PI16 Antibodies
Background
PI16 protein, as a secreted glycoprotein, is mainly present in the extracellular matrix and the matrix cells of specific tissues (such as the thymus and lymph nodes). This protein participates in maintaining tissue homeostasis by regulating the cell cycle progression and inhibiting cell proliferation, and plays a crucial role in immune regulation and fibrotic diseases. Its expression level is closely related to the remodeling of the tumor microenvironment in various cancers, and can affect immune cell infiltration and matrix remodeling. It was first identified in a T-cell screening experiment in 2004. PI16 has become a key molecule in the study of cell quiescence due to its unique function in cell cycle regulation. The relationship between its structure and function has been gradually clarified, providing an important model for understanding microenvironmental signal regulation, disease-related fibrosis, and immune escape mechanisms.
Structure of PI16
PI16 is a secreted glycoprotein, and its molecular weight varies among different species. For instance, the molecular weight of the human PI16 protein is approximately 20-22 kDa, while the homologous protein in mice is about 19 kDa. The following table lists the typical molecular weights and characteristics of this protein in some species:
| Species | Human | Mouse | Rat |
|---|---|---|---|
| Molecular Weight (kDa) | 20–22 | ~19 | ~19 |
| Primary Structural Differences | Containing a signal peptide and multiple potential glycosylation sites | High sequence homology and similar glycosylation patterns | Structurally conservative, with functions highly similar to those of humans. |
The PI16 protein is composed of approximately 160-180 amino acids. Its structure mainly includes a typical signal peptide sequence and several conserved domains, forming a secretable spherical protein. Its core functional domains can interact with extracellular matrix components and other signaling proteins. The secondary structure of this protein is mainly β-sheet, supplemented by several α-helix regions, jointly forming a stable three-dimensional conformation, enabling it to participate in regulating processes such as cell cycle arrest, immune regulation, and tissue fibrosis in the cellular microenvironment.
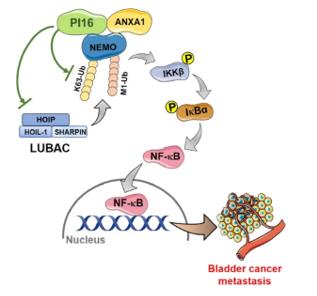 Fig. 1 Proposed model diagram of PI16 inhibition of NF-κB signalling pathway and BLCA metastasis.1
Fig. 1 Proposed model diagram of PI16 inhibition of NF-κB signalling pathway and BLCA metastasis.1
Key structural properties of PI16:
- Secretory globular glycoprotein structure
- Containing a conservative protease inhibitory domain
- The stable folding region rich in cysteine
Functions of PI16
The main function of the PI16 protein is to regulate the cell cycle and participate in immune regulation. At the same time, it is also involved in maintaining tissue homeostasis and the fibrotic processes related to diseases.
| Function | Description |
|---|---|
| Cell Cycle Regulation | By inducing cell cycle arrest at the G1 phase, it inhibits the proliferation of various types of cells. |
| Immune Regulation | It is expressed in the stromal cells of immune organs such as lymph nodes and thymus, regulating the migration and function of immune cells. |
| Maintenance of Tissue Homeostasis | It participates in the remodeling of extracellular matrix in normal tissues and maintains the stability of tissue structure. |
| Fibrosis Promotion | It is highly expressed in fibrotic lesions of organs such as the lungs and liver, promoting the activation of fibroblasts and collagen deposition. |
| Tumor Microenvironment Regulation | It affects the functions of tumor-associated fibroblasts and participates in immune evasion and tumor progression. |
Unlike typical cell cycle inhibitory proteins (such as p21), the function of PI16 does not rely on the p53 pathway. Instead, it achieves its function by influencing extracellular signals and the interaction between the extracellular matrix and cells. This makes it a key factor in regulating the tissue-specific microenvironment.
Applications of PI16 and PI16 Antibody in Literature
1. Kuang, Xiangqin, et al. "Peptidase inhibitor (PI16) impairs bladder cancer metastasis by inhibiting NF-κB activation via disrupting multiple-site ubiquitination of NEMO." Cellular & Molecular Biology Letters 28.1 (2023): 62. https://doi.org/10.1186/s11658-023-00465-6
The article indicates that the expression of PI16 is downregulated in bladder cancer and is associated with poor prognosis. Its overexpression can inhibit the ubiquitination of NEMO by interacting with ANXA1, thereby blocking the NF-κB pathway, inhibiting tumor growth, metastasis and angiogenesis, and having tumor suppressor function.
2. Sun, Yuankai, et al. "Regulatory role of PI16 in autoimmune arthritis and intestinal inflammation: implications for Treg cell differentiation and function." Journal of Translational Medicine 22.1 (2024): 327. https://doi.org/10.1186/s12967-024-05082-1
The research has found that PI16 inhibits the differentiation and function of regulatory T cells (Tregs). Specific knockout of the PI16 gene in Tregs can enhance the dominance of Tregs and improve autoimmune arthritis and colitis, providing new ideas for the treatment of related diseases.
3. McCartney, Erika E., Yein Chung, and Matthew B. Buechler. "Life of Pi: Exploring functions of Pi16+ fibroblasts." F1000Research 13 (2024): 126. https://doi.org/10.12688/f1000research.143511.2
This article reviews the studies on fibroblasts expressing PI16. As a potential precursor state of this cell lineage, PI16-labeled cells provide functional flexibility in dynamic processes such as tissue growth, inflammation, and injury repair.
4. Wang, Pusen, et al. "PI16 attenuates response to sorafenib and represents a predictive biomarker in hepatocellular carcinoma." Cancer Medicine 9.19 (2020): 6972-6983. https://doi.org/10.1002/cam4.3331
The study found that PI16 is highly expressed in liver cancer tissues. It reduces the sensitivity of liver cancer cells to sorafenib by inhibiting the p38 MAPK/caspase apoptosis pathway. Clinical data show that patients with high levels of PI16 have a poorer response to sorafenib treatment and a worse prognosis, suggesting that PI16 can serve as a potential efficacy predictor marker.
5. De Martin, Angelina, et al. "PI16+ reticular cells in human palatine tonsils govern T cell activity in distinct subepithelial niches." Nature Immunology 24.7 (2023): 1138-1148. https://doi.org/10.1038/s41590-023-01502-4
The research has found that the reticular cells expressing PI16 in human tonsils are the most significant subpopulation of matrix cells in the structural remodeling during the inflammatory response. Through interaction with lymphocytes, they play a crucial role in immune regulation in the oropharyngeal mucosa immunity.
Creative Biolabs: PI16 Antibodies for Research
Creative Biolabs specializes in the production of high-quality PI16 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom PI16 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our PI16 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Kuang, Xiangqin, et al. "Peptidase inhibitor (PI16) impairs bladder cancer metastasis by inhibiting NF-κB activation via disrupting multiple-site ubiquitination of NEMO." Cellular & Molecular Biology Letters 28.1 (2023): 62. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1186/s11658-023-00465-6
Anti-PI16 antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-C1QC Recombinant Antibody (CBFYC-0600) (CBMAB-C0654-FY)

