MEPE Antibodies
Background
The MEPE gene encodes a secretory bone matrix protein, which is mainly expressed in bone tissue and is particularly active in osteocytes and osteoblasts. This protein is involved in regulating the bone mineralization process, maintaining bone mineral balance by inhibiting the excessive deposition of hydroxyapatite crystals, and simultaneously influencing the reabsorption of phosphate by renal tubules. Research has found that the abnormal expression of the MEPE gene is closely related to metabolic bone diseases such as hypophosphatemic rickets and neoplastic osteomalacia. The acidic fragments released by its cleavage can directly act on the mineralization regulatory pathways of the kidneys and bones. This gene was first identified through localization cloning technology in 2000. The unveiling of its multi-dimensional functions has provided a key molecular basis for understanding the phosphate homeostasis mechanism of the bone-kidney axis. Related research continues to drive the development of diagnostic markers and targeted therapeutic strategies for metabolic bone diseases.
Structure of MEPE
The extracellular phosphorylated glycoprotein encoded by the MEPE gene is a secreted protein, and the molecular weight of its mature form is approximately 60-65 kDa. There is sequence conservation among different species, and their core functional regions are highly homologous among different mammals.
| Species | Human | Mouse | Rat |
| Molecular Weight (kDa) | About 60-65 | About 62 | About 63 |
| Primary Structural Differences | Contains ASARM inhibitory peptide, RGD motif | Highly conserved ASARM area, function is similar | The core zone sequence |
This protein is composed of approximately 525 amino acids, and its primary structure contains multiple characteristic functional domains. Among them, the acidic motif (ASARM motif) rich in aspartic acid and serine at the C-terminal is a key region for regulating mineralization and can directly inhibit the growth of hydroxyapatite crystals. In addition, the RGD sequence in the center of the protein mediates the interaction with integrins on the cell surface, thereby influencing cell adhesion and signal transduction. These structural features jointly endow the MEPE protein with the core function of precisely regulating phosphate homeostasis and bone matrix mineralization in the bone-kidney axis.
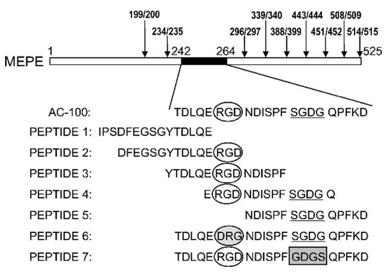 Fig. 1 Primary structure of human MEPE.1
Fig. 1 Primary structure of human MEPE.1
Key structural properties of MEPE:
- Composed of multiple module domains
- Functional acidic motif
- RGD integrin binding site
- Phosphorylation and glycosylation modification
Functions of MEPE
The core function of the MEPE gene-encoded protein is to regulate the mineralization homeostasis of bones and kidneys, but its mechanism of action is diverse and it participates in various pathophysiological processes.
| Function | Description |
| Inhibition of bone mineralization | By directly binding to hydroxyapatite crystals through its C-terminal ASARM peptide segment, it inhibits their excessive growth and prevents abnormal bone hardening. |
| Regulation of phosphate metabolism | In the kidneys, the fragments released by its lysis can inhibit the reabsorption of phosphate by renal tubules and reduce blood phosphorus levels. |
| Associated with neoplastic osteomalacia | Abnormally high expression in some mesenchymal tumors leads to renal phosphate loss and osteomalacia. |
| Osteocyte signal regulation | RGD motif interacts with osteoblast integrin to affect cell differentiation and bone remodeling process. |
| Dentin formation is involved | Express in the process of tooth development, participate in dentin matrix mineralization of fine-tuning. |
The functional regulation of MEPE protein is highly dependent on its proteolytic cleavage process: the full-length protein mainly serves as the matrix component, while the small molecule active peptides such as ASARM released by it are the key effector molecules that exercise systemic phosphate regulation and local mineralization inhibition. This dual mode of action of "precursors - active fragments" enables it to play the role of a precise regulator in the homeostasis of the bone-kidney axis.
Applications of MEPE and MEPE Antibody in Literature
- Surakka, Ida, et al. "MEPE loss-of-function variant associates with decreased bone mineral density and increased fracture risk." Nature communications 11.1 (2020): 4093. https://doi.org/10.1038/s41467-020-17315-0
This study found through sequencing that loss-of-function mutations in the MEPE gene could significantly reduce bone mineral density and increase the risk of osteoporosis and fractures. The results were verified in over 600,000 European samples. It is suggested that this rare functional variation may be more valuable for clinical risk assessment than polygenic risk scores.
- Christensen, Brian, et al. "FAM20C Mediated Phosphorylation of MEPE and Its Acidic Serine and Aspartate Rich Motif." Journal of Bone and Mineral Research Plus 4.8 (2020): e10378. https://doi.org/10.1002/jbm4.10378
Research has found that human MEPE proteins, after being phosphorylated by FAM20C, have as many as 31 phosphorylation sites, and all serines in the key ASARM motif are modified. This provides an important basis for understanding the high phosphorylation-dependent function of this protein in regulating bone mineralization and phosphate homeostasis.
- Staines, K. A., et al. "MEPE is a novel regulator of growth plate cartilage mineralization." Bone 51.3 (2012): 418-430. https://doi.org/10.1016/j.bone.2012.06.022
Research has found that MEPE protein and the phosphorylated ASARM peptide produced by its hydrolysis can specifically inhibit matrix mineralization in the hypertrophic cartilage region of growth plates without affecting cell differentiation. This effect has been confirmed in both cell and organ culture models, indicating that MEPE is a key regulatory factor for cartilage mineralization.
- Salmon, Benjamin, et al. "MEPE-derived ASARM peptide inhibits odontogenic differentiation of dental pulp stem cells and impairs mineralization in tooth models of X-linked hypophosphatemia." PloS one 8.2 (2013): e56749. https://doi.org/10.1371/journal.pone.0056749
Studies have confirmed that MEPE-derived phosphorylated ASARM peptides are the key factor leading to dentin mineralization defects in X-linked hypophosphatemic rickets (XLH) teeth. It can directly inhibit odontogenic differentiation and matrix mineralization, and up-regulate MEPE expression, forming a vicious cycle, making it a potential therapeutic target for XLH.
- Kulkarni, Rishikesh N., et al. "Inhibition of osteoclastogenesis by mechanically loaded osteocytes: involvement of MEPE." Calcified tissue international 87.5 (2010): 461-468. https://doi.org/10.1007/s00223-010-9407-7
Research has found that mechanical load can up-regulate the expression of the MEPE gene in bone cells without affecting its lyase PHEX, which may promote the expression of osteoprotegerin (OPG). This leads to the inhibition of osteoclast generation supported by bone cells, suggesting that MEPE is a key factor regulating bone resorption in the osteomechanical response.
Creative Biolabs: MEPE Antibodies for Research
Creative Biolabs specializes in the production of high-quality MEPE antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom MEPE Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our MEPE antibodies, custom preparations, or technical support, contact us at email.
Reference
- Hayashibara, Tetsuyuki, et al. "A synthetic peptide fragment of human MEPE stimulates new bone formation in vitro and in vivo." Journal of Bone and Mineral Research 19.3 (2004): 455-462. https://doi.org/10.1359/JBMR.0301263
Anti-MEPE antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ALDOA Recombinant Antibody (A2) (CBMAB-A2316-YC)

