MYH11 Antibodies
Background
The MYH11 gene encodes the heavy chain 11 of smooth muscle myosin, and is mainly expressed in smooth muscle cells of organs such as blood vessels and digestive tract. The product of this gene, as an important component of contractile proteins, drives molecular motor activities through ATP hydrolysis, thereby regulating the contraction and relaxation functions of smooth muscle and maintaining vascular tension and organ movement. Its mutations are closely related to autosomal dominant hereditary thoracic aortic aneurysm/dissection (TAAD), and were first clearly associated with the genetic study of familial aortic diseases in 2006. Abnormal expression or functional defect of the MYH11 gene will disrupt the cytoskeleton structure and mechanical signal transduction, which makes it an important molecular target for studying vascular development, the pathogenesis of cardiovascular diseases and tissue engineering repair.
Structure of MYH11
The smooth muscle myosin heavy chain 11 encoded by the MYH11 gene has a molecular weight that varies depending on the transcript. The main subtype is approximately 227 kDa. The molecular weight of this protein is highly conserved among different mammalian species, reflecting its central role in the cytoskeleton and contraction functions.
| Species | Human | Mouse | Rat | Bovine |
| Molecular Weight (kDa) | About 227 | About 227 | About 227 | About 227 |
| Primary Structural Differences | Mutations are associated with thoracic aortic diseases. | Used for constructing vascular disease models | Used for studying the physiology of vascular smooth muscle | As a reference in smooth muscle research |
This protein belongs to the myosin II family. Its primary structure consists of a typical head (motor domain), a neck (regulatory light chain binding region), and a long α-helical coiled-coil tail. The secondary structure is dominated by α-helices, especially the tail region forms a dimeric coiled-coil structure, which is crucial for the assembly of myofibrils. The head domain of the protein contains ATPase activity sites and actin binding sites, and its activity directly drives smooth muscle contraction. Its dysfunction is closely related to vascular development and diseases (such as aortic dissection), and it is one of the key molecules in cardiovascular research.
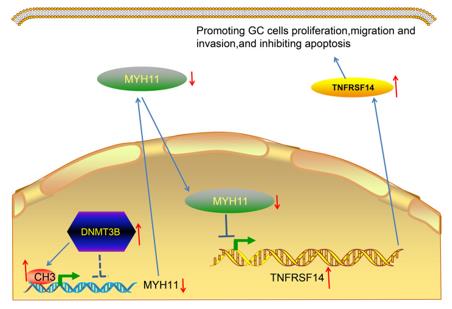 Fig. 1 Promoting GC Progression: DNMT3B Silences MYH11 to Activate TNFRSF14.1
Fig. 1 Promoting GC Progression: DNMT3B Silences MYH11 to Activate TNFRSF14.1
Key structural properties of MYH11:
- The long rod-shaped tail structure composed of α-helical coiled helices
- Spherical head containing atpase active site and actin-binding domain
- Calcium/calmodulin dependent light chain binding to the neck region
Functions of MYH11
The smooth muscle myosin heavy chain 11 encoded by the MYH11 gene has its core function as a molecular motor, driving the contraction and relaxation of smooth muscles in organs such as blood vessels and digestive tracts. Additionally, it is involved in various cellular physiological processes.
| Function | Description |
| Smooth Muscle Contraction | Through the ATPase activity in its head domain, it converts chemical energy into mechanical energy and "walks" along the actin filaments, generating contraction force. |
| Vasodilation Regulation | As the main contractile protein of the smooth muscle in the vascular wall, it directly regulates blood pressure and local blood flow through periodic contraction and relaxation. |
| Organ Movement Support | It drives the regular peristalsis of hollow organs such as the digestive tract, respiratory tract, and urinary reproductive tract, ensuring their normal physiological functions. |
| Cellular skeleton organization | The coiled helical structure at the tail end participates in the formation of myosin fibers, which are key structural components for maintaining cell shape and mechanical integrity. |
| Disease Association | The loss of function or the emergence of dominant negative effects in these genes can disrupt the integrity of the blood vessel walls, which is one of the direct causes of familial thoracic aortic aneurysm/dissection (TAAD). |
Unlike the myosin of skeletal muscle, the contraction driven by MYH11 starts slowly but lasts for a long time, with high energy utilization efficiency. This is highly compatible with its maintenance of tension function. Its dysfunction serves as an important entry point for studying vascular diseases.
Applications of MYH11 and MYH11 Antibody in Literature
1. Wang, Chao, et al. "Single‐cell RNA sequencing reveals the heterogeneity of MYH11+ tumour‐associated fibroblasts between left‐sided and right‐sided colorectal cancer."&emps; Journal of Cellular and Molecular Medicine 28.18 (2024): e70102. https://doi.org/10.1111/jcmm.70102
This study, through single-cell sequencing, revealed significant differences between left and right colon cancers. Left-sided colon cancer is more aggressive and has a poorer prognosis. The specific enriched MYH11+ cancer-associated fibroblasts (CAF) in left-sided colon cancer can promote tumor migration through interaction with macrophages, and they represent a new potential marker for poor prognosis.
2. Zhao, Yang, et al. "Generating endogenous Myh11-driven Cre mice for sex-independent gene deletion in smooth muscle cells." JCI insight 8.14 (2023): e171661. https://doi.org/10.1172/jci.insight.171661
In this study, two novel smooth muscle cell-specific gene editing tools mice were constructed using the CRISPR/Cas9 technology: Myh11-CreNLSP2A and Myh11-CreERT2-P2A. They can achieve efficient, specific and controllable gene editing in both sexes, solving the gender limitations and drug leakage problems existing in traditional models.
3. Qin, Wei, et al. "Comprehensive mutation profile in acute myeloid leukemia patients with RUNX1-RUNX1T1 or CBFB-MYH11 fusions." Turkish Journal of Hematology 39.2 (2022): 84. https://doi.org/10.4274/tjh.galenos.2022.2021.0641
The article indicates that the core binding factor acute myeloid leukemia (CBF-AML) includes two subtypes: t(8;21) and inv(16)/t(16;16), which respectively form the RUNX1-RUNX1T1 and CBFB-MYH11 fusion genes. Although the prognosis is generally good overall, the recurrence rate is as high as 40%, indicating that this subtype exhibits significant clinical and pathological heterogeneity. This study aims to understand the pathogenic mechanism and prognostic differences of this subtype in greater depth by analyzing the accompanying gene mutations through next-generation sequencing.
4. Zhou, Zhen, et al. "MYH11 rare variant augments aortic growth and induces cardiac hypertrophy and heart failure with pressure overload." PLoS genetics 21.7 (2025): e1011394. https://doi.org/10.1371/journal.pgen.1011394
This study investigated the MYH11 p.Glu1892Asp gene variant. Under the pressure load of aortic stenosis, mice carrying this mutation exhibited more severe dilation of the ascending aorta, rupture of elastic fibers, and in male mice, there was also a decline in cardiac function and myocardial hypertrophy, suggesting that abnormal smooth muscle cells can lead to cardiac lesions under pressure overload conditions.
5. Wang, Jianhua, et al. "Interaction between DNMT3B and MYH11 via hypermethylation regulates gastric cancer progression." BMC cancer 21.1 (2021): 914. https://doi.org/10.1186/s12885-021-08653-3
The article indicates that in gastric cancer, the DNA methyltransferase DNMT3B promotes the methylation of the MYH11 gene promoter and inhibits its expression. The low expression of MYH11 relieves its transcriptional inhibition of the downstream gene TNFRSF14, thereby promoting the malignant progression of gastric cancer and being associated with poor prognosis in patients.
Creative Biolabs: MYH11 Antibodies for Research
Creative Biolabs specializes in the production of high-quality MYH11 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom MYH11 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our MYH11 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Wang, Jianhua, et al. "Interaction between DNMT3B and MYH11 via hypermethylation regulates gastric cancer progression." BMC cancer 21.1 (2021): 914. https://doi.org/10.1186/s12885-021-08653-3
Anti-MYH11 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-CARTPT Recombinant Antibody (113612) (CBMAB-C2450-LY)

