CCL17 Antibodies
Background
CCL17 is a small secreted protein encoded by a gene in the CC subfamily of chemokines. It is mainly expressed in thymic dendritic cells, endothelial cells, and immune cells in certain inflammatory sites. As a specific ligand for the CCR4 receptor, this protein can selectively attract regulatory T cells (Treg) and type 2 helper T cells (Th2), thereby playing a crucial regulatory role in immune surveillance, inflammatory responses, and allergic diseases. CCL17 was first identified in 1996, and the clarification of its structure and function has deepened our understanding of the chemokine signaling network and the immune microenvironment. In particular, it provides potential therapeutic targets for Th2-mediated diseases such as asthma and atopic dermatitis. Related research continues to drive the development of immunology and clinical medicine.
Structure of CCL17
CCL17 is a relatively small chemokine with a molecular weight of approximately 8-10 kDa. This molecular weight may vary slightly among different species due to minor differences in amino acid sequences.
| Species | Human | Mouse | Rat |
|---|---|---|---|
| Molecular Weight (kDa) | About 8.0 | About 8.2 | About 8.1 |
| Primary Structural Differences | Contains signal and mature peptides with four conserved cysteine residues | The N-terminal sequence shows species-specific differences, while the core structure is similar. | High homology with mice, and the functional domains are conserved |
This protein is composed of approximately 71 amino acid residues (in its mature form), and its primary structure forms a stable β-sheet and random coil conformation through disulfide bonds (Cys11-Cys52 and Cys12-Cys34), constituting the typical "three-leaf-clover" like spatial folding characteristic of chemokines. Its tertiary structure forms a hydrophobic core and specifically binds to the CCR4 receptor through the extended region at the N-terminus. Its functional activity is highly dependent on the integrity of the first few amino acid residues at the N-terminus (such as serine), which are crucial for receptor recognition and activation.
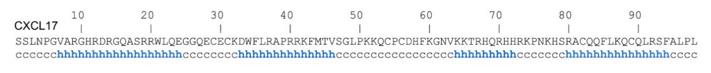 Fig. 1 Secondary structure prediction of human CXCL17 using DSC webserver.1
Fig. 1 Secondary structure prediction of human CXCL17 using DSC webserver.1
Key structural properties of CCL17:
- Typical chemokine folding structure, including reverse parallel β-folding and C-terminal α-helix
- Core region consists of two disulfide bonds of conservative (Cys11-Cys52 and Cys12-Cys34) for stability
- N-terminal extension area contains receptors and activates the key sequence
- The tertiary structure formation composed of hydrophobic residues receptor interface, that can bind CCR4 receptor specifically
Functions of CCL17
The main function of CCL17 (thymus and activation-regulated chemokine) is to act as a chemokine, selectively recruiting specific immune cells. However, it is also involved in a wide range of immune regulation and pathological processes.
| Function | Description |
| Immune cell chemotaxis | As a ligand for the CCR4 receptor, it selectively recruits regulatory T cells and type 2 helper T cells to the inflammatory or draining lymph node sites. |
| Immune Tolerance Regulation | By recruiting regulatory T cells, it participates in the establishment and maintenance of immune tolerance in the thymus and periphery, preventing excessive immune responses. |
| Inflammation and Allergic Responses | Highly expressed in allergic diseases (such as atopic dermatitis, asthma) and chronic inflammation, driving Th2-type immune responses and tissue inflammation. |
| Tumor Microenvironment Regulation | It is expressed in various tumors and, by recruiting regulatory T cells and other means, it shapes an inhibitory tumor immune microenvironment, facilitating immune evasion. |
| Vascularization Impact | Some studies suggest that it may indirectly affect pathological vascularization, especially during the tissue remodeling associated with Th2 inflammation. |
Unlike some broad-spectrum chemotactic factors, CCL17 exhibits highly specific receptor selectivity (primarily acting on CCR4), which determines its precise regulatory role in the directed migration of immune cells and the shaping of the local microenvironment.
Applications of CCL17 and CCL17 Antibody in Literature
1. Denisov, Stepan S. "CXCL17: The black sheep in the chemokine flock." Frontiers in Immunology 12 (2021): 712897. https://doi.org/10.3389/fimmu.2021.712897
The article indicates that CXCL17 is the latest discovered CXC-type chemokine. Although its structure conforms to the folding characteristics of chemokines, there is still controversy surrounding it, and whether it is a true member of the chemokine family remains undetermined.
2. Hueso, Luisa, et al. "CCL17 and CCL22 chemokines are upregulated in human obesity and play a role in vascular dysfunction." Frontiers in Endocrinology 14 (2023): 1154158. https://doi.org/10.3389/fendo.2023.1154158
Studies have shown that in the blood and visceral fat of obese patients, the expression of chemokines CCL17/CCL22 and their receptor CCR4 is significantly increased, and it is positively correlated with body mass index and insulin resistance. Inhibiting CCR4 can alleviate inflammation and the adhesion of leukocytes to endothelial cells, suggesting that targeting this pathway may be a new strategy for the treatment of obesity.
3. Zou, Chenghui, et al. "Association between CCL5, CCL11, and CCL17 polymorphisms and atopic dermatitis risk: A systematic review and meta-analysis." Medicine 103.8 (2024): e36897. https://doi.org/10.1097/MD.0000000000036897
This meta-analysis indicates that the -403G/A and -28C/G polymorphisms of the CCL5 gene are significantly associated with the risk of atopic dermatitis. However, the study on the CCL17 polymorphism involved in the analysis did not reveal a clear association.
4. Li, He, et al. "CCL17‐CCR4 axis contributes to the onset of vitiligo in mice." Immunity, Inflammation and Disease 9.3 (2021): 702-709. https://doi.org/10.1002/iid3.423
This study is the first to confirm that the expression of the CCL17-CCR4 axis is upregulated in the lesion areas of patients with vitiligo. In animal models, knocking out this axis or using neutralizing antibodies to block it can significantly inhibit the migration of CD8⁺ T cells to the skin and alleviate the symptoms of white patches, suggesting that it is a potential therapeutic target.
5. Zhang, Yang, et al. "CCL17 acts as a novel therapeutic target in pathological cardiac hypertrophy and heart failure." Journal of Experimental Medicine 219.8 (2022): e20200418. https://doi.org/10.1084/jem.20200418
The research has found that the level of CCL17 increases with age and is associated with cardiac dysfunction. Animal experiments have confirmed that knocking out the CCL17 gene or using neutralizing antibodies can significantly inhibit aging and cardiac hypertrophy and fibrosis induced by angiotensin II, suggesting that it is a new target for the treatment of age-related heart failure.
Creative Biolabs: CCL17 Antibodies for Research
Creative Biolabs specializes in the production of high-quality CCL17 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom CCL17 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our CCL17 antibodies, custom preparations, or technical support, contact us at info@creative-biolabs.com.
Reference
- Denisov, Stepan S. "CXCL17: The black sheep in the chemokine flock." Frontiers in Immunology 12 (2021): 712897. Distributed under the same Creative Commons license CC BY 4.0 as the original. Cropped from the original figure. https://doi.org/10.3389/fimmu.2021.712897
Anti-CCL17 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ATG5 Recombinant Antibody (9H197) (CBMAB-A3945-YC)

