groEL Antibodies
Background
The groEL gene encodes a highly conserved molecular chaperone protein, which is mainly present in the organelles of bacteria and eukaryotes. This protein forms a large barrel-shaped complex and helps unfolded or misfolded polypeptide chains to fold correctly through an ATP-dependent mechanism, thereby maintaining protein homeostasis within the cell. Under stress conditions, the expression of GroEL is upregulated to address the risk of protein aggregation and ensure cell survival. Since its structure and function were discovered in the 1970s, they have been deeply analyzed and have become a model system in the field of molecular chaperone research. The research on the mechanism of this protein has greatly advanced people's understanding of fundamental biological processes such as protein folding, conformational regulation, and cellular stress response.
Structure of groEL
GroEL is a relatively large molecular chaperone protein with a molecular weight of approximately 57-60 kDa (monomer). This molecular weight is relatively conserved among different species, mainly due to the highly conserved ring structure and functional requirements of groEL..
| Species | E. coli | Thermophilic bacteria | Human mitochondria |
| Molecular Weight (kDa) | 57.2 | 58.0 | 60.0 |
| Primary Structural Differences | The classic model system features two seven-ring stacks | Higher thermal stability, adapt to high temperature | With mitochondrial targeting sequences, ring structure is similar |
The groEL protein is composed of approximately 548 amino acids and folds into a large bicring-shaped barrel structure (tetramer). Its tertiary and quaternary structures form hollow cylindrical chambers, providing an isolation environment for the folding of substrate proteins. The structure of the groEL protein is mainly composed of the ATPase domain and the apical domain. The apical domain is rich in hydrophobic residues and can bind to unfolded substrate proteins. The cooperative conformational changes between the ring structures, relying on the hydrolysis of ATP, drive the hydrophobic to hydrophilic conversion of the inner wall of the chamber, thereby promoting the correct folding and release of substrate proteins.
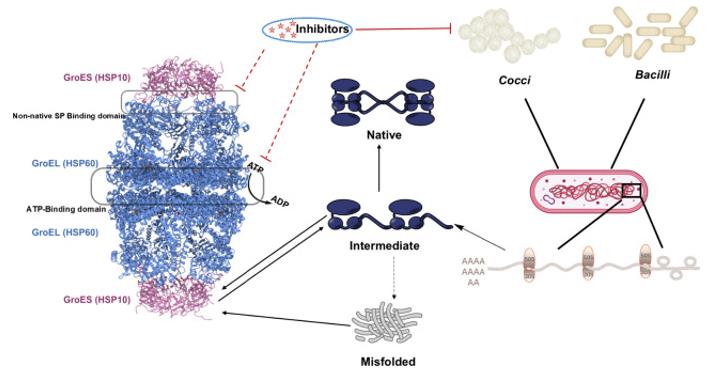 Fig. 1 Mechanistic pathway of GroEL/ES (HSP60/10) inhibitors.1
Fig. 1 Mechanistic pathway of GroEL/ES (HSP60/10) inhibitors.1
Key structural properties of groEL:
- Large bicyclic barrel-shaped complex structure (tetradomer)
- Central hydrophobic chamber can be combined with folding polypeptide
- ATP binding and hydrolysis of hinge regions that drive conformational changes
Functions of groEL
The main function of the molecular chaperone protein encoded by the groEL gene is to assist in the folding and assembly of proteins within cells. However, it is also involved in a variety of key cellular processes, including stress protection, protein quality control and cell cycle regulation.
| Function | Description |
| Protein folding | By utilizing the energy provided by ATP hydrolysis, an isolated bar-shaped chamber is offered for unfolded or misfolded polypeptide chains, assisting them in completing the correct three-dimensional conformation folding. |
| Stress protection | Under the condition of thermal shock stress expression level, prevent the cytotoxicity caused by protein denaturation gathered, is a core component of heat shock response. |
| Protein quality control | Identify and combine intermediates with partial folding or incorrect conformation to prevent irreversible aggregation and assist in guiding them towards the correct folding pathway or degradation pathway. |
| Oligomer assembly | Assist certain multi-subunit protein complexes in completing the correct assembly of their subunits to form functional oligomer structures. |
| Cell cycle and survival | Essential for the survival of many bacteria and organelles (e.g., mitochondria, chloroplasts), inactivation of which often leads to growth arrest or cell death. |
Unlike hemoglobin that relies on synergistic effects, the mechanism of action of groEL depends on the synergistic allostery of its bicyclic structure and the ATP hydrolysis cycle. This mechanism enables it to orderly bind, encapsulate and release substrate proteins through repetitive conformational changes, achieving efficient folding assistance rather than simple binding and dissociation.
Applications of groEL and groEL Antibody in Literature
1. Klaes, Simon, et al. "GroEL-proteotyping of bacterial communities using tandem mass spectrometry." International Journal of Molecular Sciences 24.21 (2023): 15692. https://doi.org/10.3390/ijms242115692
This paper proposes the GroEL protein typing method. Through mass spectrometry analysis of GroEL peptides, high-abundance bacterial families in bacterial communities can be rapidly identified without the need for amplification primers. Efficient species resolution can be directly achieved based on SDS-PAGE pre-separation and scalable databases.
2. Marchenkov, Victor V., and Gennady V. Semisotnov. "GroEL-assisted protein folding: does it occur within the chaperonin inner cavity?." International Journal of Molecular Sciences 10.5 (2009): 2066-2083. https://doi.org/10.3390/ijms10052066
The mainstream view in this article holds that GroEL, with the assistance of GroES, helps proteins fold within the lumen. This review focuses on the evidence that proteins can also complete folding after detaling from the chaperone complex and discusses the related regulatory dissociation models.
3. Iizuka, Ryo, and Takashi Funatsu. "Chaperonin GroEL uses asymmetric and symmetric reaction cycles in response to the concentration of non-native substrate proteins." Biophysics and physicobiology 13 (2016): 63-69. https://doi.org/10.2142/biophysico.13.0_63
The traditional view holds that GroEL and GroES only form asymmetric complexes for protein folding. This paper reviews the formation mechanism of symmetric complexes and points out that GroEL can switch between symmetric and asymmetric reaction cycles depending on the substrate concentration.
4. Taguchi, Hideki, and Ayumi Koike-Takeshita. "In vivo client proteins of the chaperonin GroEL-GroES provide insight into the role of chaperones in protein evolution." Frontiers in Molecular Biosciences 10 (2023): 1091677. https://doi.org/10.3389/fmolb.2023.1091677
The article indicates that the traditional view holds that proteins fold within the GroEL cavity. New research indicates that substrate proteins can be detached from GroEL to complete folding in solution and proposes a ligand-regulated dissociation model.
5. Wang, Yuming, et al. "Exploring Novel Antibiotics by Targeting the GroEL/GroES Chaperonin System." ACS Pharmacology & Translational Science 8.1 (2024): 10-20. https://doi.org/10.1021/acsptsci.4c00397
The article indicates that inhibiting the GroEL/ES chaperone protein system can disrupt bacterial protein homeostasis and is a potential target for new antibacterial drugs. This article reviews the research progress of the system's functions and its inhibitors, providing a direction for developing new strategies against drug-resistant bacteria.
Creative Biolabs: groEL Antibodies for Research
Creative Biolabs specializes in the production of high-quality groEL antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom groEL Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our groEL antibodies, custom preparations, or technical support, contact us at email.
Reference
- Wang, Yuming, et al. "Exploring Novel Antibiotics by Targeting the GroEL/GroES Chaperonin System." ACS Pharmacology & Translational Science 8.1 (2024): 10-20. https://doi.org/10.1021/acsptsci.4c00397
Anti-groEL antibodies
 Loading...
Loading...
Hot products 
-
Rabbit Anti-B2M Recombinant Antibody (CBYY-0059) (CBMAB-0059-YY)

