IGFBP2 Antibodies
Background
IGFBP2, as a secreted multifunctional protein, is mainly present in the serum, tissue fluid, and various cellular microenvironments of mammals. This protein binds to insulin-like growth factors with high affinity, not only regulating the bioavailability of IGF and its downstream signaling pathways, but also directly acting on cell surface receptors in a manner independent of IGF, thereby influencing cell proliferation, migration, and apoptosis. In embryonic development, tissue repair, and metabolic homeostasis, IGFBP2 plays a crucial regulatory role. Its abnormal expression is closely related to the progression of various tumors and metabolic diseases. Since it was first purified and identified in 1988, IGFBP2 has become a research hotspot in the insulin-like growth factor binding protein family. The determination of its crystal structure has further revealed the molecular mechanism of the interaction between IGFBP2 and ligands and targets, laying the foundation for a deeper understanding of its multiple functions under physiological and pathological conditions.
Structure of IGFBP2
IGFBP2 is a secreted glycoprotein with a molecular weight of approximately 31 kDa. There are differences in length and glycosylation sites among different species.
| Species | Human | Mouse | Rat | Bovine | Pig |
| Molecular Weight (kDa) | 31.0 | 30.8 | 30.9 | 31.2 | 31.1 |
| Primary Structural Differences | Typical GCGCCxxC motif, rich in cysteines | The genetic similarity to humans is approximately 87%. | The genetic similarity to humans is approximately 86%. | Structurally highly conservative | The sequence is similar to that of humans |
IGFBP2 consists of 289 amino acids and is composed of three parts: the N-terminal conserved region, the linker region, and the C-terminal domain. Both the N-terminal and C-terminal regions are rich in cysteines and form multiple intramolecular disulfide bonds, jointly forming a highly specific IGF binding interface. The linker region is prone to proteolysis, resulting in functional fragments with independent activity. The N-terminal domain contains a characteristic GCGCCxxC motif, which is a hallmark of the IGFBP family; the thyroglobulin type-1 repeat sequence in the C-terminal domain is involved in non-IGF-dependent regulatory processes such as cell surface adhesion.
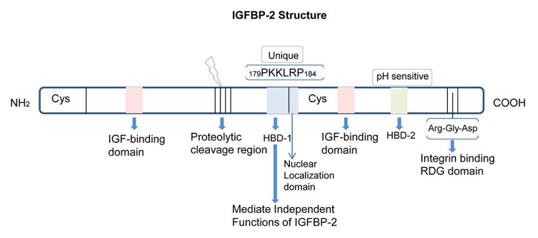 Fig. 1 Structure and domains of IGFBP-2.1
Fig. 1 Structure and domains of IGFBP-2.1
Key structural properties of IGFBP2:
- The N-terminal and C-terminal are highly conserved, each containing 12 to 14 cysteines
- Intra-chain disulfide bonds stabilize the 3D conformation and form the IGF high-affinity binding interface
- Central connection area for unstructured area, vulnerable to protease shear
Functions of IGFBP2
The main function of IGFBP2 is to regulate the bioavailability of insulin-like growth factors. It also participates in various non-IGF-dependent physiological processes, such as cell migration, adhesion and metabolic regulation.
| Function | Description |
| IGF signal regulation | It binds to IGF-I/II with high affinity, prolongs their half-life and regulates receptor activation, thereby influencing cell proliferation and differentiation. |
| Non-IGF-dependent action | It directly regulates cell migration, adhesion and survival by binding to the C-terminal domain with cell surface integrins. |
| Metabolic regulation | It affects glucose uptake and insulin sensitivity in adipose tissue and liver, and is related to obesity and diabetes. |
| Tumor progression participation | It is highly expressed in various tumors such as glioma and prostate cancer, promoting angiogenesis, invasion and metastasis. |
| Development and tissue repair | It is expressed at higher levels during embryonic development and after tissue damage, participating in neurogenesis, bone formation and wound healing. |
IGFBP2 possesses both IGF-dependent and IGF-independent regulatory mechanisms. Its functional switching mainly relies on the proteolytic cleavage of the linker region and changes in the cellular microenvironment.
Applications of IGFBP2 and IGFBP2 Antibody in Literature
1. Khan, Shumsuzzaman. "IGFBP-2 signaling in the brain: from brain development to higher order brain functions." Frontiers in Endocrinology 10 (2019): 822. https://doi.org/10.3389/fendo.2019.00822
The article indicates that IGFBP-2 is the most abundant neuro-regulatory polypeptide in cerebrospinal fluid, and is highly expressed in brain regions such as the hippocampus during brain development and in adulthood. It regulates neural plasticity through pathways that depend on or are independent of IGF receptors, and participates in advanced cognitive functions such as learning and memory. It is a neurotrophic factor throughout the entire life cycle.
2. Chen, Xiu, et al. "IGFBP2 drives epithelial-mesenchymal transition in hepatocellular carcinoma via activating the Wnt/β-catenin pathway." Infectious Agents and Cancer 18.1 (2023): 73. https://doi.org/10.1186/s13027-023-00543-6
The article indicates that insulin-like growth factor binding protein 2 (IGFBP2) is highly expressed in liver cancer. It promotes epithelial-mesenchymal transition (EMT) and tumor metastasis by activating the Wnt/β-catenin signaling pathway. Knockdown of this protein can reverse the expression of related proteins.
3. Alicea, Gretchen M., et al. "Age-related increases in IGFBP2 increase melanoma cell invasion and lipid synthesis." Cancer Research Communications 4.8 (2024): 1908-1918. https://doi.org/10.1158/2767-9764.CRC-23-0176
The article indicates that the skin fibroblasts of the elderly secrete high levels of IGFBP2. This secretion activates the PI3K pathway, promoting the fatty acid synthesis of melanoma cells, thereby enhancing their invasiveness and metastasis. Neutralizing IGFBP2 can inhibit tumor growth.
4. Li, Hui, et al. "IGFBP2 function as a novel biomarker for active lupus nephritis." Journal of Molecular Medicine 100.10 (2022): 1479-1491. https://doi.org/10.1007/s00109-022-02241-z
The article indicates that IGFBP2 is significantly elevated in patients with active lupus nephritis and can serve as a highly specific diagnostic marker. Neutralizing IGFBP2 can inhibit T cell activation, increase the proportion of regulatory T cells, and significantly improve mouse nephritis by inhibiting the AKT/mTOR pathway.
5. Lu, Hengxiao, et al. "IGFBP2/ITGA5 promotes gefitinib resistance via activating STAT3/CXCL1 axis in non-small cell lung cancer." Cell Death & Disease 15.6 (2024): 447. https://doi.org/10.1038/s41419-024-06843-y
The article indicates that IGFBP2 is highly expressed in patients with non-small cell lung cancer who are resistant to gefitinib. It upregulates CXCL1 by activating STAT3 and collaborates with ITGA5 to promote the survival of cancer cells in the drug environment, making it a potential biomarker of resistance and an intervention target.
Company A: IGFBP2 Antibodies for Research
Company A specializes in the production of high-quality IGFBP2 antibodies for research and industrial applications. Our portfolio includes monoclonal and polyclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom IGFBP2 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our IGFBP2 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Khan, Shumsuzzaman. "IGFBP-2 signaling in the brain: from brain development to higher order brain functions." Frontiers in Endocrinology 10 (2019): 822. Distributed under Open Access license CC BY 4.0. Cropped from the original figure. https://doi.org/10.3389/fendo.2019.00822
Anti-IGFBP2 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-BMI1 Recombinant Antibody (CBYC-P026) (CBMAB-P0108-YC)