-
Rabbit Anti-BRCA2 Recombinant Antibody (D9S6V) (CBMAB-CP0017-LY)

-
Mouse Anti-CD59 Recombinant Antibody (CBXC-2097) (CBMAB-C4421-CQ)

-
Rabbit Anti-AP2M1 (Phosphorylated T156) Recombinant Antibody (D4F3) (PTM-CBMAB-0610LY)

-
Mouse Anti-FAS2 Monoclonal Antibody (1D4) (CBMAB-0071-CN)

-
Mouse Anti-CCNH Recombinant Antibody (CBFYC-1054) (CBMAB-C1111-FY)

-
Mouse Anti-ASTN1 Recombinant Antibody (H-9) (CBMAB-1154-CN)

-
Mouse Anti-AAV8 Recombinant Antibody (V2-634028) (CBMAB-AP022LY)

-
Mouse Anti-ALB Recombinant Antibody (V2-55272) (CBMAB-H0819-FY)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

-
Mouse Anti-C5b-9 Recombinant Antibody (aE11) (CBMAB-AO138LY)

-
Mouse Anti-CCT6A/B Recombinant Antibody (CBXC-0168) (CBMAB-C5570-CQ)

-
Mouse Anti-CORO1A Recombinant Antibody (4G10) (V2LY-1206-LY806)

-
Rabbit Anti-ABL1 (Phosphorylated Y185) Recombinant Antibody (V2-443434) (PTM-CBMAB-0001YC)

-
Mouse Anti-CCND2 Recombinant Antibody (DCS-3) (CBMAB-G1318-LY)

-
Human Anti-SARS-CoV-2 S1 Monoclonal Antibody (CBFYR-0120) (CBMAB-R0120-FY)

-
Mouse Anti-FPR2 Recombinant Antibody (1D6) (CBMAB-F2628-CQ)

-
Mouse Anti-APOA1 Monoclonal Antibody (CBFYR0637) (CBMAB-R0637-FY)

-
Mouse Anti-ENO1 Recombinant Antibody (8G8) (CBMAB-E1329-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