-
Mouse Anti-AFM Recombinant Antibody (V2-634159) (CBMAB-AP185LY)

-
Mouse Anti-AAV8 Recombinant Antibody (V2-634028) (CBMAB-AP022LY)

-
Mouse Anti-BCL2L1 Recombinant Antibody (H5) (CBMAB-1025CQ)

-
Mouse Anti-ADAM12 Recombinant Antibody (V2-179752) (CBMAB-A1114-YC)

-
Mouse Anti-FN1 Monoclonal Antibody (D6) (CBMAB-1240CQ)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Mouse Anti-C5b-9 Recombinant Antibody (aE11) (CBMAB-AO138LY)

-
Rat Anti-(1-5)-α-L-Arabinan Recombinant Antibody (V2-501861) (CBMAB-XB0003-YC)

-
Mouse Anti-AMH Recombinant Antibody (5/6) (CBMAB-A2527-YC)

-
Mouse Anti-C4B Recombinant Antibody (CBYY-C2996) (CBMAB-C4439-YY)

-
Mouse Anti-AKT1 (Phosphorylated S473) Recombinant Antibody (V2-505430) (PTM-CBMAB-0067LY)

-
Mouse Anti-C5B-9 Recombinant Antibody (CBFYA-0216) (CBMAB-X0304-FY)

-
Mouse Anti-AGO2 Recombinant Antibody (V2-634169) (CBMAB-AP203LY)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503417) (CBMAB-V208-1369-FY)

-
Mouse Anti-CECR2 Recombinant Antibody (CBWJC-2465) (CBMAB-C3533WJ)

-
Mouse Anti-AQP2 Recombinant Antibody (E-2) (CBMAB-A3358-YC)

-
Mouse Anti-CD24 Recombinant Antibody (HIS50) (CBMAB-C10123-LY)

-
Mouse Anti-CCL18 Recombinant Antibody (64507) (CBMAB-C7910-LY)

-
Mouse Anti-BHMT Recombinant Antibody (CBYY-0547) (CBMAB-0550-YY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






