SOD3 Antibodies
Background
The protein encoded by the SOD3 gene is an extracellular superoxide dismutase, mainly present in extracellular matrix and vascular walls. It is highly expressed in lung, kidney and vascular tissues. This enzyme protects cells from oxidative damage by eliminating superoxide anion free radicals, and is crucial for maintaining vascular function. In 1982, Marklund et al. first discovered SOD3 in human plasma. It is the only member of the superoxide dismutase family that is secreted to the extracellular space. Its unique heparin-binding domain enables it to anchor to the cell surface. The discovery of SOD3 provides an important theoretical basis for understanding the mechanisms of cardiovascular diseases such as hypertension and atherosclerosis.
Structure of SOD3
SOD3 is an extracellular glycoprotein, and its molecular weight is approximately 30-35 kDa, which is affected by glycosylation modification. There are certain differences in molecular weight and structural characteristics among SOD3 from different species sources:
| Species | Human | Mouse | Rat | Pig | Bovine |
|---|---|---|---|---|---|
| Molecular Weight (kDa) | 30.9 | 32.5 | 32.3 | 31.2 | 30.5 |
| Primary Structural Differences | Containing a heparin-binding domain, anchoring the extracellular matrix | The C-terminal domain shows significant differences from that of humans. | The organizational distribution characteristics are different from those of individuals | The enzyme active center is highly conserved | With a relatively high content in the plasma |
The SOD3 protein is composed of approximately 240 amino acids and forms a homologous tetramer structure. Each subunit contains a copper-zinc binding site that constitutes the catalytic active center. The C-terminal region rich in basic amino acids has a heparin-binding domain that mediates its binding to the cell surface and extracellular matrix. The protein core is a β-barrel structure, and the conformation is maintained stable through disulfide bonds. The assembly of the tetramer forms a central pore, through which the substrate superoxide anion can enter the active center. This enzyme exerts antioxidant protection in the extracellular environment by eliminating superoxide radicals.
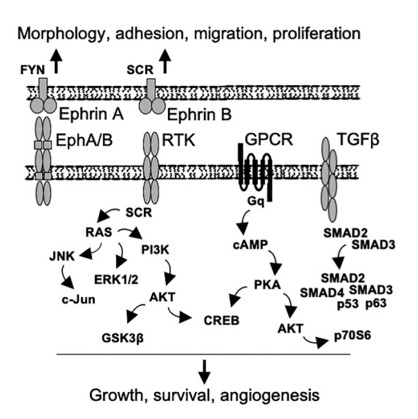 Fig. 1 Putative schematic presentation of the signaling pathways regulated by SOD3.1
Fig. 1 Putative schematic presentation of the signaling pathways regulated by SOD3.1
Key structural properties of SOD3:
- Homotrimeric structure, subunits assembled through non-covalent interactions
- C-terminal heparin-binding domain mediates binding to extracellular matrix and cell surface
- Each subunit contains a catalytic active center composed of copper and zinc ions
- Histidine residues form a metal ion coordination environment and maintain enzyme activity
Functions of SOD3
The main function of SOD3 is to eliminate superoxide anion free radicals in the extracellular space, protecting tissues from oxidative damage. Additionally, it is involved in regulating various physiological processes such as vascular function and inflammatory responses.
| Function | Description |
|---|---|
| Superoxide Anion Removal | SOD3 catalyzes the disproportionation reaction of superoxide anions, generating hydrogen peroxide and oxygen, thereby reducing the level of oxidative stress. |
| Vascular Protection | By maintaining the bioavailability of nitric oxide, it protects the function of vascular endothelium, regulates vasodilation and blood pressure . |
| Anti-inflammatory Effect | Reduces the activation of inflammatory signals mediated by superoxide anions, inhibits inflammatory cell infiltration and tissue damage. |
| Tissue Remodeling | Participates in the regulation of extracellular matrix metabolism, affecting tissue repair and fibrosis process. |
| Signal Regulation | Affects cell proliferation, differentiation and apoptosis-related signaling pathways by regulating the level of reactive oxygen species. |
SOD3, as a key component of the extracellular antioxidant defense system, has an extremely high catalytic efficiency for superoxide anions at its active center, but it has no significant affinity. This characteristic enables it to continuously eliminate superoxide free radicals that have diffused into the extracellular space.
Applications of SOD3 and SOD3 Antibody in Literature
1. Parascandolo, Alessia, and Mikko O. Laukkanen. "SOD3 is a non-mutagenic growth regulator affecting cell migration and proliferation signal transduction." Antioxidants 10.5 (2021): 635. https://doi.org/10.3390/antiox10050635
The article indicates that SOD3 regulates signal transduction by generating hydrogen peroxide. At physiological concentrations, it promotes cell growth, while at supra-physiological concentrations, it induces apoptosis. Studies have shown that although SOD3 overexpression activates multiple signaling pathways, it does not cause DNA damage and has no mutagenic properties. There is a significant difference in kinase activity between SOD3 and SV40-induced immortalized cells, suggesting a potential drug target.
2. Xu, Yidan, et al. "Placenta-derived SOD3 deletion impairs maternal behavior via alterations in FGF/FGFR-prolactin signaling axis." Cell reports 43.10 (2024). https://doi.org/10.1016/j.celrep.2024.114789
The article indicates that the deletion of placental SOD3 leads to a decrease in prolactin in female mice, as well as behavioral defects in maternal behavior, and inhibition of the pituitary FGF pathway. Mechanistically, it regulates the methylation of Fgf1/Fgfr2 through the TET/IDH/αKG pathway, affecting the expression of prolactin, and the behavioral defects of the mother can be transmitted to the offspring.
3. Li, Qilong, et al. "SOD3 suppresses early cellular immune responses to parasite infection." Nature Communications 15.1 (2024): 4913. https://doi.org/10.1038/s41467-024-49348-0
The article indicates that malaria parasites induce the secretion of SOD3 by neutrophils. This protein inhibits the expression of IL-2 in T cells and the IFN-γ response, thereby weakening the host's immune clearance ability. The survival rate of SOD3-deficient mice significantly increased, suggesting that it is a key regulatory factor for parasite immune evasion.
4. Wang, Yanwei, et al. "Uncovering SOD3 and GPX4 as new targets of Benzo [α] pyrene-induced hepatotoxicity through Metabolomics and Chemical Proteomics." Redox biology 67 (2023): 102930. https://doi.org/10.1016/j.redox.2023.102930
The article indicates that the environmental pollutant benzo[a]pyrene disrupts the redox balance of liver cells and weakens the antioxidant capacity by directly binding and inhibiting the activities of SOD3 and GPX4. This study integrates metabolomics and chemobiology methods to reveal a new mechanism of benzo[a]pyrene's liver toxicity.
5. Sun, Shuaiqi, et al. "SOD3 overexpression alleviates cerebral ischemia‐reperfusion injury in rats." Molecular genetics & genomic medicine 7.10 (2019): e00831. https://doi.org/10.1002/mgg3.831
The article indicates that the transplantation of mesenchymal stem cells with overexpressed SOD3 can significantly reduce the infarction volume in rats with cerebral ischemia-reperfusion and improve neurological function. The mechanism is related to the upregulation of Bcl-2 and the downregulation of Bax expression, which inhibits cell apoptosis. This provides a new idea for the treatment of ischemic stroke.
Creative Biolabs: SOD3 Antibodies for Research
Creative Biolabs specializes in the production of high-quality SOD3 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom SOD3 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our SOD3 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Parascandolo, Alessia, and Mikko O. Laukkanen. "SOD3 is a non-mutagenic growth regulator affecting cell migration and proliferation signal transduction." Antioxidants 10.5 (2021): 635. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/antiox10050635
Anti-SOD3 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-CIITA Recombinant Antibody (CBLC160-LY) (CBMAB-C10987-LY)