-
Armenian hamster Anti-CD40 Recombinant Antibody (HM40-3) (CBMAB-C10365-LY)

-
Mouse Anti-EMP3 Recombinant Antibody (CBFYE-0100) (CBMAB-E0207-FY)

-
Mouse Anti-ADGRL2 Recombinant Antibody (V2-58519) (CBMAB-L0166-YJ)

-
Mouse Anti-CCDC6 Recombinant Antibody (CBXC-0106) (CBMAB-C5397-CQ)

-
Mouse Anti-AMOT Recombinant Antibody (CBYC-A564) (CBMAB-A2552-YC)

-
Mouse Anti-FLT1 Recombinant Antibody (11) (CBMAB-V0154-LY)

-
Rabbit Anti-CCL5 Recombinant Antibody (R0437) (CBMAB-R0437-CN)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

-
Mouse Anti-FOXA3 Recombinant Antibody (2A9) (CBMAB-0377-YC)

-
Rabbit Anti-ATF4 Recombinant Antibody (D4B8) (CBMAB-A3872-YC)

-
Mouse Anti-ARID1B Recombinant Antibody (KMN1) (CBMAB-A3546-YC)

-
Mouse Anti-APCS Recombinant Antibody (CBYC-A663) (CBMAB-A3054-YC)

-
Mouse Anti-DMPK Recombinant Antibody (CBYCD-324) (CBMAB-D1200-YC)

-
Mouse Anti-CASP8 Recombinant Antibody (CBYY-C0987) (CBMAB-C2424-YY)

-
Mouse Anti-ALX1 Recombinant Antibody (96k) (CBMAB-C0616-FY)

-
Rabbit Anti-AKT3 Recombinant Antibody (V2-12567) (CBMAB-1057-CN)

-
Mouse Anti-C4B Recombinant Antibody (CBYY-C2996) (CBMAB-C4439-YY)

-
Rabbit Anti-ADRA1A Recombinant Antibody (V2-12532) (CBMAB-1022-CN)

-
Rabbit Anti-Acetyl-Histone H3 (Lys36) Recombinant Antibody (V2-623395) (CBMAB-CP0994-LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






