HMGA2 Antibodies
Background
HMGA2 is a non-histone chromatin-binding protein that mainly exists during the embryonic development stage of mammals and in some adult tissues. This protein binds to the minor grooves of DNA through its AT-hook domain, thereby altering the chromatin conformation and regulating the transcriptional activity of various genes. It plays a crucial role in cell proliferation, differentiation, and embryonic development. Abnormal expression of this gene is closely related to the occurrence and development of various tumors, and its overexpression often promotes epithelial-mesenchymal transition of cells and accelerates tumor metastasis. Since it was systematically studied in the 1990s, HMGA2 has become a research hotspot due to its important role in developmental biology and oncology. The study of its molecular mechanism has deepened our understanding of chromatin remodeling, gene expression regulation, and epigenetic regulation in disease occurrence.
Structure of HMGA2
HMGA2 is a non-histone chromatin regulatory factor with a molecular weight of approximately 12.5 kDa. Due to the presence of multiple isoforms generated by alternative splicing, there will be some fluctuations in its molecular weight.
| Species | Human | Mouse | Rat | Zebrafish | Bovine |
| Molecular Weight (kDa) | ~12.5 | ~12.2 | ~12.3 | ~11.8 | ~12.4 |
| Primary Structural Differences | Containing three highly conserved AT-hook domains | Highly similar to humans in terms of structure and function | Structurally highly conservative | Homologous genes exist and the structure is similar to that of mammals | Structure of domain and human are basically identical |
The HMGA2 protein is composed of approximately 109 amino acids and has a relatively loose tertiary structure, lacking the typical globular folding. The core function of this protein relies on its three consecutive AT-hook domains (formed by short peptides rich in basic amino acids), which can specifically recognize and bind to the DNA grooves rich in AT base pairs. By binding to DNA, HMGA2 can alter the local spatial conformation of chromatin, thereby acting as a "transcriptional regulatory scaffold" to recruit or exclude other transcription factors and chromatin modification complexes, thereby achieving precise regulation of downstream gene expression.
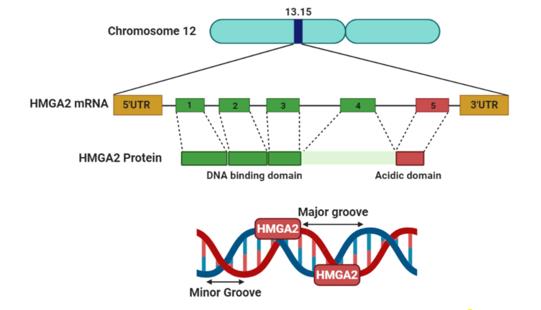 Fig. 1 Schematic of the high mobility group protein 2 (HMGA2) gene and protein.1
Fig. 1 Schematic of the high mobility group protein 2 (HMGA2) gene and protein.1
Key structural properties of HMGA2:
- Contains three consecutive AT-hook functional domains
- Lacks a stable three-dimensional structure and is a naturally disordered protein.
- Rich in basic amino acids (arginine, lysine)
Functions of HMGA2
The main function of the HMGA2 gene is to act as a chromatin structure regulatory factor in participating in gene transcription regulation. However, it is also widely involved in various pathological and physiological processes, especially in embryonic development and tumor formation.
| Function | Description |
| Chromatin Remodeling | By binding to AT-rich DNA sequences through its AT-hook domain, it alters the local chromatin spatial conformation, providing a binding platform for other transcription factors. |
| Embryo Development Regulation | It is highly expressed in the early stage of the embryo, regulating the expression of various genes (such as IGF2, PLAG1) related to cell proliferation, differentiation and organ formation. |
| Cell Proliferation and Differentiation | Promote the self-renewal of stem cells and inhibit their premature differentiation, maintaining the plasticity of tissues. |
| Tumor Formation and Metastasis | It is abnormally highly expressed in various malignant tumors. By inducing epithelial-mesenchymal transition (EMT) and regulating cell cycle-related genes, it promotes tumor invasion and metastasis. |
| Association between fat metabolism and obesity | The genetic polymorphisms are associated with individual fat content, body weight, and the risk of metabolic syndrome, and they affect the proliferation and differentiation of fat precursor cells. |
The HMGA2 protein itself does not possess the classic domains for direct activation or inhibition of transcription. Instead, it indirectly and globally affects the gene expression network by altering the accessibility of chromatin. This characteristic of "architecture transcription factor" enables it to play a crucial and complex regulatory role in development and diseases.
Applications of HMGA2 and HMGA2 Antibody in Literature
1. Wang, Xin, et al. "HMGA2 facilitates colorectal cancer progression via STAT3-mediated tumor-associated macrophage recruitment." Theranostics 12.2 (2022): 963. https://doi.org/10.7150/thno.65411
The article indicates that in colorectal cancer, HMGA2 binds to the STAT3 promoter to activate its transcription, promoting the secretion of CCL2, which then recruits and polarizes into M2-type macrophages, accelerating tumor progression. The HMGA2/STAT3/CCL2 axis may represent a potential therapeutic target.
2. Luo, Ziyang, et al. "HMGA2 alleviates ferroptosis by promoting GPX4 expression in pancreatic cancer cells." Cell Death & Disease 15.3 (2024): 220. https://doi.org/10.1038/s41419-024-06592-y
The article indicates that HMGA2 is highly expressed in pancreatic cancer. It inhibits ferroptosis by activating the transcription of GPX4 and the mTORC1 pathway, thereby enhancing cancer cell proliferation, metastasis, and chemotherapy resistance, making it a potential therapeutic target.
3. Mansoori, Behzad, et al. "HMGA2 as a critical regulator in cancer development." Genes 12.2 (2021): 269. https://doi.org/10.3390/genes12020269
The article indicates that HMGA2 is a DNA-binding protein that is highly expressed during embryonic development and is limited in adulthood. It re-expresses in various malignant tumors and drives tumor occurrence and development through multiple mechanisms such as regulating the cell cycle, DNA repair, epithelial-mesenchymal transition, and cancer stem cell phenotype. It is a potential therapeutic target.
4. Hashemi, Mehrdad, et al. "HMGA2 regulation by miRNAs in cancer: affecting cancer hallmarks and therapy response." Pharmacological Research 190 (2023): 106732. https://doi.org/10.1016/j.phrs.2023.106732
The article indicates that HMGA2 is a cancer-causing protein involved in chromatin structure, and it promotes the malignant progression of tumors by regulating key pathways such as Wnt/β-catenin and mTOR. Non-coding RNAs (especially miRNAs) can regulate the expression of HMGA2, and targeting this axis has potential therapeutic value.
5. Ma, Qing, et al. "HMGA2 promotes cancer metastasis by regulating epithelial–mesenchymal transition." Frontiers in oncology 14 (2024): 1320887. https://doi.org/10.3389/fonc.2024.1320887
The article indicates that HMGA2 is overexpressed in various cancers. It induces EMT through the TGFβ/SMAD, MAPK and WNT/β-catenin signaling pathways, promoting tumor migration and invasion. Non-coding RNAs can regulate this process, providing a new strategy for targeted therapy.
Creative Biolabs: HMGA2 Antibodies for Research
Creative Biolabs specializes in the production of high-quality HMGA2 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom HMGA2 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our HMGA2 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Mansoori, Behzad, et al. "HMGA2 as a critical regulator in cancer development." Genes 12.2 (2021): 269. https://doi.org/10.3390/genes12020269
Anti-HMGA2 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Mouse Anti-F11R Recombinant Antibody (402) (CBMAB-0026-WJ)

