APOC3 Antibodies
Background
The APOC3 gene encodes apolipoprotein C-III and is mainly present in very low-density lipoprotein and high-density lipoprotein. This protein regulates the metabolism and clearance of triglycerides in plasma by inhibiting the activity of lipoprotein lipase and hindering the liver's uptake of triglyceride-rich lipoproteins. Studies have shown that loss-of-function mutations in APOC3 are associated with low triglyceride levels and a reduced risk of cardiovascular diseases. Since its discovery in the 1970s, APOC3 has become a key target in the research of metabolic diseases. Its mechanism of action and regulatory network have been continuously and deeply explored, significantly enhancing the understanding of the pathological mechanisms of lipid metabolism disorders, atherosclerosis and related metabolic syndromes.
Structure of APOC3
APOC3 is a small apolipoprotein with a molecular weight of approximately 8.8 kDa. There are slight differences in its molecular weight among different species, which mainly result from minor changes in the amino acid sequence.
| Species | Human | Mouse | Rat |
| Molecular Weight (kDa) | 8.8 | 8.9 | 8.7 |
| Primary Structural Differences | 79 amino acids, including a domain rich in proline | Highly homologous to humans and functionally similar | The sequence is highly conserved and is a commonly used animal model |
The APOC3 protein is composed of 79 amino acids, and its primary structure features a hydrophilic N-terminal and a hydrophobic C-terminal domain rich in proline. This amphiphilic structure determines its biological function of binding to lipoprotein particles. Its core functional area is located at the C-terminal. By inhibiting the activity of lipoprotein lipase (LPL) and interacting with receptors, it negatively regulates the clearance rate of triglyceride-rich lipoproteins.
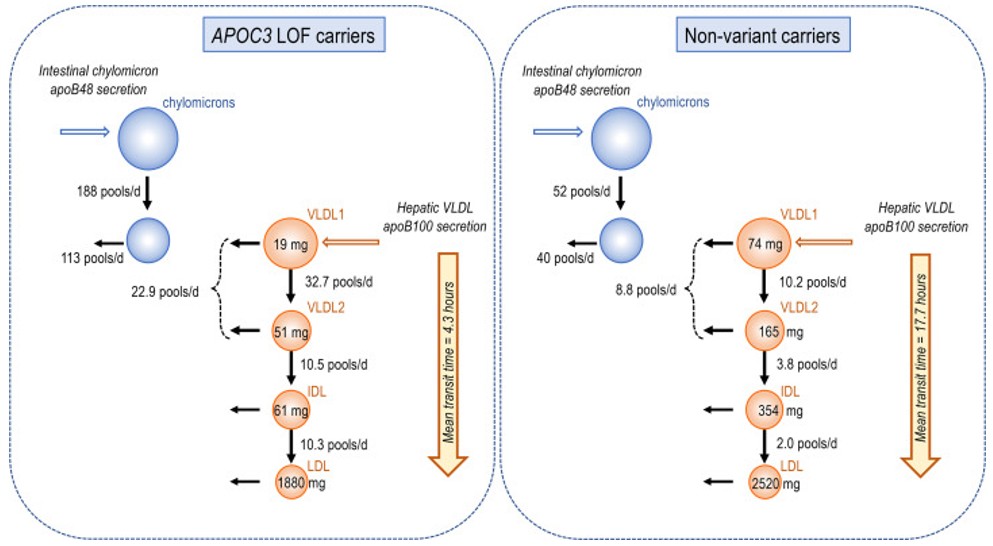 Fig. 1 Key Kinetic Parameter Differences in APOC3 Loss-of-Function Carriers.1
Fig. 1 Key Kinetic Parameter Differences in APOC3 Loss-of-Function Carriers.1
Key structural properties of APOC3:
- Distribution of amphiphilic domains
- Core functional domain
- Specific glycosylation sites
Functions of APOC3
The main function of APOC3 is to regulate the metabolism and clearance of plasma triglycerides, and it is also involved in various pathophysiological processes such as atherosclerosis and inflammatory responses.
| Function | Description |
| Regulation of lipid metabolism | By inhibiting the activity of lipoprotein lipase (LPL), the decomposition of triglyceride-rich lipoproteins (such as chylomicrons and VLDL) is delayed. |
| Inhibition of liver clearance | The uptake of lipoprotein residues by liver receptors (such as the LDL receptor family) is hindered, resulting in prolonged retention in plasma. |
| Cardiovascular risk | Elevated levels are directly associated with hypertriglyceridemia, atherosclerotic plaque formation, and increased risk of cardiovascular disease. |
| Inflammatory regulation | May be affected by lipoprotein particle composition and function, indirectly participate in the process of vascular endothelial inflammatory response. |
| Potential therapeutic targets | Silencing or inhibiting APOC3 has become an important therapeutic strategy for reducing triglycerides and preventing cardiovascular events. |
The inhibitory effect of APOC3 on lipoprotein lipase is A direct and crucial regulatory mechanism, which antagonizes the promoting function of apolipoprotein A-V and jointly maintains the dynamic balance of lipid metabolism. Its functional gain is significantly positively correlated with the risk of cardiovascular diseases.
Applications of APOC3 and APOC3 Antibody in Literature
1. Gouni-Berthold, Ioanna, Jonas Schwarz, and Heiner K. Berthold. "Updates in drug treatment of severe hypertriglyceridemia." Current Atherosclerosis Reports 25.10 (2023): 701-709. https://doi.org/10.1007/s11883-023-01140-z
The article indicates that new treatment options for severe hypertriglyceridemia have emerged: ASO (such as volanesorsen) targeting APOC3 and ANGPTL3 and siRNA drugs (such as ARO-APOC3) have shown potential, but no therapy has been proven to clearly reduce the risk of acute pancreatitis.
2. Wilkens, Trine L., et al. "Associations between alcohol consumption and HDL subspecies defined by ApoC3, ApoE and ApoJ: The Cardiovascular Health Study." Current problems in cardiology 48.1 (2023): 101395. https://doi.org/10.1016/j.cpcardiol.2022.101395
The article indicates that the analysis of 2,092 elderly subjects in this study shows that alcohol consumption is positively correlated with HDL subtypes with or without apoC3, apoE, and apoJ, as well as apoA1 levels (an increase of about 1% per week for each additional drink). apoC3 is weakly associated with alcohol consumption. The HDL protein components may affect its anti-arteriosclerosis function.
3. Zhou, Jiahe, et al. "ApoC3 is expressed in oocytes and increased expression is associated with PCOS progression." Journal of Ovarian Research 16.1 (2023): 188. https://doi.org/10.1186/s13048-023-01263-6
This study has for the first time discovered that APOC3 is highly expressed in oocytes of patients with polycystic ovary syndrome. Its level increases with the progression of the disease and is significantly correlated with multiple sex hormones, suggesting that APOC3 may be involved in the regulation of PCOS progression and oocyte development.
4. Holzer, Michael, et al. "ApoC3 Attenuates Platelet Activation Through GPIIb/IIIa Receptor Interaction." Cells 14.18 (2025): 1411. https://doi.org/10.3390/cells14181411
Research has found that apolipoprotein C3 can rapidly inhibit platelet activation, significantly reduce their aggregation and adhesion, and partially exert its effects through GPIIb/IIIa receptors. This suggests that APOC3 has a new function in the regulation of thrombosis risk.
5. Xu, Yitong, et al. "Targeting ApoC3 paradoxically aggravates atherosclerosis in hamsters with severe refractory hypercholesterolemia." Frontiers in Cardiovascular Medicine 9 (2022): 840358. https://doi.org/10.3389/fcvm.2022.840358
The study found through the construction of a APOC3/LDLR double knockout hamster model that although inhibiting APOC3 could lower triglycerides, it failed to improve refractory hypercholesterolemia and unexpectedly accelerated the progression of atherosclerosis, accompanied by abnormal platelet indicators.
Creative Biolabs: APOC3 Antibodies for Research
Creative Biolabs specializes in the production of high-quality APOC3 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom APOC3 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our APOC3 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Taskinen, Marja-Riitta, et al. "Postprandial metabolism of apolipoproteins B48, B100, C-III, and E in humans with APOC3 loss-of-function mutations." JCI insight 7.19 (2022): e160607. https://doi.org/10.1172/jci.insight.160607
Anti-APOC3 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-DHFR Recombinant Antibody (D0821) (CBMAB-D0821-YC)

