KDM6A Antibodies
Background
The KDM6A gene encodes a key histone demethylase, which mainly activates a series of key genes regulating embryonic development, cell differentiation and immune response by catalyzing the demethylation of histone H3 lysine 27 (H3K27me2/3) in the cell nucleus. As an important epigenetic regulator, the loss of function of this gene has been confirmed to be closely related to various developmental abnormalities and cancers. For instance, patients with typical Kabuki syndrome often carry mutations in this gene. As one of the earliest discovered H3K27 demethylases, the molecular mechanism analysis of KDM6A has greatly deepened the academic understanding of the relationship between the dynamics of histone modification, chromatin remodeling and the precise regulation of gene expression, making significant contributions to a groundbreaking breakthrough in the field of epigenetics.
Structure of KDM6A
The KDM6A gene encodes an important histone demethylase with a molecular weight of approximately 170 kDa. This protein plays a core role in the epigenetic regulation of gene expression by catalyzing the demethylation of lysine 27 (H3K27) on histone H3.
| Species | Human | Mouse | Zebrafish |
| Functional conservatism | High-level expression, X-linked | Homologous gene Utx, highly conserved in function | Direct homologs exist and are involved in embryonic development |
| Key domain | JmjC catalytic domain | Conservative JmjC domain | JmjC structure domain, high sequence similarity |
| Main functions | Demethylation activity regulates development and differentiation | Necessary for normal embryonic development | Participate in the development of cardiac and neural crest cells |
The primary structure of the KDM6A protein contains a highly conserved JmjC catalytic domain, which is at the core of its enzymatic activity. Its tertiary structure forms a specific spatial conformation, which can precisely recognize and bind to the modified histone tail. This protein specifically removes the dimethyl and trimethyl markers of H3K27 through its iron (Fe(II)) and α-ketoglutarate dependent catalytic mechanism, thereby activating a series of key genes that control embryonic development, cell differentiation and immune response. The functional loss of KDM6A is closely related to a variety of human diseases, among which the most famous one is Kabuki syndrome.
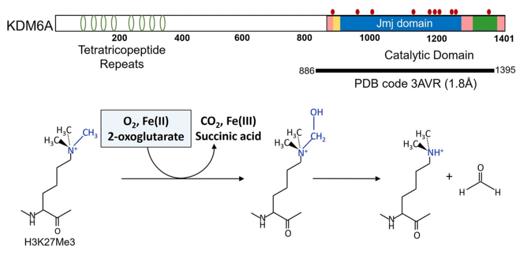 Fig. 1 Domain structure and the schematic of KMD6A catalytic mechanism.1
Fig. 1 Domain structure and the schematic of KMD6A catalytic mechanism.1
Key structural properties of KDM6A:
- Highly conserved JmjC catalytic domain
- Zinc finger domain
- Fe(II) - and α-ketoglutaric acid-dependent catalytic centers
Functions of KDM6A
The main function of the KDM6A gene is to regulate gene expression through histone demethylation and participate in a variety of key biological processes.
| Function | Description |
| Gene expression activation | Catalytically remove the dimethyl/trimethyl markers on histone H3K27, relieve transcriptional suppression, and activate the target gene. |
| Regulation of embryonic development | Regulate key embryonic development processes such as mesodermal differentiation, cardiac development and neural crest cell migration. |
| Cell fate determination | In stem cells, somatic cell reprogramming and decisive epigenetic switch role in immune cell function. |
| Tumor suppression | As an important tumor suppressor, its functional inactivation is commonly seen in various hematological malignancies and solid tumors. |
| Aging is associated with diseases | Abnormal expression or activity is closely related to Kabuki syndrome, aging-related diseases and metabolic disorders. |
Unlike simple enzymes that only catalyze a single substrate, KDM6A integrates developmental signals and epigenetic states through its complex chromatin reading and modification capabilities, precisely coordinating the sequential expression of long gene clusters.
Applications of KDM6A and KDM6A Antibody in Literature
1. Chi, Young-In, et al. "Molecular mechanics and dynamic simulations of well-known Kabuki syndrome-associated KDM6A variants reveal putative mechanisms of dysfunction." Orphanet Journal of Rare Diseases 16.1 (2021): 66. https://doi.org/10.1186/s13023-021-01692-w
This study, through molecular simulation analysis, found that 11 missense variations of KDM6A could damage the demethylase function of the protein by disrupting its structure, chemical environment and catalytic domain kinetics, among which the C1153R variation might have a key allosteric impact. The research results can provide a new direction for the analysis of the pathogenic mechanism of variations.
2. Liu, Lei, et al. "KDM6A-ARHGDIB axis blocks metastasis of bladder cancer by inhibiting Rac1." Molecular cancer 20.1 (2021): 77. https://doi.org/10.1186/s12943-021-01369-9
Research has found that FOXA1 in bladder cancer can activate the expression of histone demethylase KDM6A, and then inhibit tumor migration and invasion through ARHGDIB. The loss of KDM6A function promotes metastasis, while EZH2 inhibitors can reverse this effect, suggesting that both are potential therapeutic targets.
3. Liu, Jiye, et al. "Epigenetic regulation of CD38/CD48 by KDM6A mediates NK cell response in multiple myeloma." Nature Communications 15.1 (2024): 1367. https://doi.org/10.1038/s41467-024-45561-z
Research has found that the deletion of KDM6A significantly down-regulates the expression of CD38 by increasing the H3K27me3 modification of the CD38 gene promoter, leading to resistance of multiple myeloma to daratumumab. The use of EZH2 inhibitors can reverse this resistance, providing a new strategy for clinical combination therapy.
4. Chang, Soojeong, Sujin Yim, and Hyunsung Park. "The cancer driver genes IDH1/2, JARID1C/KDM5C, and UTX/KDM6A: crosstalk between histone demethylation and hypoxic reprogramming in cancer metabolism." Experimental & molecular medicine 51.6 (2019): 1-17. https://doi.org/10.1038/s12276-019-0230-6
Research has found that histone demethylases JARID1C/KDM5C and UTX/KDM6A have been identified as cancer driver genes. Studies have found that metabolites such as D-2HG can inhibit its activity, indicating that histone modification and metabolic reprogramming have key interactions in tumor development.
5. Guo, Wenyun, et al. "KDM6A promotes hepatocellular carcinoma progression and dictates targeted drug efficacy by upregulating FGFR4 expression." Clinical and translational medicine 13.10 (2023): e1452. https://doi.org/10.1002/ctm2.1452
Studies have found that KDM6A is highly expressed in liver cancer and has a poor prognosis. It activates the PI3K-AKT-mTOR pathway by upregulating FGFR4, promoting tumor growth and metabolic reprogramming. It is worth noting that inhibiting KDM6A can reduce the sensitivity of liver cancer to the targeted drug.
Creative Biolabs: KDM6A Antibodies for Research
Creative Biolabs specializes in the production of high-quality KDM6A antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom KDM6A Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our KDM6A antibodies, custom preparations, or technical support, contact us at email.
Reference
- Chi, Young-In, et al. "Molecular mechanics and dynamic simulations of well-known Kabuki syndrome-associated KDM6A variants reveal putative mechanisms of dysfunction." Orphanet Journal of Rare Diseases 16.1 (2021): 66. https://doi.org/10.1186/s13023-021-01692-w
Anti-KDM6A antibodies
Products List
 Loading...
Loading...
Hot products 
-
Mouse Anti-AKR1C3 Recombinant Antibody (V2-12560) (CBMAB-1050-CN)

