ACACA Antibodies
Background
The ACACA gene encodes acetyl-CoA carboxylase α, which is an enzyme that plays a crucial regulatory role in the fatty acid biosynthesis pathway. It is mainly found in metabolically active organs such as the liver and adipose tissue. This enzyme catalyzes the conversion of acetyl-CoA to malonyl-CoA, controlling the initial step of fatty acid synthesis and thereby influencing the lipid metabolism balance within cells. In the research of metabolic diseases such as obesity and diabetes, the expression and activity of ACACA are often of great interest, as they are directly related to the regulatory mechanism of energy storage and utilization. Since its function has been gradually clarified, this gene has become an important molecular target in the field of metabolism research, providing a crucial theoretical basis for understanding lipid homeostasis, cell signal transduction, and the pathogenesis of related diseases.
Structure of ACACA
The acetyl-CoA carboxylase α encoded by the ACACA gene is a large multifunctional enzyme with a molecular weight of approximately 265 kDa. Its molecular weight remains relatively stable across different species and tissues, but there may be minor variations due to alternative splicing and post-translational modifications.
| Species | Human | Mouse | Rat | Chicken |
| Molecular Weight (kDa) | ~265 | ~265 | ~265 | ~265 |
| Primary Structural Differences | Containing the BC, BCCP and CT domains | Highly homologous structure, adjust the site | Similar phosphorylation regulatory sites | The core catalytic domain is conserved, but the regulatory mechanisms are different |
This protein is composed of over 2300 amino acids and folds into a compact multi-domain overall conformation. Its primary structure determines three key functional domains: the biotin carboxylase (BC) domain at the N-terminus, the biotin carboxyl carrier protein (BCCP) domain in the middle, and the carboxyl transferase (CT) domain at the C-terminus. The BCCP domain is covalently bound to a biotin cofactor through a flexible lysine linker, which is necessary for capturing carbon dioxide. The protein's secondary structure is composed of abundant α-helices and β-sheet structures, forming multiple active pockets. The key catalytic mechanism involves: the BC domain using ATP to carboxylate the biotin cofactor, followed by the BCCP domain transferring the activated carboxyl group to the CT domain, and finally the CT domain completing the carboxylation of acetyl coenzyme A, generating propionyl coenzyme A. The entire reaction process relies on the allosteric synergy and precise spatial conformational changes between the domains.
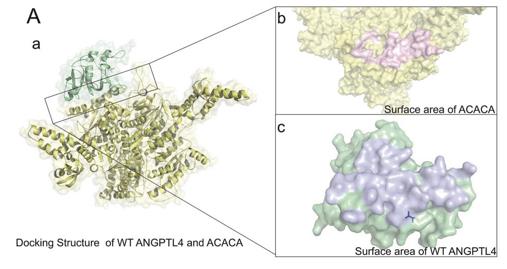 Fig. 1 Interaction docking structure between wild-type ANGPTL4 and ACACA.1
Fig. 1 Interaction docking structure between wild-type ANGPTL4 and ACACA.1
Key structural properties of ACACA:
- Modular multi-domain configuration
- The dynamic functional area connected by a flexible linker
- Carboxylation reaction centers dependent on ATP and biotin cofactors
Functions of ACACA
The main function of the ACACA gene (acetyl-CoA carboxylase alpha) is to catalyze the first step of the rate-limiting reaction in fatty acid biosynthesis. Additionally, it is deeply involved in various metabolic regulation and cellular signaling processes.
| Function | Description |
| Fatty Acid Synthesis | Catalyzes the conversion of acetyl coenzyme A to malonyl coenzyme A, which is a necessary substrate and a crucial regulatory step for the elongation of the fatty acid chain. |
| Metabolic Regulation Center | As the integration point for nutritional and hormonal signals (such as insulin, glucagon), it regulates the enzymatic activity through phosphorylation/dephosphorylation, thereby influencing the balance between energy storage and breakdown. |
| Energy homeostasis maintenance | The product, malonyl-CoA, is a building block for fatty acid synthesis. It can also inhibit carnitine palmitoyltransferase-1 (CPT-1), thereby preventing fatty acids from entering mitochondria for β-oxidation. When energy is abundant, it promotes fat storage. |
| Insulin Sensitivity Regulation | In the liver and adipose tissues, abnormal activity is closely associated with the development of insulin resistance and metabolic syndrome. |
| Cell Proliferation Support | Provide essential precursors for membrane lipid synthesis to rapidly proliferating cells (such as tumor cells), meeting their requirements for cell membrane construction and energy. |
The activity regulation curve of this enzyme exhibits the typical characteristics of a regulatory enzyme, being allosterically regulated by citrate (an activator) and long-chain fatty acyl-CoA (an inhibitor). This enables it to respond sensitively to the energy state of the cell. Its regulatory properties are more similar to "molecular switches" in metabolic pathways rather than simple linear catalysts.
Applications of ACACA and ACACA Antibody in Literature
1. Li, Xinyi, et al. "T266M variants of ANGPTL4 improve lipid metabolism by modifying their binding affinity to acetyl-CoA carboxylase in obstructive sleep apnea." Annals of Medicine 56.1 (2024): 2337740. https://doi.org/10.1080/07853890.2024.2337740
The article indicates that the serum level of ANGPTL4 significantly decreases as OSA worsens. Although the T266M mutation is not directly related to blood lipids, it can enhance the stability of binding with the ACACA protein, suggesting that it may regulate lipid metabolism.
2. Dong, Jian, et al. "ACACA reduces lipid accumulation through dual regulation of lipid metabolism and mitochondrial function via AMPK-PPARα-CPT1A axis." Journal of Translational Medicine 22.1 (2024): 196.https://doi.org/10.1186/s12967-024-04942-0
In this study, a NAFLD mouse model was established through a high-fat diet. It was found that inhibiting the key fat synthesis enzyme ACACA could reduce lipid deposition in liver cells and improve mitochondrial function. The mechanism was related to the activation of the AMPK-PPARα-CPT1A pathway.
3. Wu, Yu-Peng, et al. "ND630 controls ACACA and lipid reprogramming in prostate cancer by regulating the expression of circKIF18B_003." Journal of Translational Medicine 21.1 (2023): 877.https://doi.org/10.1186/s12967-023-04760-w
The article indicates that circKIF18B_003 is overexpressed in cancer tissues. It upregulates ACACA by adsorbing miR-370-3p and promotes the progression of prostate cancer. The new molecule ND630 inhibits ACACA and lipid metabolism reprogramming by regulating this circRNA, and may become a new therapeutic target.
4. Zhang, Hui, et al. "Down-regulation of ACACA suppresses the malignant progression of Prostate Cancer through inhibiting mitochondrial potential." Journal of Cancer 12.1 (2021): 232. https://doi.org/10.7150/jca.49560
The article indicates that ACACA is highly expressed in prostate cancer. Inhibiting ACACA can reduce the proliferation of cancer cells and their ability to form tumors in the body, while also decreasing mitochondrial ATP and mtDNA, suggesting a new therapeutic strategy targeting ACACA and mitochondria.
5. Huang, Yung-Chi, et al. "Involvement of ACACA (acetyl-CoA carboxylase α) in the lung pre-metastatic niche formation in breast cancer by senescence phenotypic conversion in fibroblasts." Cellular Oncology 46.3 (2023): 643-660. https://doi.org/10.1007/s13402-022-00767-5
This study reveals that in the pre-ecological formation of lung metastasis from breast cancer, the expression of ACACA in lung fibroblasts is downregulated, leading to cell senescence and the secretion of CXCL1, which subsequently recruits immunosuppressive cells and promotes the establishment of the pre-metastatic microenvironment.
Creative Biolabs: ACACA Antibodies for Research
Creative Biolabs specializes in the production of high-quality ACACA antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom ACACA Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our ACACA antibodies, custom preparations, or technical support, contact us at email.
Reference
- Li, Xinyi, et al. "T266M variants of ANGPTL4 improve lipid metabolism by modifying their binding affinity to acetyl-CoA carboxylase in obstructive sleep apnea." Annals of Medicine 56.1 (2024): 2337740. https://doi.org/10.1080/07853890.2024.2337740
Anti-ACACA antibodies
 Loading...
Loading...
Hot products 
-
Rabbit Anti-CBL Recombinant Antibody (D4E10) (CBMAB-CP0149-LY)

