PTPRO Antibodies
Background
PTPRO is a transmembrane receptor-like protein tyrosine phosphatase, mainly expressed in the kidneys, immune cells and the nervous system. This gene participates in regulating key life activities such as cell growth, differentiation and apoptosis by specifically catalyzing the dephosphorylation process of substrate proteins. During the development of the kidneys, PTPRO can maintain the integrity of the podocyte cytoskeleton structure. The loss of its function can lead to proteinuria and the destruction of the glomerular filtration barrier. In 1994, researchers first identified the PTPRO gene through cloning technology. Its unique extracellular fibronectin type III repeat domain, single transmembrane region and intracellular phosphatase domain provided an important model for studying the regulatory mechanism of tyrosine phosphorylation. The regulatory role of this gene in tumor suppression, autoimmune diseases and neurodegenerative disorders has continuously attracted academic attention. Its complex signaling pathway network has become an important example in the study of cell signal transduction.
Structure of PTPRO
PTPRO is a transmembrane protein tyrosine phosphatase with a molecular weight of approximately 200-220 kDa. The difference in molecular weight mainly results from the variations in transcript splicing and glycosylation modifications.
| Species | Human | Mouse | Rat |
| Molecular Weight (kDa) | 214.5 (Isomer 1) | 145.8 (Isomer 3) | 215.2 |
| Primary Structural Differences | Extracellular region containing fibronectin III repeat sequences | The structure of extracellular domain is shorter | Highly homologous to the human PTPRO structure |
This protein is composed of 1,443 amino acid residues, and its intracellular region contains a highly conserved tyrosine phosphatase domain, which is responsible for catalyzing the dephosphorylation process of the substrate. The extracellular region of PTPRO is composed of multiple fibronectin type III repeat modules, which form an extended structure in β -folded lamellae and participate in the interactions between cells and between cells and the matrix. Its single transmembrane helix conducts extracellular signals into the intracellular, while the intracellular phosphatase domain precisely regulates signal transduction by specifically binding to the substrate through a conserved catalytic motif (HCXXGXXR).
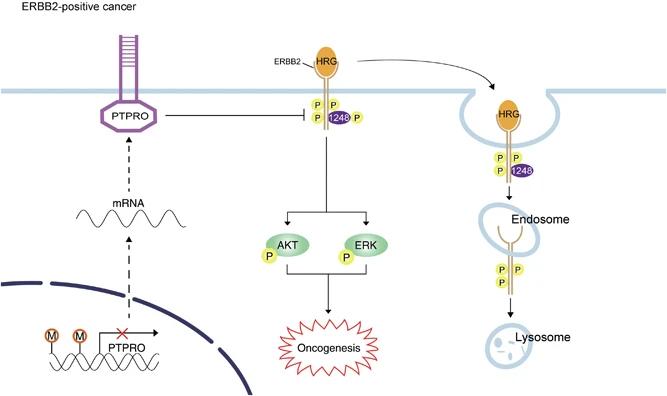 Fig. 1 Schematic summary of the role of PTPRO in ERBB2-driven breast carcinogenesis.1
Fig. 1 Schematic summary of the role of PTPRO in ERBB2-driven breast carcinogenesis.1
Key structural properties of PTPRO:
- Extracellular fibronectin III repetitive structure domain component ligand recognition interface
- A single transmembrane helix connects extracellular and intracellular domains
- Intracellular highly conserved phosphatase catalytic domain (PTP domain)
Functions of PTPRO
As a receptor tyrosine phosphatase, the core function of PTPRO is to regulate intracellular signal transduction through dephosphorylation modification. This protein plays multiple regulatory roles in different tissues and physiological and pathological processes.
| Function | Description |
| Cell proliferation inhibition | By dephosphorylating multiple growth factor receptors and downstream signaling proteins, it negatively regulates the cell cycle process and mitotic signaling. |
| Immune regulation | Regulatory T cells in lymphocyte receptors signal strength, affect the immune synapse formation and the activation of the immune response threshold. |
| Renal filtration barrier is maintained | In glomerular podocytes, adaptor proteins such as phosphorylated nephrin are passed through to maintain the integrity of the pore membrane structure and filtration function. |
| Tumor suppression | In various cancer cells, it inhibits the abnormal proliferation, invasion and metastasis of tumor cells by dephosphorylating oncogenic signaling proteins. |
| Nervous system development | By regulating neurotrophic factor signaling pathways such as Trk receptors, it affects neuronal differentiation, axon orientation and synaptic plasticity. |
PTPRO functions as a "molecular switch" for various tyrosine phosphorylation signaling events through its highly specific phosphatase activity. Unlike the broad action characteristics of some kinases, PTPRO exhibits strict substrate selectivity and pathway specificity. This precise regulation makes it play a key role in maintaining tissue homeostasis and inhibiting pathological processes.
Applications of PTPRO and PTPRO Antibody in Literature
1. Yao, Zhimeng, et al. "Age-related decline in hippocampal tyrosine phosphatase PTPRO is a mechanistic factor in chemotherapy-related cognitive impairment." JCI insight 8.14 (2023): e166306. https://doi.org/10.1172/jci.insight.166306
This article reveals that the protein tyrosine phosphatase PTPRO in the hippocampal region significantly decreases with aging, and its deficiency can exacerbate chemotherapy-related cognitive impairment. Studies have confirmed that PTPRO exerts neuroprotective effects by regulating the phosphorylation of SRC/EPHA4, while berberine can upregulate PTPRO by inhibiting miR-25-3p and improve cognitive impairment.
2. Hou, Xuben, Jintong Du, and Hao Fang. "PTPRO is a therapeutic target and correlated with immune infiltrates in pancreatic cancer." Journal of Cancer 12.24 (2021): 7445. https://doi.org/10.7150/jca.64661
This study, through bioinformatics analysis, found that the protein tyrosine phosphatase PTPRO plays a potential carcinogenic role in various tumors including pancreatic cancer. Experiments have confirmed that inhibiting PTPRO can significantly curb the proliferation of cancer cells and induce their apoptosis. In addition, the expression of PTPRO is associated with immune cell infiltration. The results show that PTPRO is a potential therapeutic target for pancreatic cancer.
3. Huan, Zhirong, et al. "PTPRO knockdown protects against inflammation in hemorrhage shock-induced lung injury involving the NF-κB signaling pathway." Respiratory Research 23.1 (2022): 195. https://doi.org/10.1186/s12931-022-02118-2
This study reveals that in the hemorrhagic shock model, the expression of lung tissue protein tyrosine phosphatase PTPRO significantly increases. Knockdown of PTPRO can enhance the integrity of lung epithelium by up-regulating tight junction proteins and inhibiting the activation of the NF-κB pathway, thereby reducing inflammatory cell infiltration and cytokine release, and ultimately alleviating lung injury.
4. Dong, Hongmei, et al. "PTPRO-related CD8+ T-cell signatures predict prognosis and immunotherapy response in patients with breast cancer." Frontiers in immunology 13 (2022): 947841. https://doi.org/10.3389/fimmu.2022.947841
This study found that in breast cancer, the high expression of protein tyrosine phosphatase PTPRO is closely related to the elevated infiltration levels of immune cells such as CD8+ T cells. The CD8+ T cell characteristic model constructed based on PTPRO can not only evaluate the prognosis of patients, but also effectively predict their therapeutic response to immune checkpoint inhibitors, providing a new basis for cancer immunotherapy decision-making.
5. Xiong, Xiao, et al. "PTPRO represses breast cancer lung metastasis by inhibiting the JAK2-YAP axis." Scientific Reports 15.1 (2025): 7065. https://doi.org/10.1038/s41598-025-91341-0
This study reveals that the protein tyrosine phosphatase PTPRO plays a key role in inhibiting lung metastasis of breast cancer. PTPRO inhibits the activation of the JAK2-YAP pathway by dephosphorylating JAK2 and YAP, ultimately reducing the metastatic ability of cancer cells. The research results indicate that enhancing the function of PTPRO or targeting the pathways it mediates is a potential strategy for inhibiting breast cancer metastasis.
Creative Biolabs: PTPRO Antibodies for Research
Creative Biolabs specializes in the production of high-quality PTPRO antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom PTPRO Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our PTPRO antibodies, custom preparations, or technical support, contact us at email.
Reference
- Dong, H., et al. "PTPRO represses ERBB2-driven breast oncogenesis by dephosphorylation and endosomal internalization of ERBB2." Oncogene 36.3 (2017): 410-422. https://doi.org/10.1038/onc.2016.213
Anti-PTPRO antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-AAV9 Recombinant Antibody (V2-634029) (CBMAB-AP023LY)

