IGD-type multiple myeloma and chronic infections, providing a new research direction for a deeper understanding of its biological functions.
Structure of IgD
IgD is an immunoglobulin with a molecular weight of approximately 185 kDa, and its molecular weight varies to some extent among different species. This protein is composed of two heavy chains (δ chains, approximately 55 kDa) and two light chains (κ or λ chains, approximately 25 kDa) linked by disulfide bonds.
| Species | Human | Bovine | Mouse |
| Molecular Weight (kDa) | 185 | 180-190 | 200-210 |
| Primary Structural Differences | Easy to hydrolysis long hinge area | Hinge region is shorter | More stable structure |
IgD has a unique hinge region structure, which is rich in O-glycosylation sites but also makes it prone to degradation by proteases at the same time. Its heavy chain contains three constant regions (Cδ1-Cδ3) and one variable region, and the antigen recognition function is achieved through the Fab segment. It is worth noting that human IgD is often co-expressed with IgM on the surface of B cells, forming a double BCR complex. The three-dimensional structure of this protein presents a typical "Y" type antibody conformation, but its longer hinge region endows the structure with higher flexibility. During the activation process of B cells, IgD binds to specific receptors through its Fc segment and participates in signal transduction. Recent studies have found that certain pathogens (such as Streptococcus pneumoniae) can produce specific IGD-binding proteins, suggesting their special role in infection immunity.
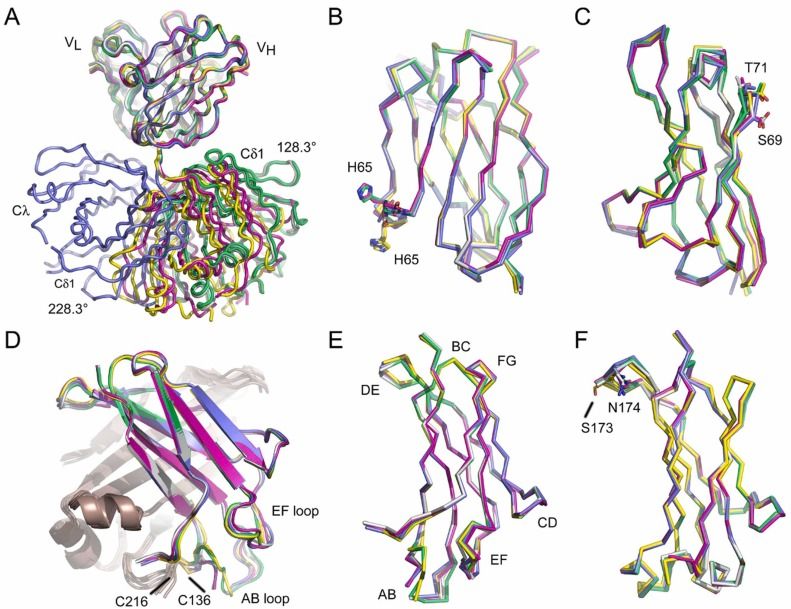 Fig. 1 Overall structure of the IgD Fab.1
Fig. 1 Overall structure of the IgD Fab.1
Key structural properties of IgD:
- Typical "Y" type antibody structure
- Unique super-long hinge area
- Rich in O-glycosylation modification sites
- Membrane-bound and secretory forms exist
Functions of IgD
The main function of IgD is related to immune regulation, and its special functions are mainly reflected in B cell activation and mucosal immunity:
| Function | Description |
| Activation of B cells | As a core component of the B-cell receptor (BCR), it participates in the activation and signal transduction of initial B cells. |
| Regulation of immune tolerance | Prevent the occurrence of autoimmune reactions by regulating the B-cell response threshold. |
| Mucosal immune defense | It is highly expressed in the mucosal tissues of the respiratory tract and digestive tract and participates in the local immune response. |
| Inflammation regulation | By binding to the basophilic CD300a receptor, it regulates the release of inflammatory factors. |
| Immune memory formation | Affect the differentiation process of B cells into memory B cells. |
IgD has the characteristics of co-expression with IgM to form a dual antigen recognition system, temperature sensitivity that is prone to detach from the surface of B cells at 37 °C, evolutionary features conserved in all mammals, and clinical features that abnormal levels are associated with autoimmune diseases and lymphoma. The latest research also found that secretory IgD can participate in mucosal immune surveillance by binding to immune cells such as mast cells.
Applications of IgD and IgD Antibody in Literature
1. Davies, Anna M., et al. "The Crystal Structure of Human IgD-Fc Reveals Unexpected Differences With Other Antibody Isotypes." Proteins: Structure, Function, and Bioinformatics 93.4 (2025): 786-800. https://doi.org/10.1002/prot.26771
This article indicates that the latest research findings show that IGD-FC has a unique structure, and its orientation of the Cδ3 domain and interface characteristics are significantly different from those of other antibody subtypes. There are special binding pockets at the Cδ2/Cδ3 interface, which may play a key role in maintaining the stability of IgD dimers.
2. Dirks, Johannes, et al. "IgD shapes the pre-immune naïve B cell compartment in humans." Frontiers in Immunology 14 (2023): 1096019. https://doi.org/10.3389/fimmu.2023.1096019
This article indicates that although IgD deletion does not affect the generation of initial B cells, it can change the expression of surface markers such as CD19/CD21, resulting in a weakened response of B cells to BCR/TLR signals and affecting the selection of specific VH regions, suggesting that IgD plays a key role in regulating the survival of self-reactive B cell clones.
3. Kasahara, Taissa de M., and Sudhir Gupta. "IgD+ IgM− B Cells in Common Variable Immunodeficiency." Pathogens 13.2 (2024): 136. https://doi.org/10.3390/pathogens13020136
This article indicates that in patients with common variant immunodeficiency disease (CVID), the proportion of IgD+IgM- memory B cells significantly increases when combined with allergic rhinitis/asthma, suggesting that such cells may play a special role in respiratory allergic reactions, while the function of IgD+IgM-CD27- non-reactive B cells remains normal.
4. Sabouri, Zahra, et al. "IgD attenuates the IgM-induced anergy response in transitional and mature B cells." Nature communications 7.1 (2016): 13381. https://doi.org/10.1038/ncomms13381
This article indicates that IgD maintains the survival of non-reactive B cells by reducing its own antigen reactivity: When B cells express only IgD, although calcium signaling and the expression of core non-responsive genes can be normally induced, compared with the co-expression state of IgM-IgD, the expression of IgD alone will enhance the expression of tolerance markers such as Syndecan-1, thereby inhibiting excessive autoimmune responses and protecting the diversity of the antibody library.
5. Noviski, Mark, et al. "IgM and IgD B cell receptors differentially respond to endogenous antigens and control B cell fate." Elife 7 (2018): e35074. https://doi.org/10.7554/eLife.35074
Studies have found that although IgD can effectively conduct BCR signals in vitro, its reactivity to self-antigens in vivo is significantly lower than that of IgM. Experiments show that B cells expressing only IgD cannot differentiate into short-life plasma cells, and the production of autoantibodies is limited. This indicates that IgD maintains the resting state of autoreactive B cells by reducing the activation threshold of them, thereby inhibiting the occurrence of autoimmune reactions.
Creative Biolabs: IgD Antibodies for Research
Creative Biolabs specializes in the production of high-quality IgD antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Western Blot (WB), Flow Cytometry (FC), and Immunohistochemistry (IHC), and other diagnostic methodologies.
- Custom IgD Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our IgD antibodies, custom preparations, or technical support, contact us at info@creative-biolabs.com.
Reference
- Davies, Anna M., et al. "Crystal structures of the human IgD Fab reveal insights into CH1 domain diversity." Molecular immunology 159 (2023): 28-37. https://doi.org/10.1016/j.molimm.2023.05.006
Anti-IgD antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-FN1 Monoclonal Antibody (D6) (CBMAB-1240CQ)

