CD1d Antibodies
Background
CD1d exists as a non-classical major histocompatibility complex I molecule and is mainly expressed on the surface of antigen-presenting cells. This protein binds to endogenous lipids and exogenous glycolipids and presents them to natural killer T cells, thereby achieving immunoregulatory functions and playing a key role in autoimmune diseases, infections, and tumor surveillance. Mammals such as humans and mice maintain immune balance by relying on CD1d, as their immune systems need to recognize different types of lipid antigens. It was first identified and cloned by the Michael Brenner team in 1995. CD1d is also the first member of the CD1 family to have its three-dimensional structure revealed, and the characteristics of its antigen-binding groove were determined through X-ray crystallography analysis. This discovery laid the foundation for understanding the mechanism of lipid antigen recognition. Its unique hydrophobic binding pocket structure has since been widely studied, greatly promoting our understanding of non-peptide antigen presentation, lipid immune recognition, and immune regulatory pathways.
Structure of CD1d
CD1d is a type I transmembrane glycoprotein with a relative molecular mass of approximately 37 kDa. This protein varies among different species, and the specific parameters are shown in the table below:
| Species | Human | Mouse | Rat |
|---|---|---|---|
| Molecular Weight (kDa) | 37 | 38 | 38 |
| Primary Structural Differences | The α1 and α2 domains form the antigen-binding groove | The slot structure is slightly different | Highly homologous to mice |
This protein is composed of a heavy chain and β2-microglobulin. Through its extracellular α1 and α2 domains, it forms a deep and hydrophobic antigen-binding groove, specifically designed for binding and presenting lipid antigens. The tertiary structure of the CD1d molecule consists of the two-sided walls formed by α helices and the bottom composed of β sheets. Two large hydrophobic pockets (A' and F' pockets) can accommodate lipid chains of different lengths. The key amino acid residues on the α helices are stabilized in the binding of lipid antigens through hydrogen bonds and hydrophobic interactions, while the β2-microglobulin maintains the stability of the overall structure.
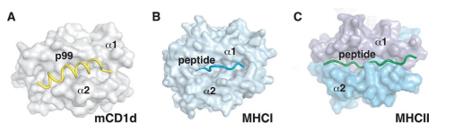 Fig. 1 Peptide binding to MHC and CD1d molecules.1
Fig. 1 Peptide binding to MHC and CD1d molecules.1
Key structural properties of CD1d:
- Heterodimeric structure consisting of a heavy chain and β2-microglobulin
- The deep hydrophobic antigen binding slot is formed by the α1 and α2 domains
- Two hydrophobic pockets (A' and F' pockets) accommodating fatty acid chains
- Polar residues within the binding groove mediate the recognition of lipid antigens
Functions of CD1d
The main function of the CD1d molecule is to present lipid antigens to natural killer T cells. However, it also plays a role in various immune regulatory processes, including autoimmune regulation and anti-infection immunity.
| Function | Description |
| Lipid Antigen Presentation | CD1d binds and presents endogenous and exogenous lipid antigens, activating natural killer T cells. |
| Immune Regulation | By activating natural killer T cells and rapidly releasing various cytokines, the direction of the immune response can be regulated. |
| Autoimmune Protection | In experimental models such as autoimmune encephalomyelitis, the absence of CD1d leads to the aggravation of the disease. |
| Anti-infection immunity | Participates in the recognition of bacterial lipid antigens, exerting immune surveillance function in the early stage of microbial infection. |
| Tumor Immune Surveillance | The CD1d-NK T cell axis recognizes abnormal lipids on the surface of tumor cells and mediates anti-tumor immunity. |
The lipid antigen recognition mechanism mediated by CD1d is different from the presentation of peptide antigens by MHC molecules. It accommodates the acyl chains of lipid antigens in a hydrophobic binding groove and exposes the polar heads for recognition by the T cell receptor.
Applications of CD1d and CD1d Antibody in Literature
1. Girardi, Enrico, Jing Wang, and Dirk M. Zajonc. "Structure of an α-helical peptide and lipopeptide bound to the nonclassical major histocompatibility complex (MHC) class I molecule CD1d." Journal of Biological Chemistry 291.20 (2016): 10677-10683. https://doi.org/10.1074/jbc.M115.702118
The article indicates that mouse CD1d can bind and present hydrophobic peptide segment p99. Its crystal structure shows that the peptide segment is bound in an α-helix conformation in the antigen binding groove, with the motif residues embedded inwardly in the deep groove, elucidating the molecular mechanism of peptide binding. Lipid peptides can also bind to CD1d in a similar conformation, revealing a new class of antigenic molecules.
2. Iwamura, Chiaki, and Toshinori Nakayama. "Role of CD1d-and MR1-restricted T cells in asthma." Frontiers in immunology 9 (2018): 1942. https://doi.org/10.3389/fimmu.2018.01942
The article indicates that congenital T cells rapidly exert their effector functions by recognizing non-peptide antigens presented by CD1d or MR1 through constant receptors. This article reviews the mechanism of action of iNKT and MAIT cells in allergic diseases such as asthma, and explores their potential as therapeutic targets.
3. Lee, Sung Won, et al. "Roles and therapeutic potential of CD1d-Restricted NKT cells in inflammatory skin diseases." Frontiers in immunology 13 (2022): 979370. https://doi.org/10.3389/fimmu.2022.979370
The article indicates that NKT cells are a type of innate T cells that recognize glycolipid antigens presented by CD1d. This article reviews the immunoregulatory role of CD1d-dependent NKT cells in skin inflammation such as atopic dermatitis, and explores the functional heterogeneity of its subgroups and its therapeutic potential.
4. King, Lisa A., et al. "CD1d-invariant natural killer T cell-based cancer immunotherapy: α-galactosylceramide and beyond." Frontiers in immunology 9 (2018): 1519. https://doi.org/10.3389/fimmu.2018.01519
The article indicates that after iNKT cells are activated by antigens presented via CD1d, they can directly kill tumors or initiate anti-tumor immunity by releasing cytokines. This article reviews various immunotherapy strategies based on iNKT cells, including glycolipid antigens, adoptive transfer, and CAR modification.
5. Nishioka, Yusuke, et al. "CD1d-restricted type II NKT cells reactive with endogenous hydrophobic peptides." Frontiers in immunology 9 (2018): 548. https://doi.org/10.3389/fimmu.2018.00548
The article indicates that CD1d-restricted type II NKT cells recognize endogenous hydrophobic peptides through diverse TCRs. They are opposite to the pro-inflammatory function of type I NKT cells and exert anti-inflammatory or pro-inflammatory effects in different models, participating in the regulation of autoimmune diseases.
Creative Biolabs: CD1d Antibodies for Research
Creative Biolabs specializes in the production of high-quality CD1d antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom CD1d Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our CD1d antibodies, custom preparations, or technical support, contact us at email.
Reference
- Girardi, Enrico, Jing Wang, and Dirk M. Zajonc. "Structure of an α-helical peptide and lipopeptide bound to the nonclassical major histocompatibility complex (MHC) class I molecule CD1d." Journal of Biological Chemistry 291.20 (2016): 10677-10683. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1074/jbc.M115.702118
Anti-CD1d antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-DDC Recombinant Antibody (8E8) (CBMAB-0992-YC)

