OAS3 Antibodies
Background
The OAS3 gene encodes a double-stranded RNA sensor protein that is induced by interferons and is mainly present in various cytoplasmic compartments of human cells. This protein, as a member of the 2'-5' oligoadenylate synthetase family, can detect viral double-stranded RNA and catalyze the synthesis of 2'-5' linked oligoadenylates, thereby activating the potential ribonuclease L, leading to the degradation of viral RNA and inhibition of viral replication. Studies have found that OAS3 plays a crucial role in the innate immune response, especially in resisting infections by viruses such as dengue virus and West Nile virus. Its genetic polymorphism is associated with the susceptibility to various diseases, including the progression of chronic hepatitis C infection and the clinical course of severe acute respiratory syndrome. Through the study of the relationship between the structure and function of the OAS3 protein, scientists have deepened their understanding of the antiviral mechanism of the interferon system and provided new molecular targets for the development of broad-spectrum antiviral treatment strategies.
Structure of OAS3
The protein encoded by the OAS3 gene has a molecular weight of approximately 134 kDa and shows some variations among different species.
| Species | Human | Mouse | Rat | Macaque monkey | Bovine |
| Molecular Weight (kDa) | 134 | 133 | 133 | 134 | 135 |
| Primary Structural Differences | Contains three classical domains | The second domain is functionally absent | Highly homologous to rats | Highly similar to humans | There are specific variations |
The OAS3 protein consists of 1087 amino acids and is composed of three tandem 2′-5′ oligoadenylate synthetase domains. This protein is expressed in response to interferon induction and specifically recognizes viral double-stranded RNA through its C-terminal domain, catalyzing the polymerization of ATP to generate 2′-5′ linked oligoadenylates. These products activate latent ribonuclease L as second messengers, leading to the degradation of viral RNA. Among the three domains of OAS3, only the first and the third have complete catalytic active centers, while the second domain, although lacking catalytic function, still participates in dsRNA binding. A highly conserved D-D/E-D/F motif exists in the C-terminal domain of the protein, which is crucial for catalytic activity.
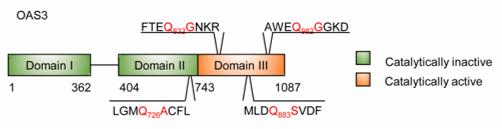 Fig. 1 Amino acid sequences of four possible cleavage sites in OAS3.1
Fig. 1 Amino acid sequences of four possible cleavage sites in OAS3.1
Key structural properties of OAS3:
- Three consecutive 2'-5' oligoadenylate synthetase domains
- Each domain contains a conserved catalytic active center
- The C-terminal domain is responsible for specific recognition of viral double-stranded RNA
- The second domain, although lacking catalytic function, retains RNA binding ability
- The D-D/E-D/F motif is crucial for catalytic activity
Functions of OAS3
The main function of the OAS3 gene is to act as a sensor for viral double-stranded RNA, triggering the innate immune response. At the same time, it also participates in regulating various antiviral signaling pathways and inflammatory responses.
| Function | Description |
| Virus RNA Recognition | OAS3 specifically binds to viral double-stranded RNA through its C-terminal domain, acting as a pattern recognition receptor to initiate antiviral immunity. |
| Catalyzes the synthesis of oligoadenylates | After activation, it catalyzes the production of 2'-5' linked oligoadenylates from ATP, serving as a second messenger to activate downstream effector molecules. |
| RNase L Activation | The synthesized oligoadenylate binds to and activates the latent ribonuclease L, leading to the degradation of viral and cellular RNA and inhibiting viral replication. |
| Interferon signal enhancement | As an interferon-stimulated gene, its expression is induced by interferons and simultaneously forms a positive feedback regulation by amplifying the interferon signal. |
| Cell Apoptosis Regulation | During viral infection, apoptosis of infected cells is induced through the RNase L pathway to limit the spread of the virus. |
OAS3 exhibits length-dependent recognition of double-stranded RNA, preferentially binding to longer double-stranded RNA, which is associated with the cooperative action of multiple structural domains of the protein.
Applications of OAS3 and OAS3 Antibody in Literature
1. Zhou, Xiaolei, et al. "EV71 3C protease cleaves host anti-viral factor OAS3 and enhances virus replication." Virologica Sinica 37.3 (2022): 418-426. https://doi.org/10.1016/j.virs.2022.04.013
The article indicates that OAS3 can effectively inhibit the replication of various enteroviruses, while EV71 evades the antiviral effect by cutting OAS3 through its 3C protease. The 3C protease of CA16 is unable to cut OAS3 due to a key amino acid variation, resulting in weakened replication.
2. Wang, Xinyue, et al. "Rescue RM/CS-AKI by blocking strategy with one-dose anti-myoglobin RabMAb." Nature Communications 16.1 (2025): 1044. https://doi.org/10.7150/ijbs.96089
The article indicates that in acute lung injury caused by sepsis, the downregulation of E3 ubiquitin ligase TRIM21 leads to the accumulation of OAS3 protein, which subsequently promotes apoptosis of lung epithelial cells through RNase L. OAS3 may represent a new therapeutic target.
3. Li, Xin-yu, et al. "OAS3 is a co-immune biomarker associated with tumour microenvironment, disease staging, prognosis, and treatment response in multiple cancer types." Frontiers in Cell and Developmental Biology 10 (2022): 815480. https://doi.org/10.3389/fcell.2022.815480
The article indicates that OAS3 is abnormally highly expressed in various tumors and is associated with tumor stage, metastasis, and poor prognosis. Its level is positively correlated with the infiltration of immunosuppressive cells, which affects the efficacy of chemotherapy and may serve as a potential prognostic marker.
4. Zhang, Yujie, and Chaoran Yu. "Prognostic characterization of OAS1/OAS2/OAS3/OASL in breast cancer." BMC cancer 20.1 (2020): 575. https://doi.org/10.1186/s12885-020-07034-6
The article indicates that high expression of OAS1 and OAS3 predicts poor prognosis for breast cancer, while OAS2 has the opposite effect. The expression of this family is associated with neutrophil infiltration, and its methylation sites also have prognostic value.
5. Abdelhafez, Mohammad, et al. "Association of IFNAR2 rs2236757 and OAS3 rs10735079 Polymorphisms with Susceptibility to COVID‐19 Infection and Severity in Palestine." Interdisciplinary Perspectives on Infectious Diseases 2023.1 (2023): 9551163. https://doi.org/10.1155/2023/9551163
The article indicates that the rs10735079A variant of the OAS3 gene can reduce the risk of muscle soreness and sore throat in COVID-19 patients, while the IFNAR2 gene variant is associated with severe cases and breathing difficulties.
Creative Biolabs: OAS3 Antibodies for Research
Creative Biolabs specializes in the production of high-quality OAS3 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom OAS3 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our OAS3 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Zhou, Xiaolei, et al. "EV71 3C protease cleaves host anti-viral factor OAS3 and enhances virus replication." Virologica Sinica 37.3 (2022): 418-426. Distributed under Open Access license CC BY 4.0, cropped from the original figure. https://doi.org/10.1016/j.virs.2022.04.013
Anti-OAS3 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-CDKL5 Recombinant Antibody (CBFYC-1629) (CBMAB-C1689-FY)

