GSDMD Antibodies
Background
GSDMD is a pore-forming protein that is widely expressed in immune cells such as neutrophils and macrophages. It usually exists in the cytoplasm in an auto-inhibited state. After being cleaved by caspase-1 or caspase-4/5/11, the N-terminal domain of this protein can oligomerize and form pores on the cell membrane, mediating cell pyroptosis and the release of inflammatory factors. It is a key molecule connecting the activation of inflammasomes and the execution of pyroptosis. Since it was simultaneously identified as a pyroptosis effector protein by multiple research teams in 2015, GSDMD has rapidly become a core target in the study of innate immunity and inflammatory diseases. The elucidation of its mechanism not only promotes the paradigm shift in the field of cell death but also provides new intervention strategies for the treatment of excessive inflammation-related diseases such as sepsis and autoimmune diseases.
Structure of GSDMD
GSDMD is a pore-forming protein with a molecular weight of approximately 48 kDa. Its precise molecular weight may vary slightly among different species due to differences in amino acid sequences.
| Species | Human | Mouse | Rat |
| Molecular Weight (kDa) | About 48 | About 48 | About 48 |
| Primary Structural Differences | Containing a C-terminal autoinhibitory domain | High sequence homology | Conservative functional domain |
This protein is composed of approximately 480 amino acids. Its three-dimensional structure includes an N-terminal pore-forming domain (about 275 amino acids) in a β-barrel-like conformation and a C-terminal inhibitory domain composed of multiple helices. The core functional structure is the pore-forming module located at the N-terminal, which can oligomerize on the cell membrane to form a pore channel with a diameter of approximately 10-21 nanometers. Activated caspase will specifically cleave at the GSDMD linker region (such as human Asp275), relieving the self-inhibition of the C-terminal, thereby releasing the N-terminal domain to execute its pore-forming function.
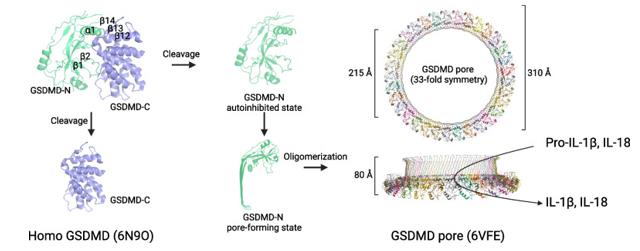 Fig. 1 Schematic representation of the structural changes in GSDMD during pyroptosis.1
Fig. 1 Schematic representation of the structural changes in GSDMD during pyroptosis.1
Key structural properties of GSDMD:
- Dual domain structure of N-terminal pore-forming domain and C-terminal inhibitory domain
- Caspase-specific cleavage sites within the linker region
- Oligomerization interface of the N-terminal domain
Functions of GSDMD
The main function of GSDMD is to act as the executor of cell pyroptosis. The core mechanism of its activation is to respond to upstream inflammatory signals and form pores on the cell membrane. However, it also exhibits diverse biological effects in different cellular environments and under various signaling conditions.
| Function | Description |
| Pyroptosis Execution | After being activated by caspase cleavage, its N-terminal domain oligomerizes and forms channels on the cell membrane, causing cell swelling and rupture, and releasing contents. |
| Release of Inflammatory Mediators | The formation of cell membrane channels facilitates the release of pro-inflammatory cytokines such as IL-1β and IL-18 into the extracellular space, amplifying the inflammatory signal. |
| Membrane Permeability Regulation | In addition to the plasma membrane, pores can also be formed on intracellular membrane systems such as the mitochondrial membrane, affecting membrane potential and possibly promoting the release of other factors related to cell death. |
| Bacterial Defense | In macrophages, by inducing pyroptosis to lyse infected cells, intracellular pathogens can be exposed to the extracellular immune surveillance, thereby limiting their spread. |
| Non-classical pyroptosis pathway | When caspase-1 is absent, caspase-4/5/11 directly recognize and cleave GSDMD, triggering the pyroptosis through a pathway known as "non-classical" inflammasome pathway. |
The activation process of GSDMD exhibits a "switch-like" on-off characteristic, which is different from the gradual oxygen-binding curve of myoglobin; once the inhibitory domain is removed, its pore-forming activity will rapidly burst forth, ensuring that the inflammatory response can be initiated quickly and thoroughly.
Applications of GSDMD and GSDMD Antibody in Literature
1. Li, Yujuan, and Bin Guo. "GSDMD-mediated pyroptosis: molecular mechanisms, diseases and therapeutic targets." Molecular Biomedicine 6.1 (2025): 11. https://doi.org/10.1186/s43556-025-00249-8
The article indicates that Gasdermin D (GSDMD) is a key executor protein of pyroptosis and has complex functions in different diseases: it often aggravates damage in inflammation and can both promote immune escape and kill cancer cells within tumors. Targeting GSDMD offers a new direction for precision treatment. Currently, the related inhibitors are still in the preclinical research stage.
2. Li, Shanshan, et al. "NLRP3/caspase-1/GSDMD–mediated pyroptosis exerts a crucial role in astrocyte pathological injury in mouse model of depression." JCI insight 6.23 (2021): e146852. https://doi.org/10.1172/jci.insight.146852
Research has found that astrocyte pyroptosis depends on GSDMD and is the key mechanism leading to the loss of this cell in the hippocampus of depression model mice. Targeting the NLRP3/Casp-1/GSDMD pathway can inhibit pyroptosis and improve depressive behaviors, providing a new potential strategy for treatment.
3. Wang, Yujia, et al. "GSDMD-dependent neutrophil extracellular traps promote macrophage-to-myofibroblast transition and renal fibrosis in obstructive nephropathy." Cell death & disease 13.8 (2022): 693. https://doi.org/10.1038/s41419-022-05138-4
This study reveals that in obstructive nephropathy, neutrophils form NETs through the caspase-11/GSDMD pathway, which in turn activates the transformation of macrophages into myofibroblasts and drives renal fibrosis. Targeting this pathway can become a new therapeutic strategy.
4. Karmakar, Mausita, et al. "N-GSDMD trafficking to neutrophil organelles facilitates IL-1β release independently of plasma membrane pores and pyroptosis." Nature communications 11.1 (2020): 2212. https://doi.org/10.1038/s41467-020-16043-9
Research has found that there are key differences in the mechanism of action of GSDMD between neutrophils and macrophages: in neutrophils, N-GSDMD does not cause cytosis but acts on the granulotropin granules and autophagosomes, releasing IL-1β through autophagy-dependent pathways.
5. Yao, Yihan, et al. "A CD4+ T lymphocyte–specific TCR/GSDMD/IL-2 axis facilitates antitumor immunity." The Journal of Clinical Investigation 135.15 (2025). https://doi.org/10.1172/JCI191119
Research has found that GSDMD exerts a new non-ptosis function in tumor-infiltrating T cells: it forms channels through caspase-8 cutting, promoting calcium influx and IL-2 secretion, thereby enhancing the auxiliary effect of CD4+ T cells on CD8+ T cells and improving anti-tumor immunity and immunotherapy response.
Creative Biolabs: GSDMD Antibodies for Research
Creative Biolabs specializes in the production of high-quality GSDMD antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom GSDMD Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our GSDMD antibodies, custom preparations, or technical support, contact us at email.
Reference
- Li, Yujuan, and Bin Guo. "GSDMD-mediated pyroptosis: molecular mechanisms, diseases and therapeutic targets." Molecular Biomedicine 6.1 (2025): 11. https://doi.org/10.1186/s43556-025-00249-8
Anti-GSDMD antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ADRB2 Recombinant Antibody (V2-180026) (CBMAB-A1420-YC)

