PGK1 Antibodies
Background
PGK1 is a crucial glycolytic enzyme that is mainly present in the cytoplasm of all eukaryotic cells. This enzyme catalyzes the key steps in the glycolytic pathway, converting 1,3-diphosphoglyceric acid into 3-phosphoglyceric acid while generating ATP, thereby providing an energy foundation for cellular metabolism. Due to its significant role, the expression and activity of PGK1 are closely related to cell proliferation, stress responses, and various disease processes. This enzyme was first isolated and identified in the 1960s, and its three-dimensional structure was resolved through X-ray crystallography in the 1980s, becoming a classic model for studying enzyme kinetics and metabolic regulation. The highly conserved domain and allosteric regulation mechanism of PGK1 remain important entry points for studying the association between energy metabolism and diseases, providing a theoretical basis for understanding cell adaptation mechanisms and related drug development.
Structure of PGK1
PGK1 (Phosphoglycerate Kinase 1) is a key enzyme in glycolysis with a molecular weight of approximately 44.7 kDa. Its precise molecular weight may vary slightly among different species due to minor differences in amino acid sequences.
| Species | Human | Mouse | Rat | Yeast |
| Molecular Weight (kDa) | 44.7 | 44.6 | 44.8 | 44.5 |
| Primary Structural Differences | Highly conservative catalytic core | Extremely similar to humans in origin | The sequence similarity exceeds 95%. | Having the same functional domains |
The PGK1 protein is composed of approximately 417 amino acids and its three-dimensional structure exhibits a characteristic "bifacial" spherical conformation. This structure results from its clearly defined two-domain architecture: the N-terminal domain and the C-terminal domain, which are connected by a flexible hinge region. The catalytic active site is located in the gap between the two domains, and this spatial arrangement is crucial for substrate binding and phosphate group transfer. The secondary structure of the protein is mainly composed of α-helices and β-sheets, which together form a stable hydrophobic core and an accurate substrate binding pocket. A key lysine residue (such as Lys219 in human PGK1) plays a central role in the catalytic mechanism, responsible for stabilizing the transition state intermediate and promoting ATP synthesis.
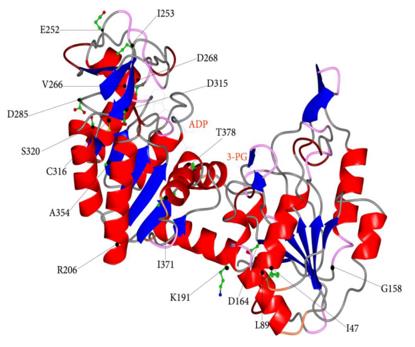 Fig. 1 Ribbons representation of human PGK1 in open conformation.1
Fig. 1 Ribbons representation of human PGK1 in open conformation.1
Key structural properties of PGK1:
- Highly conservative bistructural globular conformation
- Catalytic active site is located in the n-terminal and flexible hinge between the c-terminal domain structure zone
- Phosphorylation catalytic centers dependent on ATP binding and transfer
- Key lysine and aspartic acid residues cooperate to regulate substrate binding and energy conversion
Functions of PGK1
The core function of the PGK1 gene is to catalyze the key steps in glycolysis and generate ATP. However, it has also been found to be involved in a variety of important cellular processes, including regulating cell proliferation, responding to oxidative stress, and participating in DNA repair.
| Function | Description |
| Energy Production | Catalyzes the conversion of 1,3-diphosphoglycerate to 3-phosphoglycerate and ATP, which is one of the main ATP generation steps in the glycolysis pathway. |
| Metabolic Hub | As the central enzyme of glycolysis, its activity directly regulates the flux and affects the energy supply of the entire cell as well as the biosynthetic precursors. |
| Oxidative Stress Response | Under oxidative stress, some PGK1 can be translocated to the mitochondria, thereby regulating mitochondrial function to protect the cells. |
| DNA Repair Involvement | The research has found that PGK1 can interact with DNA repair proteins and plays a role in processes such as base excision repair, maintaining genomic stability. |
| Cellular Proliferation Regulation | The activity of PGK1 is closely related to the cell cycle progression and the growth of tumor cells, as it affects energy metabolism and signaling pathways. |
The catalytic kinetics of PGK1 conforms to the classic Michaelis-Menten equation. Its activity is inhibited by the allosteric product ATP and regulated by the substrate level, which ensures the homeostasis and on-demand supply of cellular energy metabolism.
Applications of PGK1 and PGK1 Antibody in Literature
1. Valentini, Giovanna, Maristella Maggi, and Angel L. Pey. "Protein stability, folding and misfolding in human PGK1 deficiency." Biomolecules 3.4 (2013): 1030-1052. https://doi.org/10.3390/biom3041030
This article reviews the effects of human phosphoglycerate kinase 1 (hPGK1) mutations on protein folding and aggregation. It is found that pathogenic mutations alter the folding cooperativity and the denaturation transition state structure, thereby intensifying the tendency of protein aggregation. The study reveals the mechanism of hPGK1 misfolding, providing a theoretical basis for therapeutic strategies targeting protein stability and folding ability.
2. Liu, Yu, et al. "Effects of PGK1 on immunoinfiltration by integrated single-cell and bulk RNA-sequencing analysis in sepsis." Frontiers in Immunology 15 (2024): 1449975. https://doi.org/10.3389/fimmu.2024.1449975
The study found that PGK1 was significantly upregulated in patients with sepsis (with an AUC > 0.9), suggesting its potential as a diagnostic marker. PGK1 is associated with inflammatory pathways and the infiltration of various immune cells, and may play an important role in the immune regulation of sepsis.
3. Chen, Zhenzhen, et al. "mcPGK1-dependent mitochondrial import of PGK1 promotes metabolic reprogramming and self-renewal of liver TICs." Nature Communications 14.1 (2023): 1121. https://doi.org/10.1038/s41467-023-36651-5
The study found that the mitochondrial circular RNA mcPGK1 is highly expressed in liver cancer initiating cells. It regulates metabolic reprogramming through the PGK1-PDK1-PDH axis, inhibits oxidative phosphorylation and promotes glycolysis, thereby activating the Wnt/β-catenin pathway and driving the self-renewal of liver cancer initiating cells.
4. Liu, Hao, et al. "PRMT1-mediated PGK1 arginine methylation promotes colorectal cancer glycolysis and tumorigenesis." Cell Death & Disease 15.2 (2024): 170. https://doi.org/10.1038/s41419-024-06544-6
The study found that PRMT1 methylates the PGK1 protein (meR206-PGK1) and enhances its phosphorylation (pS203-PGK1), thereby inhibiting mitochondrial function and promoting glycolysis, driving the proliferation and tumor formation of colorectal cancer. meR206-PGK1 is associated with poor prognosis in patients and is expected to become a new prognostic biomarker and therapeutic target.
5. Cui, Jiayong, et al. "Targeting PGK1: A New Frontier in Breast Cancer Therapy Under Hypoxic Conditions." Current Issues in Molecular Biology 46.11 (2024): 12214-12229. https://doi.org/10.3390/cimb46110725
This article reviews the crucial role of PGK1 in the hypoxic microenvironment of breast cancer. PGK1 regulates the glycolytic process and hypoxia-inducible factors, thereby influencing tumor energy metabolism and progression. The mechanism research of PGK1 provides important theoretical basis for the development of new therapeutic targets and strategies.
Creative Biolabs: PGK1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality PGK1 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom PGK1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our PGK1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Valentini, Giovanna, Maristella Maggi, and Angel L. Pey. "Protein stability, folding and misfolding in human PGK1 deficiency." Biomolecules 3.4 (2013): 1030-1052. https://doi.org/10.3390/biom3041030
Anti-PGK1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ATM Recombinant Antibody (2C1) (CBMAB-A3970-YC)