-
Mouse Anti-BACE1 Recombinant Antibody (CBLNB-121) (CBMAB-1180-CN)

-
Mouse Anti-CRYAB Recombinant Antibody (A4345) (CBMAB-A4345-YC)

-
Mouse Anti-C5B-9 Recombinant Antibody (CBFYA-0216) (CBMAB-X0304-FY)

-
Mouse Anti-ASH1L Monoclonal Antibody (ASH5H03) (CBMAB-1372-YC)

-
Mouse Anti-AQP2 Recombinant Antibody (E-2) (CBMAB-A3358-YC)

-
Mouse Anti-CASQ1 Recombinant Antibody (CBFYC-0863) (CBMAB-C0918-FY)

-
Mouse Anti-AOC3 Recombinant Antibody (CBYY-0014) (CBMAB-0014-YY)

-
Mouse Anti-BAD (Phospho-Ser136) Recombinant Antibody (CBYY-0138) (CBMAB-0139-YY)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Mouse Anti-CEMIP Recombinant Antibody (3C12) (CBMAB-K0296-LY)

-
Mouse Anti-CD24 Recombinant Antibody (SN3) (CBMAB-C1037-CQ)

-
Rabbit Anti-CCN1 Recombinant Antibody (CBWJC-3580) (CBMAB-C4816WJ)

-
Mouse Anti-AZGP1 Recombinant Antibody (CBWJZ-007) (CBMAB-Z0012-WJ)

-
Mouse Anti-CAPZB Recombinant Antibody (CBYY-C0944) (CBMAB-C2381-YY)

-
Mouse Anti-DMD Recombinant Antibody (D1190) (CBMAB-D1190-YC)

-
Mouse Anti-FAS2 Monoclonal Antibody (1D4) (CBMAB-0071-CN)

-
Rat Anti-ADAM10 Recombinant Antibody (V2-179741) (CBMAB-A1103-YC)

-
Mouse Anti-GFAP Recombinant Antibody (5) (CBMAB-G0346-LY)

-
Mouse Anti-ADIPOR2 Recombinant Antibody (V2-179983) (CBMAB-A1369-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






