CDC42 Antibodies
Background
CDC42 is a tiny GTPase protein belonging to the Rho family and is widely present in eukaryotic cells. This protein regulates key life activities such as cytoskeletal recombination, cell polarity and cell division through the transition of GTP/GDP binding state, thereby maintaining the stability of cell morphology and function. During the development of neurons and the migration of immune cells, CDC42 directly guides the direction of cell movement by coordinating the dynamic assembly of actin. Its gene function was first systematically revealed through yeast mutant research in the 1990s. Subsequent studies have confirmed that this protein is one of the core hubs of the cell's signal transduction network. An in-depth analysis of the relationship between the structure and function of CDC41 not only reveals the mechanism of action of the GTPase molecular switch, but also provides an important molecular basis for pathological research such as tumor metastasis and neurodegenerative diseases.
Structure of CDC42
CDC42 is a small GTP-binding protein with a molecular weight of approximately 21 kDa. Its precise molecular weight varies slightly among different homologies, mainly due to the evolutionary divergence of the gene coding sequence. The following is a comparison of the typical characteristics of CDC42 protein in different species:
| Species | Human | Mouse | Yeast | Fruit fly | Nematode |
| Molecular Weight (kDa) | 21.4 | 21.3 | 21.6 | 21.2 | 21.5 |
| Primary Structural Differences | Containing 191 amino acids, C terminal with lipid modification sites | Homology with human sequences > 90% | Maintain polar growth regulatory function | Participate in the morphogenesis of epithelial cells | Functionally conserved during cell migration |
This protein is composed of five highly conserved domains forming its GTPase core. Among them, the conformations of the Switch I and Switch II regions undergo reversible changes with the GTP/GDP binding state, thereby determining its molecular switching activity. Glycine at position 12 and glutamine at position 61, as key catalytic residues, directly participate in the regulation of GTP hydrolysis. This mechanism is highly conserved in eukaryotes, from yeast to humans.
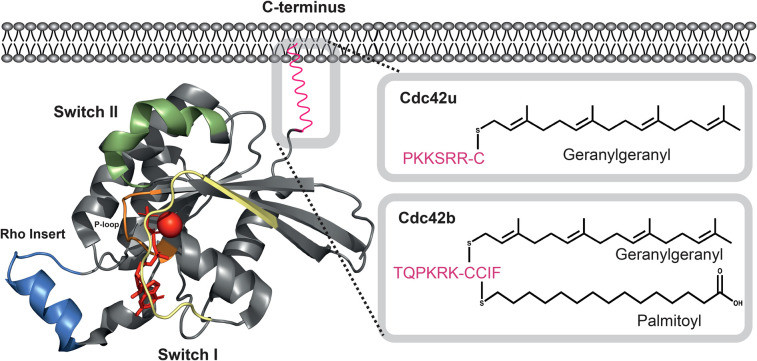 Fig. 1 The structure of Cdc42.1
Fig. 1 The structure of Cdc42.1
Key structural properties of CDC42:
- The classic GTPase folding conformation is adopted
- Has a highly conservative GTP/GDP combined domain
- Contains molecular Switch mechanism is the core of the Switch I/II area
Functions of CDC42
The core function of the CDC42 protein is to act as a molecular switch for intracellular signal transduction. However, it is also widely involved in a variety of key cellular physiological activities, including the establishment of cell polarity and the regulation of the cell cycle.
| Function | Description |
| Cytoskeletal recombination | By activating downstream effectors (such as WASP proteins), the polymerization and rearrangement of actin are directly initiated to control the morphological changes of cells. |
| Establishment of cell polarity | Locating specific protein complexes in asymmetric cells provides a directional basis for processes such as cell movement and division. |
| Cell cycle regulation | Through regulating the G1 / S phase transformation affect the process of cell division, its abnormal expression is closely related to tumorigenesis. |
| Vesicle transport orientation | Participate in the vesicle transport process from the Golgi apparatus to the cell membrane to ensure the accuracy of directional transport of substances. |
| Regulation of gene expression | As a signal hub, it conducts extracellular stimuli to the nucleus through pathways such as MAPK, influencing the transcriptional activity of specific genes. |
The activity of CDC42 is precisely controlled by the GTP/GDP binding cycle. The conformational difference between its GTP binding state and GDP binding state determines the efficient initiation and timely termination of this signaling pathway. This characteristic makes it the core regulator of cellular life activities.
Applications of CDC42 and CDC42 Antibody in Literature
1. Murphy, Natasha P., et al. "Molecular subversion of Cdc42 signalling in cancer." Biochemical Society Transactions 49.3 (2021): 1425-1442. https://doi.org/10.1042/BST20200557
The article indicates that Cdc42, as a small GTPase of the Rho family, is a key molecule regulating cell movement, polarity and cycle. Its role in cancer is clear, but the specific mechanism is still under exploration. Given the challenges of directly targeting Cdc42, targeting its regulatory factors and downstream signaling pathways will become a more feasible therapeutic strategy.
2. Tang, Xinyi, et al. "CDC42 deficiency leads to endometrial stromal cell senescence in recurrent implantation failure." Human Reproduction 39.12 (2024): 2768-2784. https://doi.org/10.1093/humrep/deae246
Research has found that in patients with repeated implantation failure (RIF), the down-regulation of CDC42 protein in endometrial stromal cells can induce cellular senescence and decidualization defects by activating the Wnt signaling pathway, thereby damaging uterine receptivity. The use of the Wnt inhibitor XAV-939 can reverse this defect.
3. Swaine, Thomas, and Matthias T. Dittmar. "CDC42 Use in viral cell entry processes by RNA viruses." Viruses 7.12 (2015): 6526-6536. https://doi.org/10.3390/v7122955
Research has found that the Rho family GTPase Cdc42 is a key factor regulating the dynamics of actin in cells. Existing studies have shown that Cdc42 is utilized in the cellular invasion process of various RNA viruses such as HIV-1 to overcome the cytoskeletal barrier, which provides an important research direction for in-depth exploration of the mechanism of viral invasion.
4. Murphy, Natasha P., Helen R. Mott, and Darerca Owen. "Progress in the therapeutic inhibition of Cdc42 signalling." Biochemical Society Transactions 49.3 (2021): 1443-1456. https://doi.org/10.1042/BST20210112
Research has found that Cdc42, as a key Rho GTP enzyme, affects tumor development and metastasis by regulating the cytoskeleton. Its overexpression is associated with a poor prognosis for various cancers. The current targeted therapy strategies focus on inhibiting its lipid modification, regulating the GEF/GDI interaction, and blocking downstream effector molecules, providing a new research and development direction for cancer treatment.
5. Jiang, Tao, et al. "CDC42—A promising immune-related target in glioma." Frontiers in Neuroscience 17 (2023): 1192766. https://doi.org/10.3389/fnins.2023.1192766
Studies have shown that the cell polarity regulator Cdc42 is significantly highly expressed in glioma, and its enrichment degree is closely related to the malignancy of the tumor, poor prognosis and the level of immune infiltration. Cdc42 may become a potential new target for glioma immunotherapy by regulating the tumor immune microenvironment.
Creative Biolabs: CDC42 Antibodies for Research
Creative Biolabs specializes in the production of high-quality CDC42 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom CDC42 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our CDC42 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Murphy, Natasha P., et al. "Molecular subversion of Cdc42 signalling in cancer." Biochemical Society Transactions 49.3 (2021): 1425-1442. https://doi.org/10.1042/BST20200557
Anti-CDC42 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ADGRL2 Recombinant Antibody (V2-58519) (CBMAB-L0166-YJ)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7697) (CBMAB-1869CQ)