-
Mouse Anti-CCDC25 Recombinant Antibody (CBLC132-LY) (CBMAB-C9786-LY)

-
Mouse Anti-BLK Recombinant Antibody (CBYY-0618) (CBMAB-0621-YY)

-
Mouse Anti-CSPG4 Recombinant Antibody (CBFYM-1050) (CBMAB-M1203-FY)

-
Mouse Anti-FAS2 Monoclonal Antibody (1D4) (CBMAB-0071-CN)

-
Mouse Anti-AFDN Recombinant Antibody (V2-58751) (CBMAB-L0408-YJ)

-
Mouse Anti-CCND2 Recombinant Antibody (DCS-3) (CBMAB-G1318-LY)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

-
Human Anti-SARS-CoV-2 Spike Recombinant Antibody (CR3022) (CBMAB-CR014LY)

-
Mouse Anti-ADIPOR1 Recombinant Antibody (V2-179982) (CBMAB-A1368-YC)

-
Mouse Anti-CASP7 Recombinant Antibody (10-01-62) (CBMAB-C2005-LY)

-
Mouse Anti-CD46 Recombinant Antibody (CBFYC-0076) (CBMAB-C0085-FY)

-
Mouse Anti-FYN Recombinant Antibody (10) (CBMAB-S6332-CQ)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Rabbit Anti-Acetyl-Histone H4 (Lys16) Recombinant Antibody (V2-623415) (CBMAB-CP1021-LY)

-
Mouse Anti-Acetyl SMC3 (K105/K106) Recombinant Antibody (V2-634053) (CBMAB-AP052LY)

-
Mouse Anti-ALX1 Recombinant Antibody (96k) (CBMAB-C0616-FY)

-
Mouse Anti-CCS Recombinant Antibody (CBFYC-1093) (CBMAB-C1150-FY)

-
Rabbit Anti-AKT2 (Phosphorylated S474) Recombinant Antibody (V2-556130) (PTM-CBMAB-0605LY)

-
Mouse Anti-AMH Recombinant Antibody (5/6) (CBMAB-A2527-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