-
Mouse Anti-CALR Recombinant Antibody (CBFYC-0763) (CBMAB-C0818-FY)

-
Rabbit Anti-ABL1 (Phosphorylated Y185) Recombinant Antibody (V2-443434) (PTM-CBMAB-0001YC)

-
Mouse Anti-ARIH1 Recombinant Antibody (C-7) (CBMAB-A3563-YC)

-
Mouse Anti-ENO1 Recombinant Antibody (CBYC-A950) (CBMAB-A4388-YC)

-
Mouse Anti-BBS2 Recombinant Antibody (CBYY-0253) (CBMAB-0254-YY)

-
Mouse Anti-BACE1 Recombinant Antibody (CBLNB-121) (CBMAB-1180-CN)

-
Mouse Anti-F11R Recombinant Antibody (402) (CBMAB-0026-WJ)

-
Mouse Anti-ARHGAP5 Recombinant Antibody (54/P190-B) (CBMAB-P0070-YC)

-
Mouse Anti-CTCF Recombinant Antibody (CBFYC-2371) (CBMAB-C2443-FY)

-
Mouse Anti-BHMT Recombinant Antibody (CBYY-0547) (CBMAB-0550-YY)

-
Rat Anti-ADAM10 Recombinant Antibody (V2-179741) (CBMAB-A1103-YC)

-
Mouse Anti-APOA1 Monoclonal Antibody (CBFYR0637) (CBMAB-R0637-FY)

-
Mouse Anti-AQP2 Recombinant Antibody (E-2) (CBMAB-A3358-YC)

-
Mouse Anti-FYN Recombinant Antibody (10) (CBMAB-S6332-CQ)

-
Rat Anti-AChR Recombinant Antibody (V2-12500) (CBMAB-0990-CN)

-
Mouse Anti-C1QC Recombinant Antibody (CBFYC-0600) (CBMAB-C0654-FY)

-
Mouse Anti-ACVR1C Recombinant Antibody (V2-179685) (CBMAB-A1041-YC)

-
Mouse Anti-CD164 Recombinant Antibody (CBFYC-0077) (CBMAB-C0086-FY)

-
Mouse Anti-BPGM Recombinant Antibody (CBYY-1806) (CBMAB-2155-YY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