-
Mouse Anti-FPR2 Recombinant Antibody (1D6) (CBMAB-F2628-CQ)

-
Mouse Anti-CD164 Recombinant Antibody (CBFYC-0077) (CBMAB-C0086-FY)

-
Mouse Anti-ARID3A Antibody (A4) (CBMAB-0128-YC)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503417) (CBMAB-V208-1369-FY)

-
Mouse Anti-CAPZB Recombinant Antibody (CBYY-C0944) (CBMAB-C2381-YY)

-
Mouse Anti-BRD3 Recombinant Antibody (CBYY-0801) (CBMAB-0804-YY)

-
Mouse Anti-ACE2 Recombinant Antibody (V2-179293) (CBMAB-A0566-YC)

-
Mouse Anti-CD24 Recombinant Antibody (SN3) (CBMAB-C1037-CQ)

-
Mouse Anti-CD247 Recombinant Antibody (6B10.2) (CBMAB-C1583-YY)

-
Mouse Anti-ADGRE2 Recombinant Antibody (V2-261270) (CBMAB-C0813-LY)

-
Mouse Anti-C4B Recombinant Antibody (CBYY-C2996) (CBMAB-C4439-YY)

-
Rabbit Anti-AKT3 Recombinant Antibody (V2-12567) (CBMAB-1057-CN)

-
Mouse Anti-C5B-9 Recombinant Antibody (CBFYA-0216) (CBMAB-X0304-FY)

-
Mouse Anti-BAX Recombinant Antibody (CBYY-0216) (CBMAB-0217-YY)

-
Mouse Anti-AMH Recombinant Antibody (5/6) (CBMAB-A2527-YC)

-
Mouse Anti-DES Monoclonal Antibody (440) (CBMAB-AP1857LY)

-
Mouse Anti-ADAM12 Recombinant Antibody (V2-179752) (CBMAB-A1114-YC)

-
Mouse Anti-CALR Recombinant Antibody (CBFYC-0763) (CBMAB-C0818-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