-
Mouse Anti-FTH1 Recombinant Antibody (CBXF-1896) (CBMAB-F3426-CQ)

-
Mouse Anti-C5b-9 Recombinant Antibody (aE11) (CBMAB-AO138LY)

-
Mouse Anti-ALB Recombinant Antibody (V2-363290) (CBMAB-S0173-CQ)

-
Mouse Anti-EIF4G1 Recombinant Antibody (2A9) (CBMAB-A2544-LY)

-
Mouse Anti-FLI1 Recombinant Antibody (CBXF-0733) (CBMAB-F0435-CQ)

-
Mouse Anti-CCDC6 Recombinant Antibody (CBXC-0106) (CBMAB-C5397-CQ)

-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Rat Anti-C5AR1 Recombinant Antibody (8D6) (CBMAB-C9139-LY)

-
Mouse Anti-CASP7 Recombinant Antibody (10-01-62) (CBMAB-C2005-LY)

-
Mouse Anti-Acetyl-α-Tubulin (Lys40) Recombinant Antibody (V2-623485) (CBMAB-CP2897-LY)

-
Mouse Anti-dsDNA Recombinant Antibody (22) (CBMAB-AP1954LY)

-
Mouse Anti-FPR2 Recombinant Antibody (1D6) (CBMAB-F2628-CQ)

-
Mouse Anti-CALR Recombinant Antibody (CBFYC-0763) (CBMAB-C0818-FY)

-
Mouse Anti-CORO1A Recombinant Antibody (4G10) (V2LY-1206-LY806)

-
Mouse Anti-APP Recombinant Antibody (5C2A1) (CBMAB-A3314-YC)

-
Mouse Anti-CIITA Recombinant Antibody (CBLC160-LY) (CBMAB-C10987-LY)

-
Mouse Anti-ADIPOR1 Recombinant Antibody (V2-179982) (CBMAB-A1368-YC)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