-
Mouse Anti-CCN2 Recombinant Antibody (CBFYC-2383) (CBMAB-C2456-FY)

-
Mouse Anti-CD83 Recombinant Antibody (HB15) (CBMAB-C1765-CQ)

-
Mouse Anti-CCS Recombinant Antibody (CBFYC-1093) (CBMAB-C1150-FY)

-
Mouse Anti-CIITA Recombinant Antibody (CBLC160-LY) (CBMAB-C10987-LY)

-
Mouse Anti-BCL6 Recombinant Antibody (CBYY-0435) (CBMAB-0437-YY)

-
Mouse Anti-CD24 Recombinant Antibody (SN3) (CBMAB-C1037-CQ)

-
Mouse Anti-ARHGDIA Recombinant Antibody (CBCNA-009) (CBMAB-R0415-CN)

-
Mouse Anti-GLP1R Recombinant Antibody (4F3) (CBMAB-G0521-LY)

-
Mouse Anti-BLNK Recombinant Antibody (CBYY-0623) (CBMAB-0626-YY)

-
Mouse Anti-CECR2 Recombinant Antibody (CBWJC-2465) (CBMAB-C3533WJ)

-
Mouse Anti-ABL2 Recombinant Antibody (V2-179121) (CBMAB-A0364-YC)

-
Mouse Anti-BBS2 Recombinant Antibody (CBYY-0253) (CBMAB-0254-YY)

-
Mouse Anti-HTLV-1 gp46 Recombinant Antibody (CBMW-H1006) (CBMAB-V208-1154-FY)

-
Rabbit Anti-ADRA1A Recombinant Antibody (V2-12532) (CBMAB-1022-CN)

-
Mouse Anti-ASB9 Recombinant Antibody (1D8) (CBMAB-A0529-LY)

-
Mouse Anti-DLL4 Recombinant Antibody (D1090) (CBMAB-D1090-YC)

-
Mouse Anti-CEMIP Recombinant Antibody (3C12) (CBMAB-K0296-LY)

-
Rabbit Anti-Acetyl-Histone H3 (Lys36) Recombinant Antibody (V2-623395) (CBMAB-CP0994-LY)

-
Rat Anti-CD300A Recombinant Antibody (172224) (CBMAB-C0423-LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