-
Mouse Anti-AMH Recombinant Antibody (5/6) (CBMAB-A2527-YC)

-
Mouse Anti-CEMIP Recombinant Antibody (3C12) (CBMAB-K0296-LY)

-
Mouse Anti-G6PD Recombinant Antibody (13B331) (CBMAB-G1553-LY)

-
Mouse Anti-ATG5 Recombinant Antibody (9H197) (CBMAB-A3945-YC)

-
Rabbit Anti-ABL1 (Phosphorylated Y185) Recombinant Antibody (V2-443434) (PTM-CBMAB-0001YC)

-
Mouse Anti-EPO Recombinant Antibody (CBFYR0196) (CBMAB-R0196-FY)

-
Mouse Anti-BCL2L1 Recombinant Antibody (H5) (CBMAB-1025CQ)

-
Mouse Anti-AQP2 Recombinant Antibody (E-2) (CBMAB-A3358-YC)

-
Mouse Anti-CD46 Recombinant Antibody (CBFYC-0076) (CBMAB-C0085-FY)

-
Mouse Anti-ACVR1C Recombinant Antibody (V2-179685) (CBMAB-A1041-YC)

-
Mouse Anti-ELAVL4 Recombinant Antibody (6B9) (CBMAB-1132-YC)

-
Mouse Anti-CCDC6 Recombinant Antibody (CBXC-0106) (CBMAB-C5397-CQ)

-
Mouse Anti-ADAM12 Recombinant Antibody (V2-179752) (CBMAB-A1114-YC)

-
Mouse Anti-AAV-5 Recombinant Antibody (V2-503417) (CBMAB-V208-1369-FY)

-
Mouse Anti-ARHGAP5 Recombinant Antibody (54/P190-B) (CBMAB-P0070-YC)

-
Mouse Anti-CD63 Recombinant Antibody (CBXC-1200) (CBMAB-C1467-CQ)

-
Mouse Anti-CAT Recombinant Antibody (724810) (CBMAB-C8431-LY)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Mouse Anti-DISP2 Monoclonal Antibody (F66A4B1) (CBMAB-1112CQ)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






