EXO1 Antibodies
Background
The EXO1 gene encodes an exonuclease that plays a crucial role in the DNA repair process, mainly existing in the cell nucleus of eukaryotes. This protein, as a nucleic acid exonuclease, can recognize and remove the nucleotide sequences around the DNA damage sites, thereby participating in important genomic maintenance mechanisms such as mismatch repair and double-strand break repair. Cells need this precise repair process to maintain the stability of genetic information and prevent the accumulation of mutations that lead to cancer or other diseases. The human EXO1 gene was independently cloned and identified by multiple research teams as early as 1998. Subsequent studies revealed its functions in meiotic recombination and the generation of immunoglobulin diversity. Abnormal expression regulation of this gene is closely related to the occurrence and development of various cancers, making it an important focus of cancer research and potential therapeutic targets. Through in-depth studies of the structure and function of EXO1, scientists have been able to more comprehensively understand the complexity of the DNA repair network and its core role in maintaining genomic stability.
Structure of EXO1
The molecular weight of EXO1 protein is approximately 94 kDa. There are slight variations among different species due to differences in amino acid sequences.
| Species | Human | Mouse | Rat | Yeast |
| Molecular Weight (kDa) | 94 | 94 | 94 | ~106 |
| Primary Structural Differences | Contains multiple domains and has a high degree of homology with other species | Highly homologous to human EXO1 | Conservative functional area | The functions are the same but the sequences differ significantly |
This protein is composed of 846 amino acids and its structure contains multiple functional domains, including the N-terminal catalytic domain and the C-terminal protein interaction domain. EXO1, as a structure-specific nucleases, plays a central role in DNA metabolism. The secondary structure of the protein is mainly composed of α-helices and β-sheets, forming a specific catalytic active center. Its activity depends on the metal ion binding sites in the conserved nucleic acid exonuclease domain, and DNA strand cutting is achieved through the cooperative action of two metal ions. The C-terminal region of the protein mediates interactions with other DNA repair factors, ensuring the correct assembly of the repair complex.
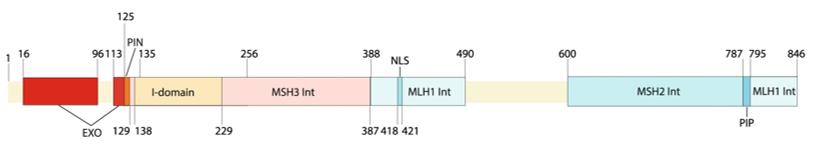 Fig. 1 Schematic overview of domains in the human EXO1 protein.1
Fig. 1 Schematic overview of domains in the human EXO1 protein.1
Key structural properties of EXO1:
- Conserved exonuclease domains containing metal ion binding sites
- The C-terminal protein interaction domain mediates binding to DNA repair factors
- Structure-specific nuclease activity, capable of recognizing and cutting various DNA structures
- The N-terminal catalytic core is responsible for enzyme activity, and phosphorylation sites are involved in cell cycle regulation
Functions of EXO1
The main function of EXO1 is to participate in DNA repair and maintain genomic stability. However, it is also involved in various cellular processes, including replication fork maintenance and immune system development.
| Function | Description |
| DNA mismatch repair | EXO1 identifies and removes mismatched bases generated during DNA replication, ensuring the accurate transmission of genetic information. |
| DNA double-strand break repair | By removing the nucleotides at the broken ends, a 3' single-strand bulge is produced, creating conditions for homologous recombination repair. |
| Meiotic recombination | During meiotic division of reproductive cells, it participates in the cross-over and exchange of homologous chromosomes, promoting genetic diversity. |
| Replication fork protection | When DNA replication is blocked, EXO1 participates in the handling and restart of the replication fork, preventing genomic instability. |
| Immune globulin maturation | In lymphocytes, it participates in frequent somatic mutations and class-switch recombination, promoting the generation of antibody diversity. |
The nucleic acid exonuclease activity of EXO1 is dependent on its conserved catalytic domain. It can gradually remove nucleotides from the single-stranded DNA in a 5'→3' direction. Unlike many DNA repair enzymes, EXO1 exhibits structural specificity and can act on various DNA substrates including gaps, ends, and branched structures, enabling it to play multifunctional roles in the DNA metabolic network.
Applications of EXO1 and EXO1 Antibody in Literature
1. Gao, Yuandi, et al. "A CRISPR-Cas9 screen identifies EXO1 as a formaldehyde resistance gene." Nature communications 14.1 (2023): 381. https://doi.org/10.1038/s41467-023-35802-y
In Fanconi anemia, the mechanism of DNA damage induced by formaldehyde is unclear. The study found that EXO1, by removing DNA-protein cross-links and protecting replication forks, responds to the replication stress and genomic instability caused by formaldehyde, and its function is independent of the FA pathway.
2. Roy, Megha, et al. "EXO1 promotes the meiotic MLH1-MLH3 endonuclease through conserved interactions with MLH1, MSH4 and DNA." Nature communications 16.1 (2025): 4141. https://doi.org/10.1038/s41467-025-59470-2
The article indicates that EXO1 interacts with MLH1 through the MIP/I403 motif and with MSH4 through the W371 residue, participating in the meiotic resolvase complex in a structural component form, and promoting the dissociation of the double Holliday Junction into crossover products.
3. Maric, Marija, et al. "EXO1 as a therapeutic target for Fanconi Anaemia, ZRSR2 and BRCA1-A complex deficient cancers." Nature communications 16.1 (2025): 8476. https://doi.org/10.1038/s41467-025-63349-7
The article indicates that the absence of EXO1 is associated with synthetic lethality in Fanconi anemia and ZRSR2 deficiency. The underlying mechanism is the reversal defect of the replication fork, accelerated speed, resulting in ssDNA exposure and DNA damage, suggesting that EXO1 is a potential target for DDR-deficient cancers.
4. Wang, Zefeng, et al. "EXO1/P53/SREBP1 axis-regulated lipid metabolism promotes prostate cancer progression." Journal of Translational Medicine 22.1 (2024): 104. https://doi.org/10.1186/s12967-023-04822-z
The article indicates that EXO1 is a new biomarker for prostate cancer. By inhibiting the P53 signal and upregulating SREBP1, it reprograms lipid metabolism to promote tumor progression, providing a potential therapeutic target for advanced prostate cancer.
5. Yang, Hongbin, et al. "Cyclin F–EXO1 axis controls cell cycle–dependent execution of double-strand break repair." Science Advances 10.32 (2024): eado0636. https://doi.org/10.1126/sciadv.ado0636
The article indicates that the SCF cyclin F ubiquitin ligase mediates the degradation of EXO1, preventing DNA end excision during mitosis, thereby promoting microhomology-mediated end-joining repair, and revealing a new mechanism for cell cycle regulation of DSB repair.
Creative Biolabs: EXO1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality EXO1 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom EXO1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our EXO1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Gao, Yuandi, et al. "A CRISPR-Cas9 screen identifies EXO1 as a formaldehyde resistance gene." Nature communications 14.1 (2023): 381. Distributed under Open Access license CC BY 4.0, and cropped from the original figure. https://doi.org/10.1038/s41467-023-35802-y
Anti-EXO1 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-GLP1R Recombinant Antibody (4F3) (CBMAB-G0521-LY)