-
Mouse Anti-CECR2 Recombinant Antibody (CBWJC-2465) (CBMAB-C3533WJ)

-
Human Anti-SARS-CoV-2 S1 Monoclonal Antibody (CBFYR-0120) (CBMAB-R0120-FY)

-
Mouse Anti-CARTPT Recombinant Antibody (113612) (CBMAB-C2450-LY)

-
Mouse Anti-AHCYL1 Recombinant Antibody (V2-180270) (CBMAB-A1703-YC)

-
Rabbit Anti-ALOX5AP Recombinant Antibody (CBXF-1219) (CBMAB-F0750-CQ)

-
Mouse Anti-DLC1 Recombinant Antibody (D1009) (CBMAB-D1009-YC)

-
Mouse Anti-BIRC7 Recombinant Antibody (88C570) (CBMAB-L0261-YJ)

-
Mouse Anti-CGAS Recombinant Antibody (CBFYM-0995) (CBMAB-M1146-FY)

-
Mouse Anti-COL1A2 Recombinant Antibody (CF108) (V2LY-1206-LY626)

-
Mouse Anti-CTNND1 Recombinant Antibody (CBFYC-2414) (CBMAB-C2487-FY)

-
Mouse Anti-ABIN2 Recombinant Antibody (V2-179106) (CBMAB-A0349-YC)

-
Mouse Anti-AMACR Recombinant Antibody (CB34A) (CBMAB-CA034LY)

-
Mouse Anti-ADGRE5 Recombinant Antibody (V2-360335) (CBMAB-C2088-CQ)

-
Mouse Anti-APOA1 Monoclonal Antibody (CBFYR0637) (CBMAB-R0637-FY)

-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Mouse Anti-DMPK Recombinant Antibody (CBYCD-324) (CBMAB-D1200-YC)

-
Mouse Anti-BIRC3 Recombinant Antibody (315304) (CBMAB-1214-CN)

-
Mouse Anti-CSPG4 Recombinant Antibody (CBFYM-1050) (CBMAB-M1203-FY)

-
Rat Anti-CD63 Recombinant Antibody (7G4.2E8) (CBMAB-C8725-LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






