VWA1 Antibodies
Background
The VWA1 gene encodes an extracellular matrix protein containing the von Willebrand factor A domain. It is mainly expressed in connective tissues such as cartilage, skeletal muscle, and vascular walls. This protein mediates the interaction between cells and the extracellular matrix through its domain, participating in processes such as matrix assembly, cell adhesion, and migration. It plays a crucial role in tissue development and homeostasis maintenance. Mutations in the VWA1 gene are associated with various genetic connective tissue disorders, and its abnormal expression may also affect bone development and muscle function. This gene was initially identified through genomic methods, and subsequent studies have revealed its key position in the extracellular matrix network. The functional analysis of its domain provides an important model for understanding the mechanism of cell-matrix interaction.
Structure of VWA1
The protein encoded by the VWA1 gene has a molecular weight of approximately 52 kDa and shows certain differences among different species. The composition of its domains and the conservation of its amino acid sequence reflect the similarities and specificities in function.
| Species | Human | Mouse | Rat | Zebrafish |
| Molecular Weight (kDa) | 52 | 53 | 53 | 50 |
| Primary Structural Differences | Contains VWA domain, sequence highly conserved | High homology, similar functions | High homology, similar functions | Consistent domain composition, adapted to aquatic environment |
The VWA1 protein is composed of approximately 450 amino acids. Its three-dimensional structure is centered around the von Willebrand factor A domain, which folds into a typical α/β Rossmann-like topology. The central part is a hydrophobic β-sheet, and the two sides are surrounded by α-helices. This domain mediates the interaction with ligands through a metal ion-dependent coordinating MIDAS motif in the extracellular matrix, performing functions such as cell adhesion and matrix assembly. The secondary structure of the VWA1 protein alternately arranges α-helices and β-strands, jointly forming a stable conformation. The key aspartic and serine residues in the MIDAS motif participate in the coordination of metal ions, directly influencing the binding specificity of the protein. Additionally, the C-terminal extension region of VWA1 may be involved in regulating its interaction with collagen or proteoglycans, thereby playing multiple roles in the homeostasis of connective tissues.
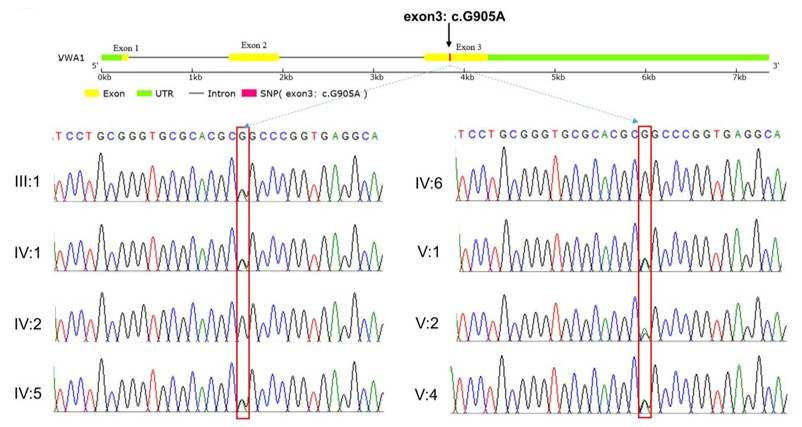 Fig. 1 Sanger sequencing results of the VWA1 mutation in the pedigree.1
Fig. 1 Sanger sequencing results of the VWA1 mutation in the pedigree.1
Key structural properties of VWA1:
- The characteristic VWA domain
- The MIDAS motif mediates ligand binding dependent on divalent metal ions
- Multiple cysteine residues form intramolecular disulfide bonds to stabilize the conformation
- The C-terminal domain participates in the interaction of the extracellular matrix network
- The hydrophobic amino acid residue-rich region promotes the correct folding of the protein
Functions of VWA1
The extracellular matrix protein encoded by the VWA1 gene mainly mediates the interaction between cells and the matrix through its VWA domain, and plays multiple roles in tissue development and homeostasis maintenance.
| Function | Description |
| Cell adhesion | The VWA domain binds to ligands through the MIDAS motif, facilitating specific adhesion of cells to the extracellular matrix. |
| Matrix assembly | Participates in the formation and stabilization of the collagen fiber network, maintaining the integrity of the connective tissue structure. |
| Cartilage development | Highly expressed in cartilage tissue, it regulates the extracellular matrix environment to support the differentiation and functional maintenance of chondrocytes. |
| Vascularization regulation | By interacting with the matrix components of the vascular wall, it indirectly affects the behavior of endothelial cells and the formation of new blood vessels. |
| Tissue injury repair | Expresses at an elevated level at the injury site, participates in the process of matrix remodeling, and promotes tissue regeneration and repair. |
The ligand-binding kinetics of the protein encoded by the VWA1 gene exhibit a typical saturation curve characteristic. The affinity of this protein for integrins and extracellular matrix ligands is jointly regulated by metal ions in the MIDAS motif. This binding mode is in line with the dynamic and reversible interactions required during cell adhesion.
Applications of VWA1 and VWA1 Antibody in Literature
1. Wang, Yibei, et al. "A mutation in VWA1, encoding von Willebrand factor A domain-containing protein 1, is associated with hemifacial microsomia." Frontiers in Cell and Developmental Biology 8 (2020): 571004. https://doi.org/10.3389/fcell.2020.571004
The article indicates that the discovery of the VWA1 gene p.R302Q mutation through whole exome sequencing is associated with familial hemifacial microsomia. The functional experiments in zebrafish confirmed that this gene causes facial deformities by inhibiting the proliferation of cranial neural crest cells and disrupting the structure of pharyngeal cartilage cells.
2. Niu, Xiaomin, et al. "vwa1 knockout in zebrafish causes abnormal craniofacial chondrogenesis by regulating FGF pathway." Genes 14.4 (2023): 838. https://doi.org/10.3390/genes14040838
The article indicates that by using CRISPR/Cas9 to construct a zebrafish vwa1 gene knockout model, it was confirmed that this gene regulates the aggregation, differentiation and proliferation of cranial neural crest cells by influencing the FGF signaling pathway, thereby causing malformations in the development of pharyngeal cartilage.
3. Nagy, Sara, et al. "Autosomal recessive VWA1-related disorder: comprehensive analysis of phenotypic variability and genetic mutations." Brain communications 6.6 (2024): fcae377. https://doi.org/10.1093/braincomms/fcae377
The study found that the double allelic variation of the VWA1 gene leads to a new type of hereditary motor neuron disease, characterized by early-onset proximal weakness, joint laxity, and upper motor neuron signs. This disease mimics various neuromuscular disorders and accounts for 1% of cases in undiagnosed neuropathies.
4. Athamneh, Mohammed, et al. "Proteomic studies in VWA1‐related neuromyopathy allowed new pathophysiological insights and the definition of blood biomarkers." Journal of Cellular and Molecular Medicine 28.8 (2024): e18122. https://doi.org/10.1111/jcmm.18122
Through proteomics analysis of the blood and muscles of patients with VWA1-related neuromuscular diseases, differences in expression of proteins such as HNRNPDL and oxidative stress-related plasma markers were discovered. This provides new evidence for the study of disease mechanisms and clinical detection.
5. Pagnamenta, Alistair T., et al. "An ancestral 10-bp repeat expansion in VWA1 causes recessive hereditary motor neuropathy." Brain 144.2 (2021): 584-600.https://doi.org/10.1093/brain/awaa420
The study found that the double allelic variation of the VWA1 gene leads to autosomal recessive hereditary motor neuropathy. The carrier frequency in Europe is approximately 1/1000, accounting for 1% of the undiagnosed cases. The ancestral mutation p.G25Rfs*74 causes the disease through nonsense-mediated decay.
Creative Biolabs: VWA1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality VWA1 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom VWA1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our VWA1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Wang, Yibei, et al. "A mutation in VWA1, encoding von Willebrand factor A domain-containing protein 1, is associated with hemifacial microsomia." Frontiers in Cell and Developmental Biology 8 (2020): 571004. Distributed under Open Access license CC BY 4.0, cropped from the original figure. https://doi.org/10.3389/fcell.2020.571004
Anti-VWA1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-FN1 Monoclonal Antibody (D6) (CBMAB-1240CQ)