-
Mouse Anti-BrdU Recombinant Antibody (IIB5) (CBMAB-1038CQ)

-
Mouse Anti-ARIH1 Recombinant Antibody (C-7) (CBMAB-A3563-YC)

-
Rabbit Anti-Acetyl-Histone H3 (Lys36) Recombinant Antibody (V2-623395) (CBMAB-CP0994-LY)

-
Mouse Anti-FAS2 Monoclonal Antibody (1D4) (CBMAB-0071-CN)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-0790) (CBMAB-0793-YY)

-
Mouse Anti-CCS Recombinant Antibody (CBFYC-1093) (CBMAB-C1150-FY)

-
Mouse Anti-ARSA Recombinant Antibody (CBYC-A799) (CBMAB-A3679-YC)

-
Mouse Anti-Acetyl SMC3 (K105/K106) Recombinant Antibody (V2-634053) (CBMAB-AP052LY)

-
Mouse Anti-ACTG1 Recombinant Antibody (V2-179597) (CBMAB-A0916-YC)

-
Mouse Anti-CRTAM Recombinant Antibody (CBFYC-2235) (CBMAB-C2305-FY)

-
Mouse Anti-AKT1 (Phosphorylated S473) Recombinant Antibody (V2-505430) (PTM-CBMAB-0067LY)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Rat Anti-ABCC11 Recombinant Antibody (V2-179001) (CBMAB-A0236-YC)

-
Mouse Anti-EIF4G1 Recombinant Antibody (2A9) (CBMAB-A2544-LY)

-
Mouse Anti-ENPP1 Recombinant Antibody (CBFYE-0159) (CBMAB-E0375-FY)

-
Mouse Anti-8-oxoguanine Recombinant Antibody (V2-7697) (CBMAB-1869CQ)

-
Mouse Anti-CGAS Recombinant Antibody (CBFYM-0995) (CBMAB-M1146-FY)

-
Mouse Anti-AGO2 Recombinant Antibody (V2-634169) (CBMAB-AP203LY)

-
Mouse Anti-ADIPOR2 Recombinant Antibody (V2-179983) (CBMAB-A1369-YC)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