-
Mouse Anti-AK4 Recombinant Antibody (V2-180419) (CBMAB-A1891-YC)

-
Rat Anti-ADGRE4 Recombinant Antibody (V2-160163) (CBMAB-F0011-CQ)

-
Mouse Anti-Acetyl-α-Tubulin (Lys40) Recombinant Antibody (V2-623485) (CBMAB-CP2897-LY)

-
Mouse Anti-CCN2 Recombinant Antibody (CBFYC-2383) (CBMAB-C2456-FY)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

-
Mouse Anti-COL1A2 Recombinant Antibody (CF108) (V2LY-1206-LY626)

-
Mouse Anti-CD33 Recombinant Antibody (P67.6) (CBMAB-C10189-LY)

-
Mouse Anti-DISP2 Monoclonal Antibody (F66A4B1) (CBMAB-1112CQ)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

-
Mouse Anti-AQP2 Recombinant Antibody (G-3) (CBMAB-A3359-YC)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-1728) (CBMAB-2077-YY)

-
Mouse Anti-GFP Recombinant Antibody (28) (CBMAB-G3038-LY)

-
Mouse Anti-FTH1 Recombinant Antibody (CBXF-1896) (CBMAB-F3426-CQ)

-
Rabbit Anti-DLK1 Recombinant Antibody (9D8) (CBMAB-D1061-YC)

-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Mouse Anti-ACTB Recombinant Antibody (V2-179553) (CBMAB-A0870-YC)

-
Mouse Anti-CTNND1 Recombinant Antibody (CBFYC-2414) (CBMAB-C2487-FY)

-
Mouse Anti-Acetyl SMC3 (K105/K106) Recombinant Antibody (V2-634053) (CBMAB-AP052LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






