DNER Antibodies
Background
The DNER gene encodes a transmembrane protein that is specifically expressed in the nervous system. It is mainly distributed on the surfaces of developing neurons and some glial cells. This protein acts as an atypical Notch ligand and regulates the differentiation direction of neural stem cells by binding to Notch receptors. It also participates in dendrite growth and synapse formation during the maturation of neurons. Its expression pattern in specific brain regions is associated with neurodevelopmental disorders. Recent studies suggest that abnormal DNER function may be involved in the pathological processes of schizophrenia and cerebellar ataxia. This gene was first identified by Eiraku et al. in 2002 through screening specific molecules in mouse brain tissues. Subsequent studies have confirmed that it plays an irreplaceable role in maintaining the structural and functional integrity of Purkinje cells in the cerebellum. Currently, this molecule has become an important research object in the field of neurodevelopmental biology, providing a new perspective for revealing the mechanism of injury repair in the central nervous system and exploring intervention targets for related diseases.
Structure of DNER
The DNER gene encodes a transmembrane protein with a molecular weight of approximately 80 kDa. The molecular weight of this protein varies slightly among different species due to differences in amino acid sequences.
| Species | Human | Mouse | Rat | Chicken | Zebrafish |
| Molecular Weight (kDa) | 80.2 | 79.8 | 80.0 | 81.5 | 82.1 |
| Primary Structural Differences | The extracellular region contained 10 EGF-like replicates | The intracellular region is slightly shorter | Highly homologous to mice | Different numbers of EGF repetitions | Differences in the arrangement of extracellular domains |
The DNER protein contains approximately 700 amino acids and is a type I transmembrane protein. The extracellular region is rich in EGF-like repetitive sequences, while the intracellular region contains conserved tyrosine residues. This protein binds to the Notch receptor through the extracellular region and participates in signal transduction in the intracellular region. The disulfide bonds in the EGF domain of the extracellular region are crucial for maintaining conformational stability, and the transmembrane region anchors the protein to the membrane.
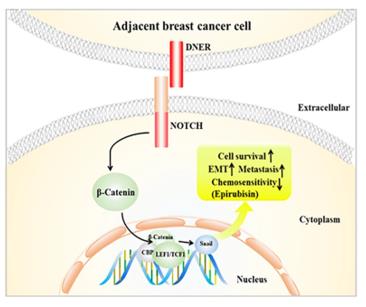 Fig. 1 Schematic model for DNER-induced biological function of BC cells.1
Fig. 1 Schematic model for DNER-induced biological function of BC cells.1
Key structural properties of DNER:
- Extracellular domain containing EGF-like repeat sequences
- Single transmembrane helical region
- Intracellular region contains conserved tyrosine residues
- Disulfide bonds stabilize the conformation of the EGF domain
- Non-classical Notch ligand, lacking DSL motif
Functions of DNER
The main function of DNER is to regulate the differentiation of neural stem cells and the maturation of neurons. In addition, it is also involved in processes such as synapse formation, glial cell activation, and cerebellar development.
| Function | Description |
| Neuronal Differentiation Regulation | As an atypical Notch ligand, it activates downstream signaling pathways and inhibits premature neuronal differentiation . |
| Axon Development | Promotes the formation of dendritic fields and the establishment of synaptic connections in cerebellar Purkinje cells. |
| Synaptic Plasticity | Participates in the maintenance of glutamatergic synaptic function and affects motor coordination and learning memory . |
| Glial Cell Activation | Expressed in Bergmann glial cells of the cerebellum, it regulates their protrusions and interaction with neurons. |
| Disease Association | Abnormal expression is associated with schizophrenia, ataxia, and repair of myelin sheath damage. |
DNER binds to the Notch receptor through its extracellular domain, without relying on the classical DSL motif. Its intracellular domain has no transcriptional activity, and the signal transduction is different from the classical Notch pathway.
Applications of DNER and DNER Antibody in Literature
1. Wang, Zhong, et al. "DNER promotes epithelial–mesenchymal transition and prevents chemosensitivity through the Wnt/β-catenin pathway in breast cancer." Cell death & disease 11.8 (2020): 642. https://doi.org/10.1038/s41419-020-02903-1
The article indicates that DNER is highly expressed in breast cancer, especially in triple-negative breast cancer, and is associated with poor prognosis. It induces epithelial-mesenchymal transition and inhibits apoptosis by activating the Wnt/β-catenin pathway, thereby promoting tumor proliferation and metastasis. DNER can serve as a potential therapeutic target.
2. Jin, Ran, et al. "A Multitask Deep Learning Framework for DNER." Computational Intelligence and Neuroscience 2022.1 (2022): 3321296. https://doi.org/10.1155/2022/3321296
This paper proposes a multi-task learning model named Multi-DTR. By jointly modeling the DNER and DNEN tasks, and integrating CNN, ELMo and BiLSTM-CRF to recognize drug names, this model performs exceptionally well on the DDI2011/2013 datasets, effectively enhancing the efficiency of biomedical text processing.
3. To, Han Thi Ngoc, et al. "Delta/Notch-like epidermal growth factor-related receptor (DNER), a potential prognostic marker of gastric cancer regulates cell survival and cell cycle progression." International Journal of Molecular Sciences 24.12 (2023): 10077. https://doi.org/10.3390/ijms241210077
The article indicates that DNER is highly expressed during the progression of gastric cancer. It promotes cell proliferation, invasion and inhibits apoptosis by activating the Notch signaling pathway, and is associated with the TGF-β signal. Studies have shown that DNER can serve as a potential prognostic marker and therapeutic target for gastric cancer.
4. Christensen, Peter Broegger, Henrik Gregersen, and Charlotte Almasi. "Anti-Tr/DNER antibody paraneoplastic cerebellar degeneration preceding a very late relapse of Hodgkin Lymphoma after 12 years." Cerebellum & Ataxias 8.1 (2021): 14. https://doi.org/10.1186/s40673-021-00137-1
The article indicates that a 76-year-old male patient with Hodgkin's lymphoma experienced gait instability and other cerebellar symptoms 12 years after achieving complete remission. Initially, he was misdiagnosed as having a stroke. Eventually, through positive anti-Tr/DNER antibodies and imaging examinations, it was confirmed that the tumor recurrence had caused paraneoplastic cerebellar degeneration. This case suggests that for patients with acute cerebellar syndromes, one should be vigilant of the possibility of rare paraneoplastic syndromes.
5. He, Rui, et al. "Transcriptional profiling reveals the regulatory role of DNER in promoting pancreatic neuroendocrine neoplasms." Frontiers in genetics 11 (2020): 587402. https://doi.org/10.3389/fgene.2020.587402
The article indicates that in pancreatic neuroendocrine tumors, the expression of DNER is significantly upregulated and is positively correlated with the expressions of RBPJ, SKP1, CTNNB1 and CDH2. Together, they promote tumor occurrence and development by activating the Wnt/Notch interaction signaling pathway.
Creative Biolabs: DNER Antibodies for Research
Creative Biolabs specializes in the production of high-quality DNER antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom DNER Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our DNER antibodies, custom preparations, or technical support, contact us at info@creative-biolabs.com.
Reference
- Wang, Zhong, et al. "DNER promotes epithelial–mesenchymal transition and prevents chemosensitivity through the Wnt/β-catenin pathway in breast cancer." Cell death & disease 11.8 (2020): 642. Distributed under the same Creative Commons license CC BY 4.0 as the original. Cropped from the original figure. https://doi.org/10.1038/s41419-020-02903-1
Anti-DNER antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-BAD (Phospho-Ser136) Recombinant Antibody (CBYY-0138) (CBMAB-0139-YY)

