MPC1 Antibodies
Background
The MPC1 gene encodes mitochondrial pyruvate vector protein 1, and its product is mainly distributed in the inner mitochondrial membrane of vertebrate cells. This protein specifically mediates the transmembrane transport of pyruvate from the cytoplasm to the mitochondrial matrix, thereby providing a core substrate for the tricarboxylic acid cycle to drive aerobic energy metabolism. The proliferation of tumor cells highly depends on the MPC1-mediated pyruvate metabolic pathway to maintain their abnormal energy requirements. This gene was jointly identified by multiple research teams through bioinformatics and functional experiments in 2012, becoming the first mitochondrial pyruvate transporter confirmed at the molecular level. The resolution of its four-helix bundle transmembrane domain provides new targets for the treatment of metabolic diseases and cancer, promoting systematic research on cellular metabolic transport mechanisms and energy homeostasis regulation.
Structure of MPC1
Mitochondrial pyruvate carrier protein 1 encoded by the MPC1 gene is a transmembrane protein with a molecular weight of approximately 15 kDa. Its molecular weight remains relatively stable among different species, mainly due to the highly conserved core transmembrane domain.
| Species | Human | Mouse | Rat | Yeast |
| Molecular Weight (kDa) | About 15 | About 15 | About 15 | About 14 |
| Primary Structural Differences | Contains bimodal mitochondrial targeting sequences | High sequence homology | Highly conserved across the membrane area | Homolog Mpc1, which is functionally similar |
This protein is composed of approximately 120 amino acids, and its primary structure forms four highly hydrophobic transmembrane α -helices, which constitute the transport channels for pyruvate. The secondary structure is almost entirely composed of these α -helices, which assemble into homologous or heterodimers in the inner mitochondrial membrane, forming functional pores. The key aspartic acid residues are located in the transmembrane region and are crucial for substrate recognition and the transport activity of proton coupling.
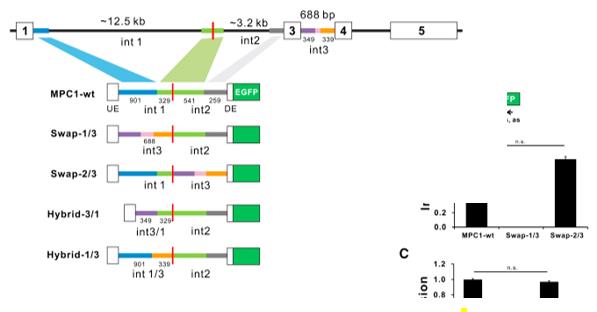 Fig. 1 Schematic of MPC1 minigene constructs.1
Fig. 1 Schematic of MPC1 minigene constructs.1
Key structural properties of MPC1:
- Four-helix transmembrane bundle structure
- Hydrophobic pores are used for pyruvate transport
- Key aspartic acid residues achieve proton coupled transport
Functions of MPC1
The core function of the protein encoded by the MPC1 gene is to regulate cellular energy metabolism. However, it also plays a role in a variety of pathophysiological processes, including tumor metabolic reprogramming and apoptosis regulation.
| Function | Description |
| Pyruvate transport | As a specific carrier on the inner mitochondrial membrane, it transports pyruvate produced by glycolysis from the cytoplasm to the mitochondrial matrix, providing a substrate for the tricarboxylic acid cycle. |
| Energy metabolism hub | The transfer activity direct coupling glycolysis and oxidative phosphorylation, is to determine cells use glucose for aerobic or anaerobic glycolysis of critical control points. |
| Metabolic reprogramming regulation | In many cancer cells, the expression is down-regulated or the function is impaired, leading to the "Warburg effect", that is, cells tend to undergo glycolysis even under aerobic conditions to support rapid proliferation. |
| Cell fate influence | By regulating mitochondrial pyruvate flow, it affects reactive oxygen species production, acetyl-CoA levels and apoptotic signals, and participates in determining cell survival or death. |
| Association with metabolic diseases | Its functional defects are closely related to metabolic diseases such as type 2 diabetes and non-alcoholic fatty liver disease, affecting the homeostasis of blood glucose and lipid metabolism throughout the body. |
Unlike myoglobin, which reversibly binds oxygen, MPC1-mediated pyruvate transport is an active and cooperative transport process dependent on membrane potential. Its high specificity for substrates determines its "gateway" status in the metabolic network rather than a simple reservoir.
Applications of MPC1 and MPC1 Antibody in Literature
- Zou, Hongbo, et al. "MPC1 deficiency accelerates lung adenocarcinoma progression through the STAT3 pathway." Cell death & disease 10.3 (2019): 148. https://doi.org/10.1038/s41419-019-1324-8
Studies have found that MPC1 is lowly expressed in lung adenocarcinoma and predicts a better prognosis. Research has found that MPC1 inhibits the characteristics, invasion and migration of tumor stem cells by binding to mitochondrial STAT3 and reducing cytoplasmic STAT3 and its phosphorylation. This axis may become a new target for the treatment of lung adenocarcinoma.
- Koh, Eunjin, Daye Shin, and Kyung-Sup Kim. "Exon definitive regions for MPC1 microexon splicing and its usage for splicing modulation." Molecular Therapy Nucleic Acids 31 (2023): 398-410. https://doi.org/10.1016/j.omtn.2023.01.010
Research has found that the composition of a 4bp microexon in the MPC1 gene is retained in mRNA. The "core exon island" fragment in its adjacent introns can change the splicing pattern of the target gene according to the insertion position, which provides a new strategy for splice-mediated gene correction.
- Li, Xiaoli, et al. "MPC1 and MPC2 expressions are associated with favorable clinical outcomes in prostate cancer." BMC cancer 16.1 (2016): 894. https://doi.org/10.1186/s12885-016-2941-6
This study reveals that the expression of MPC1 and MPC2 in prostate cancer is positively correlated, and both positive expressions are significantly associated with a longer overall survival period of patients. Multivariate analysis indicated that both were independent favorable prognostic factors.
- Li, Mingkang, et al. "LONP1 facilitates pulmonary artery smooth muscle cell glycolytic reprogramming by degrading MPC1 in pulmonary hypertension." Clinical Science 139.10 (2025): 479-501. https://doi.org/10.1042/CS20255922
Research has found that LONP1 is highly expressed in pulmonary arterial hypertension (PH) and drives glycolytic reprogramming, proliferation and migration of pulmonary artery smooth muscle cells by degrading the mitochondrial pyruvate vector MPC1. Targeted inhibition of LONP1 can alleviate PH, suggesting its potential as a new therapeutic target.
- Wang, Leiming, et al. "MPC1, a key gene in cancer metabolism, is regulated by COUPTFII in human prostate cancer." Oncotarget 7.12 (2016): 14673. https://doi.org/10.18632/oncotarget.7405
The transcription factor COUP-TFII has been found to induce a glycolytic metabolic shift in prostate cancer cells when MPC1 expression is inhibited, thereby promoting tumor growth and metastasis. This reveals a potential new therapeutic direction targeting COUP-TFII to regulate metabolism.
Creative Biolabs: MPC1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality MPC1 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom MPC1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our MPC1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Koh, Eunjin, Daye Shin, and Kyung-Sup Kim. "Exon definitive regions for MPC1 microexon splicing and its usage for splicing modulation." Molecular Therapy Nucleic Acids 31 (2023): 398-410. https://doi.org/10.1016/j.omtn.2023.01.010
Anti-MPC1 antibodies
Products List
 Loading...
Loading...
Hot products 
-
Mouse Anti-COL1A2 Recombinant Antibody (CF108) (V2LY-1206-LY626)

