VCAM1 Antibodies
Background
VCAM1 is an immunoglobulin superfamily transmembrane protein located on the cell surface, mainly expressed by endothelial cells under inflammatory stimulation. The protein encoded by this gene mediates the adhesion process between immune cells and vascular endothelium by binding to α4β1 integrin on the surface of white blood cells, thereby regulating inflammatory responses and immune cell migration. In chronic inflammatory diseases such as atherosclerosis and rheumatoid arthritis, the high expression of VCAM1 has been confirmed to be closely related to the development of the lesion. This gene was first identified in 1989. The study of its three-dimensional structure revealed the specific binding mechanism between immunoglobulin domains and integrins. Continuous research on VCAM1 has deepened people's understanding of the molecular mechanisms underlying inflammatory regulation, intercellular recognition, and the formation of the immune microenvironment.
Structure of VCAM1
VCAM1 is a transmembrane glycoprotein with a molecular weight of approximately 81-110 kDa. Its differences mainly stem from variations in gene splicing and glycosylation modifications among different species.
| Species | Human | Mouse | Rat | Bovine |
| Molecular Weight (kDa) | 81-110 | 78-108 | 79-109 | 82-111 |
| Primary Structural Differences | Seven IG-like domains and a relatively long cytoplasmic region | Six IG-like domains, shorter cytoplasmic regions | Highly homologous to mice | Seven IG-like domains, highly conserved to humans |
The extracellular region of this protein contains 6 to 7 immunoglobulin-like domains, among which the 1st and 4th domains are the key regions for binding to integrins. Its tertiary structure forms a rigidly extended rod-like conformation and is anchored to the cell membrane through transmembrane regions. N-terminal glycosylation modification plays an important regulatory role in protein stability and ligand recognition, while the phosphorylation of serine residues at the cytoplasmic tail is involved in the regulation of intracellular signal transduction processes.
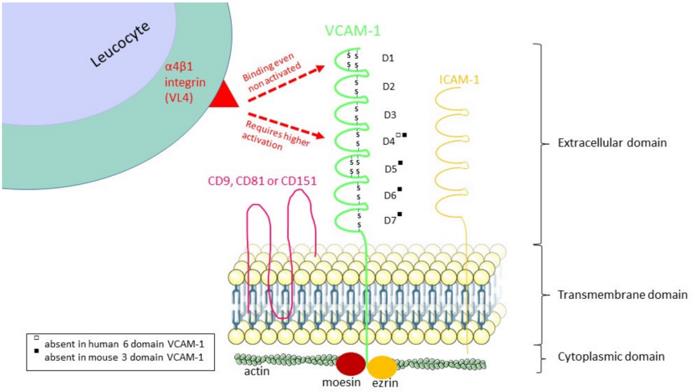 Fig. 1 The structure of VCAM-1 and its α4β1 integrin binding regions.1
Fig. 1 The structure of VCAM-1 and its α4β1 integrin binding regions.1
Key structural properties of VCAM1:
- Extracellular immunoglobulin-like domains are repeatedly arranged
- Rigidly extended rod-shaped spatial conformation
- Specific integrin binding interface
Functions of VCAM1
The core function of VCAM1 is to mediate the adhesion and migration of immune cells to vascular endothelium. However, this molecule is also involved in a variety of pathophysiological processes, including inflammatory signal transduction and regulation of the immune microenvironment.
| Function | Description |
| Leukocyte adhesion | By specifically binding to α4β1 integrins through domains 1 and 4, it mediates the stable adhesion of lymphocytes, monocytes and activated endothelium. |
| Migration of inflammatory cells | Activate intracellular signal transduction to guide immune cells to cross the vascular endothelial barrier and migrate directionally towards the inflammatory site. |
| Regulation of immune response | Increased expression in chronic inflammatory lesions, and promote the immune cell infiltration and maintain a stable local immune microenvironment. |
| Atherosclerosis formation | Promote the retention and differentiation of monocytes under the endothelium, and accelerate the formation and development of atherosclerotic plaques. |
| Regulation of tumor metastasis | Abnormal expression in tumor vascular endothelium affects the homing and distant metastasis process of circulating tumor cells. |
The binding of VCAM1 to integrins exhibits dynamically tunable characteristics, and its affinity is regulated by both the cytokine microenvironment and the glycosylation status. This feature makes it an important intervention target for inflammatory and immune diseases.
Applications of VCAM1 and VCAM1 Antibody in Literature
1. Perner, Caroline, et al. "Plasma VCAM1 levels correlate with disease severity in Parkinson's disease." Journal of neuroinflammation 16.1 (2019): 94. https://doi.org/10.1186/s12974-019-1482-8
Research has found that the expression of VLA4 in patients with Parkinson's disease is down-regulated, the level of plasma sVCAM1 is significantly increased, and it is related to the degree of movement disorder, suggesting dysfunction of the VCAM1-VLA4 axis.
2. Hession, Catherine, et al. "Cloning of an alternate form of vascular cell adhesion molecule-1 (VCAM1)." Journal of Biological Chemistry 266.11 (1991): 6682-6685. https://doi.org/10.1016/S0021-9258(20)89551-9
Research has found that VCAM1 exists in two mRNA forms, long and short, which are produced by alternative splicing. The long type is the main form of existence, and both can mediate VLA-4 integrin-dependent immune cell adhesion.
3. Wang, Tongyu, Jiahu Tian, and Yuanzhe Jin. "VCAM1 expression in the myocardium is associated with the risk of heart failure and immune cell infiltration in myocardium." Scientific Reports 11.1 (2021): 19488. https://doi.org/10.1038/s41598-021-98998-3
Research has found that VCAM1 plays a core role in heart failure caused by ischemic and dilated cardiomyopathy. Its expression disorder is associated with immune cell infiltration and the risk of disease, and it may serve as a potential biomarker and therapeutic target.
4. Uosaki, Hideki, et al. "Efficient and scalable purification of cardiomyocytes from human embryonic and induced pluripotent stem cells by VCAM1 surface expression." PloS one 6.8 (2011): e23657.https://doi.org/10.1371/journal.pone.0023657
Research has found that VCAM1 can serve as a highly specific surface marker for human stem cell-derived cardiomyocytes, enabling high-purity and high-efficiency separation without genetic modification, providing a key technical foundation for cardiac cell therapy.
5. Maglinger, Benton, et al. "Intracranial VCAM1 at time of mechanical thrombectomy predicts ischemic stroke severity." Journal of Neuroinflammation 18.1 (2021): 109. https://doi.org/10.1186/s12974-021-02157-4
Research has found that elevated intracranial and systemic VCAM1 levels in patients with acute large vessel occlusion stroke are significantly associated with hypertension, infarct volume, and poor recovery of neurological function, suggesting that it can serve as a potential therapeutic target.
Creative Biolabs: VCAM1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality VCAM1 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom VCAM1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our VCAM1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Thayse, Kathleen, et al. "VCAM-1 target in non-invasive imaging for the detection of atherosclerotic plaques." Biology 9.11 (2020): 368. https://doi.org/10.3390/biology9110368
Anti-VCAM1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-ENO1 Recombinant Antibody (8G8) (CBMAB-E1329-FY)