-
Mouse Anti-ADV Recombinant Antibody (V2-503423) (CBMAB-V208-1364-FY)

-
Mouse Anti-BSN Recombinant Antibody (219E1) (CBMAB-1228-CN)

-
Mouse Anti-ALDOA Recombinant Antibody (A2) (CBMAB-A2316-YC)

-
Mouse Anti-ACKR3 Recombinant Antibody (V2-261265) (CBMAB-C1023-LY)

-
Mouse Anti-FeLV g27 Recombinant Antibody (1) (CBMAB-V208-1714-FY)

-
Mouse Anti-ATP1A2 Recombinant Antibody (M7-PB-E9) (CBMAB-A4013-YC)

-
Mouse Anti-CD46 Recombinant Antibody (CBFYC-0076) (CBMAB-C0085-FY)

-
Rabbit Anti-B2M Recombinant Antibody (CBYY-0059) (CBMAB-0059-YY)

-
Mouse Anti-C5B-9 Recombinant Antibody (CBFYA-0216) (CBMAB-X0304-FY)

-
Mouse Anti-CARD11 Recombinant Antibody (CBFYC-0811) (CBMAB-C0866-FY)

-
Mouse Anti-ADAM12 Recombinant Antibody (V2-179752) (CBMAB-A1114-YC)

-
Mouse Anti-CD19 Recombinant Antibody (CBXC-1224) (CBMAB-C1491-CQ)

-
Mouse Anti-GFP Recombinant Antibody (28) (CBMAB-G3038-LY)

-
Rat Anti-FABP3 Recombinant Antibody (CBXF-2299) (CBMAB-F1612-CQ)

-
Mouse Anti-CCDC25 Recombinant Antibody (CBLC132-LY) (CBMAB-C9786-LY)

-
Mouse Anti-ANXA7 Recombinant Antibody (A-1) (CBMAB-A2941-YC)

-
Mouse Anti-ACLY Recombinant Antibody (V2-179314) (CBMAB-A0610-YC)

-
Mouse Anti-AFDN Recombinant Antibody (V2-58751) (CBMAB-L0408-YJ)

-
Mouse Anti-ENO2 Recombinant Antibody (85F11) (CBMAB-0276CQ)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