-
Mouse Anti-BAX Recombinant Antibody (CBYY-0216) (CBMAB-0217-YY)

-
Mouse Anti-EPO Recombinant Antibody (CBFYR0196) (CBMAB-R0196-FY)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7719) (CBMAB-1898CQ)

-
Rabbit Anti-AKT2 (Phosphorylated S474) Recombinant Antibody (V2-556130) (PTM-CBMAB-0605LY)

-
Mouse Anti-ADIPOR2 Recombinant Antibody (V2-179983) (CBMAB-A1369-YC)

-
Mouse Anti-ALB Recombinant Antibody (V2-363290) (CBMAB-S0173-CQ)

-
Mouse Anti-BAD (Phospho-Ser136) Recombinant Antibody (CBYY-0138) (CBMAB-0139-YY)

-
Mouse Anti-dsDNA Recombinant Antibody (22) (CBMAB-AP1954LY)

-
Mouse Anti-CCN2 Recombinant Antibody (CBFYC-2383) (CBMAB-C2456-FY)

-
Mouse Anti-CD63 Recombinant Antibody (CBXC-1200) (CBMAB-C1467-CQ)

-
Rabbit Anti-BRCA2 Recombinant Antibody (D9S6V) (CBMAB-CP0017-LY)

-
Mouse Anti-CRTAM Recombinant Antibody (CBFYC-2235) (CBMAB-C2305-FY)

-
Rat Anti-FABP3 Recombinant Antibody (CBXF-2299) (CBMAB-F1612-CQ)

-
Mouse Anti-APOE Recombinant Antibody (A1) (CBMAB-0078CQ)

-
Mouse Anti-CCDC25 Recombinant Antibody (CBLC132-LY) (CBMAB-C9786-LY)

-
Mouse Anti-AKT1/AKT2/AKT3 (Phosphorylated T308, T309, T305) Recombinant Antibody (V2-443454) (PTM-CBMAB-0030YC)

-
Mouse Anti-BPGM Recombinant Antibody (CBYY-1806) (CBMAB-2155-YY)

-
Human Anti-SARS-CoV-2 Spike Recombinant Antibody (CBC05) (CBMAB-CR005LY)

-
Mouse Anti-DMD Recombinant Antibody (D1190) (CBMAB-D1190-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