-
Mouse Anti-EPO Recombinant Antibody (CBFYR0196) (CBMAB-R0196-FY)

-
Mouse Anti-GFAP Recombinant Antibody (20) (CBMAB-G2914-LY)

-
Mouse Anti-ADV Recombinant Antibody (V2-503423) (CBMAB-V208-1364-FY)

-
Mouse Anti-AKR1C3 Recombinant Antibody (V2-12560) (CBMAB-1050-CN)

-
Rabbit Anti-ABL1 (Phosphorylated Y185) Recombinant Antibody (V2-443434) (PTM-CBMAB-0001YC)

-
Mouse Anti-ANXA7 Recombinant Antibody (A-1) (CBMAB-A2941-YC)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

-
Mouse Anti-CCL18 Recombinant Antibody (64507) (CBMAB-C7910-LY)

-
Mouse Anti-CCNH Recombinant Antibody (CBFYC-1054) (CBMAB-C1111-FY)

-
Mouse Anti-BMI1 Recombinant Antibody (CBYC-P026) (CBMAB-P0108-YC)

-
Mouse Anti-CD33 Recombinant Antibody (6C5/2) (CBMAB-C8126-LY)

-
Mouse Anti-ATP1B3 Recombinant Antibody (1E9) (CBMAB-A4021-YC)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7697) (CBMAB-1869CQ)

-
Mouse Anti-BAX Recombinant Antibody (CBYY-0216) (CBMAB-0217-YY)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Mouse Anti-AKT1 Recombinant Antibody (V2-180546) (CBMAB-A2070-YC)

-
Mouse Anti-CORO1A Recombinant Antibody (4G10) (V2LY-1206-LY806)

-
Mouse Anti-ATG5 Recombinant Antibody (9H197) (CBMAB-A3945-YC)

-
Mouse Anti-ENO1 Recombinant Antibody (CBYC-A950) (CBMAB-A4388-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