-
Mouse Anti-BRD3 Recombinant Antibody (CBYY-0801) (CBMAB-0804-YY)

-
Mouse Anti-ARID3A Antibody (A4) (CBMAB-0128-YC)

-
Mouse Anti-CSPG4 Recombinant Antibody (CBFYM-1050) (CBMAB-M1203-FY)

-
Mouse Anti-ESR1 Recombinant Antibody (Y31) (CBMAB-1208-YC)

-
Mouse Anti-Acetyl-α-Tubulin (Lys40) Recombinant Antibody (V2-623485) (CBMAB-CP2897-LY)

-
Mouse Anti-ACKR3 Recombinant Antibody (V2-261265) (CBMAB-C1023-LY)

-
Rabbit Anti-Acetyl-Histone H3 (Lys36) Recombinant Antibody (V2-623395) (CBMAB-CP0994-LY)

-
Mouse Anti-ACO2 Recombinant Antibody (V2-179329) (CBMAB-A0627-YC)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Mouse Anti-CORO1A Recombinant Antibody (4G10) (V2LY-1206-LY806)

-
Mouse Anti-ACVR1C Recombinant Antibody (V2-179685) (CBMAB-A1041-YC)

-
Mouse Anti-DDC Recombinant Antibody (8E8) (CBMAB-0992-YC)

-
Mouse Anti-ENO2 Recombinant Antibody (H14) (CBMAB-E1341-FY)

-
Mouse Anti-CD8 Recombinant Antibody (C1083) (CBMAB-C1083-LY)

-
Mouse Anti-CEMIP Recombinant Antibody (3C12) (CBMAB-K0296-LY)

-
Mouse Anti-B2M Recombinant Antibody (CBYY-0050) (CBMAB-0050-YY)

-
Mouse Anti-AFDN Recombinant Antibody (V2-58751) (CBMAB-L0408-YJ)

-
Mouse Anti-BHMT Recombinant Antibody (CBYY-0547) (CBMAB-0550-YY)

-
Mouse Anti-ASTN1 Recombinant Antibody (H-9) (CBMAB-1154-CN)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