-
Mouse Anti-CD33 Recombinant Antibody (P67.6) (CBMAB-C10189-LY)

-
Mouse Anti-ARSA Recombinant Antibody (CBYC-A799) (CBMAB-A3679-YC)

-
Mouse Anti-EGR1 Recombinant Antibody (CBWJZ-100) (CBMAB-Z0289-WJ)

-
Mouse Anti-BIRC3 Recombinant Antibody (315304) (CBMAB-1214-CN)

-
Mouse Anti-BAX Recombinant Antibody (CBYY-0216) (CBMAB-0217-YY)

-
Mouse Anti-BHMT Recombinant Antibody (CBYY-0547) (CBMAB-0550-YY)

-
Mouse Anti-ATG5 Recombinant Antibody (9H197) (CBMAB-A3945-YC)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-0790) (CBMAB-0793-YY)

-
Mouse Anti-ACKR3 Recombinant Antibody (V2-261265) (CBMAB-C1023-LY)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Mouse Anti-C5B-9 Recombinant Antibody (CBFYA-0216) (CBMAB-X0304-FY)

-
Mouse Anti-BIRC3 Recombinant Antibody (16E63) (CBMAB-C3367-LY)

-
Mouse Anti-BAD (Phospho-Ser136) Recombinant Antibody (CBYY-0138) (CBMAB-0139-YY)

-
Rabbit Anti-B2M Recombinant Antibody (CBYY-0059) (CBMAB-0059-YY)

-
Mouse Anti-CTNND1 Recombinant Antibody (CBFYC-2414) (CBMAB-C2487-FY)

-
Mouse Anti-AMACR Recombinant Antibody (CB34A) (CBMAB-CA034LY)

-
Mouse Anti-ANXA7 Recombinant Antibody (A-1) (CBMAB-A2941-YC)

-
Mouse Anti-COL12A1 Recombinant Antibody (CBYY-C3117) (CBMAB-C4560-YY)

-
Mouse Anti-ACE2 Recombinant Antibody (V2-179293) (CBMAB-A0566-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