-
Mouse Anti-FeLV g27 Recombinant Antibody (1) (CBMAB-V208-1714-FY)

-
Mouse Anti-ASH1L Monoclonal Antibody (ASH5H03) (CBMAB-1372-YC)

-
Mouse Anti-ADIPOR2 Recombinant Antibody (V2-179983) (CBMAB-A1369-YC)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-0790) (CBMAB-0793-YY)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503417) (CBMAB-V208-1369-FY)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

-
Mouse Anti-ALB Recombinant Antibody (V2-55272) (CBMAB-H0819-FY)

-
Mouse Anti-AHCYL1 Recombinant Antibody (V2-180270) (CBMAB-A1703-YC)

-
Mouse Anti-ACTG1 Recombinant Antibody (V2-179597) (CBMAB-A0916-YC)

-
Mouse Anti-BZLF1 Recombinant Antibody (BZ.1) (CBMAB-AP705LY)

-
Rabbit Anti-B2M Recombinant Antibody (CBYY-0059) (CBMAB-0059-YY)

-
Mouse Anti-AOC3 Recombinant Antibody (CBYY-0014) (CBMAB-0014-YY)

-
Human Anti-SARS-CoV-2 Spike Recombinant Antibody (CBC05) (CBMAB-CR005LY)

-
Mouse Anti-CD24 Recombinant Antibody (SN3) (CBMAB-C1037-CQ)

-
Mouse Anti-BPGM Recombinant Antibody (CBYY-1806) (CBMAB-2155-YY)

-
Mouse Anti-CFL1 Recombinant Antibody (CBFYC-1771) (CBMAB-C1833-FY)

-
Mouse Anti-ADV Recombinant Antibody (V2-503423) (CBMAB-V208-1364-FY)

-
Mouse Anti-BCL2L1 Recombinant Antibody (H5) (CBMAB-1025CQ)

-
Mouse Anti-CD83 Recombinant Antibody (HB15) (CBMAB-C1765-CQ)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