-
Mouse Anti-CD24 Recombinant Antibody (SN3) (CBMAB-C1037-CQ)

-
Mouse Anti-AKT1 (Phosphorylated S473) Recombinant Antibody (V2-505430) (PTM-CBMAB-0067LY)

-
Mouse Anti-ENO1 Recombinant Antibody (CBYC-A950) (CBMAB-A4388-YC)

-
Mouse Anti-C5B-9 Recombinant Antibody (CBFYA-0216) (CBMAB-X0304-FY)

-
Mouse Anti-ALDOA Recombinant Antibody (A2) (CBMAB-A2316-YC)

-
Rabbit Anti-BRCA2 Recombinant Antibody (D9S6V) (CBMAB-CP0017-LY)

-
Rat Anti-CCR2 Recombinant Antibody (475301) (CBMAB-C1338-LY)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

-
Mouse Anti-EPO Recombinant Antibody (CBFYR0196) (CBMAB-R0196-FY)

-
Mouse Anti-APOE Recombinant Antibody (A1) (CBMAB-0078CQ)

-
Mouse Anti-FLT1 Recombinant Antibody (11) (CBMAB-V0154-LY)

-
Mouse Anti-CCL18 Recombinant Antibody (64507) (CBMAB-C7910-LY)

-
Mouse Anti-CIITA Recombinant Antibody (CBLC160-LY) (CBMAB-C10987-LY)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-0790) (CBMAB-0793-YY)

-
Mouse Anti-CD2AP Recombinant Antibody (BR083) (CBMAB-BR083LY)

-
Rat Anti-(1-5)-α-L-Arabinan Recombinant Antibody (V2-501861) (CBMAB-XB0003-YC)

-
Mouse Anti-DISP2 Monoclonal Antibody (F66A4B1) (CBMAB-1112CQ)

-
Mouse Anti-ALOX5 Recombinant Antibody (33) (CBMAB-1890CQ)

-
Mouse Anti-ALB Recombinant Antibody (V2-55272) (CBMAB-H0819-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