-
Mouse Anti-CD1C Recombinant Antibody (L161) (CBMAB-C2173-CQ)

-
Mouse Anti-ANXA7 Recombinant Antibody (A-1) (CBMAB-A2941-YC)

-
Mouse Anti-dsDNA Recombinant Antibody (22) (CBMAB-AP1954LY)

-
Mouse Anti-BLNK Recombinant Antibody (CBYY-0623) (CBMAB-0626-YY)

-
Mouse Anti-CCS Recombinant Antibody (CBFYC-1093) (CBMAB-C1150-FY)

-
Rabbit Anti-ALOX5AP Recombinant Antibody (CBXF-1219) (CBMAB-F0750-CQ)

-
Mouse Anti-FOXL1 Recombinant Antibody (CBXF-0845) (CBMAB-F0462-CQ)

-
Mouse Anti-CAT Recombinant Antibody (724810) (CBMAB-C8431-LY)

-
Human Anti-SARS-CoV-2 S1 Monoclonal Antibody (CBFYR-0120) (CBMAB-R0120-FY)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Rabbit Anti-DLK1 Recombinant Antibody (9D8) (CBMAB-D1061-YC)

-
Mouse Anti-AZGP1 Recombinant Antibody (CBWJZ-007) (CBMAB-Z0012-WJ)

-
Mouse Anti-EMP3 Recombinant Antibody (CBFYE-0100) (CBMAB-E0207-FY)

-
Rabbit Anti-CAMK2A Recombinant Antibody (BA0032) (CBMAB-0137CQ)

-
Mouse Anti-Acetyl SMC3 (K105/K106) Recombinant Antibody (V2-634053) (CBMAB-AP052LY)

-
Rabbit Anti-AP2M1 (Phosphorylated T156) Recombinant Antibody (D4F3) (PTM-CBMAB-0610LY)

-
Mouse Anti-HTLV-1 gp46 Recombinant Antibody (CBMW-H1006) (CBMAB-V208-1154-FY)

-
Mouse Anti-ARID1B Recombinant Antibody (KMN1) (CBMAB-A3546-YC)

-
Mouse Anti-BIRC5 Recombinant Antibody (6E4) (CBMAB-CP2646-LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