-
Mouse Anti-GIPC2 Recombinant Antibody (10) (CBMAB-G0476-LY)

-
Mouse Anti-ACKR3 Recombinant Antibody (V2-261265) (CBMAB-C1023-LY)

-
Mouse Anti-CCDC25 Recombinant Antibody (CBLC132-LY) (CBMAB-C9786-LY)

-
Rat Anti-AChR Recombinant Antibody (V2-12500) (CBMAB-0990-CN)

-
Rat Anti-EPO Recombinant Antibody (16) (CBMAB-E1578-FY)

-
Mouse Anti-C1QC Recombinant Antibody (CBFYC-0600) (CBMAB-C0654-FY)

-
Mouse Anti-EPO Recombinant Antibody (CBFYR0196) (CBMAB-R0196-FY)

-
Mouse Anti-CALR Recombinant Antibody (CBFYC-0763) (CBMAB-C0818-FY)

-
Mouse Anti-A2M Recombinant Antibody (V2-178822) (CBMAB-A0036-YC)

-
Mouse Anti-BAX Recombinant Antibody (CBYY-0216) (CBMAB-0217-YY)

-
Mouse Anti-APOH Recombinant Antibody (4D9A4) (CBMAB-A3249-YC)

-
Rat Anti-CD63 Recombinant Antibody (7G4.2E8) (CBMAB-C8725-LY)

-
Mouse Anti-BLNK Recombinant Antibody (CBYY-0623) (CBMAB-0626-YY)

-
Mouse Anti-4-Hydroxynonenal Recombinant Antibody (V2-502280) (CBMAB-C1055-CN)

-
Rabbit Anti-ABL1 (Phosphorylated Y245) Recombinant Antibody (V2-505716) (PTM-CBMAB-0465LY)

-
Mouse Anti-AAV9 Recombinant Antibody (V2-634029) (CBMAB-AP023LY)

-
Mouse Anti-ATP1B1 Recombinant Antibody (E4) (CBMAB-0463-LY)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7719) (CBMAB-1898CQ)

-
Mouse Anti-Acetyl SMC3 (K105/K106) Recombinant Antibody (V2-634053) (CBMAB-AP052LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