-
Mouse Anti-AFDN Recombinant Antibody (V2-58751) (CBMAB-L0408-YJ)

-
Mouse Anti-APOA1 Monoclonal Antibody (CBFYR0637) (CBMAB-R0637-FY)

-
Mouse Anti-CCL18 Recombinant Antibody (64507) (CBMAB-C7910-LY)

-
Mouse Anti-BIRC3 Recombinant Antibody (315304) (CBMAB-1214-CN)

-
Mouse Anti-GFP Recombinant Antibody (28) (CBMAB-G3038-LY)

-
Mouse Anti-AHCYL1 Recombinant Antibody (V2-180270) (CBMAB-A1703-YC)

-
Mouse Anti-F11R Recombinant Antibody (402) (CBMAB-0026-WJ)

-
Mouse Anti-CD83 Recombinant Antibody (HB15) (CBMAB-C1765-CQ)

-
Mouse Anti-CEMIP Recombinant Antibody (3C12) (CBMAB-K0296-LY)

-
Mouse Anti-APOE Recombinant Antibody (A1) (CBMAB-0078CQ)

-
Mouse Anti-ABIN2 Recombinant Antibody (V2-179106) (CBMAB-A0349-YC)

-
Mouse Anti-AMIGO2 Recombinant Antibody (CBYY-C0756) (CBMAB-C2192-YY)

-
Mouse Anti-BLK Recombinant Antibody (CBYY-0618) (CBMAB-0621-YY)

-
Mouse Anti-BRD3 Recombinant Antibody (CBYY-0801) (CBMAB-0804-YY)

-
Rabbit Anti-Acetyl-Histone H4 (Lys16) Recombinant Antibody (V2-623415) (CBMAB-CP1021-LY)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7697) (CBMAB-1869CQ)

-
Human Anti-SARS-CoV-2 S1 Monoclonal Antibody (CBFYR-0120) (CBMAB-R0120-FY)

-
Mouse Anti-CIITA Recombinant Antibody (CBLC160-LY) (CBMAB-C10987-LY)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






