S1pr1 Antibodies
Background
The S1pr1 gene encodes the sphingosine-1-phosphate receptor 1 (S1PR1), which is a G protein-coupled receptor mainly expressed in immune cells and endothelial cells. This receptor participates in regulating physiological processes such as lymphocyte migration, vascular barrier function, and inflammatory responses by binding to the extracellular signaling molecule sphingosine-1-phosphate (S1P). Its signaling pathway plays a crucial role in the circulation distribution of immune cells and the regulation of inflammation, making it a key target for research on autoimmune diseases and vascular dysfunction. The structure of this receptor was resolved in the early 21st century, and its specific inhibitor fingolimod has been applied in the clinical treatment of multiple sclerosis. Related research has significantly advanced the progress in the fields of immune regulation and vascular biology.
Structure of S1pr1
The S1pr1 gene encodes the sphingosine-1-phosphate receptor 1 (S1PR1), which is a seven-transmembrane G protein-coupled receptor (GPCR) with a molecular weight of approximately 42-45 kDa. The molecular weight varies slightly among different species, mainly due to the sequence polymorphism in the intracellular and extracellular loop regions.
| Species | Human | Mouse | Rat |
| Molecular Weight (kDa) | ~42.8 | ~42.5 | ~43.1 |
| Primary Structural Differences | Seven highly conserved transmembrane helical domains (TM I-VII) | Highly homologous to humans (>90%) | The ligand-binding pocket (formed by TM III, V, VI, and VII) is highly conserved. |
This receptor protein is composed of approximately 380 amino acid residues and its three-dimensional structure forms a characteristic seven-transmembrane α-helical bundle (TM I-VII). The key functional structural units include: a hydrophobic pocket formed by transmembrane helices, which is used for specific binding of the ligand sphingosine-1-phosphate (S1P); and the domain in the intracellular region that couples with the downstream Gi protein, particularly the third intracellular loop (ICL3) and the C-terminal tail, which are crucial for signal transduction. The ligand binding and activation mechanism depends on the precise interaction between the key residues in the transmembrane region (such as Arg120, Glu121) and the polar head group of the S1P molecule.
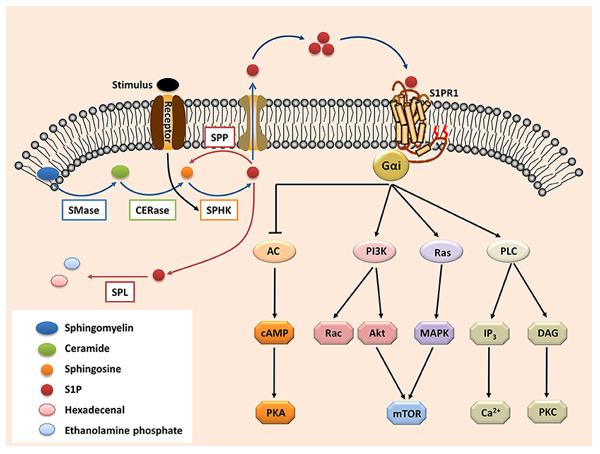 Fig. 1 The S1P-S1PR1 signaling.1
Fig. 1 The S1P-S1PR1 signaling.1
Key structural properties of S1pr1:
- Typical seven-transmembrane α-helical beam (7TM) structure
- Hydrophobic ligand (S1P) binding pocket formed by transmembrane helices III, V, VI, VII
- Conservative DRY base sequence (Asp - Arg - Tyr) is located in the second cell inner ring
- Intracellular C-terminal Serine/threonine phosphorylation sites
Functions of S1pr1
The S1P1 receptor encoded by the S1pr1 gene mainly functions to regulate the migration of immune cells and the integrity of the vascular barrier. It is also widely involved in various physiological and pathological processes, including inflammatory responses, lymphocyte recirculation, and regulation of endothelial function.
| Function | Description |
| Immune cell migration | By sensing the concentration gradient of S1P in the blood and lymph fluid, it guides lymphocytes to enter the circulatory system from lymphoid organs, which is the basis of immune surveillance. |
| Vascular Barrier Regulation | Activated in endothelial cells, it enhances intercellular connections, maintains vascular permeability homeostasis, and prevents plasma leakage. |
| Inflammation Regulation | Inhibit the abnormal aggregation of immune cells (such as T cells, dendritic cells) in inflammatory tissues, thereby exerting anti-inflammatory effects. |
| Lymphocyte Recirculation | As an "exit signal", it drives naive lymphocytes and central memory T cells to leave the thymus and secondary lymphoid organs and enter the peripheral circulation. |
| Cardiac Development and Function | It participates in the migration of cardiac progenitor cells and the formation of the atrioventricular septum during the embryonic stage, and affects angiogenesis and cardiac protection in adulthood. |
The signal transduction of S1PR1 is mainly mediated through the Gi protein pathway. Its activation leads to a decrease in the activity of inhibitory adenylate cyclase, activation of Rac GTPase, and mobilization of intracellular calcium ions, thereby coordinating cytoskeleton rearrangement and directed migration. Unlike other S1P receptors (such as S1PR2 and S1PR3), the signals of these receptors often antagonize each other, jointly finely regulating cell localization.
Applications of S1pr1 and S1pr1 Antibody in Literature
1. Chai, Yanfei, et al. "S1PR1 suppresses lung adenocarcinoma progression through p-STAT1/miR-30c-5 p/FOXA1 pathway." Journal of Experimental & Clinical Cancer Research 43.1 (2024): 304. https://doi.org/10.1186/s13046-024-03230-5
This study reveals that S1PR1 is lowly expressed in lung adenocarcinoma and is positively correlated with prognosis. S1PR1 inhibits the expression of COL5A1, MMP1 and SERPINE1 through the p-STAT1/miR-30c-5p/FOXA1 axis, thereby inhibiting the proliferation, migration and invasion of lung adenocarcinoma cells.
2. Dixit, Dhaval, et al. "S1PR1 inhibition induces proapoptotic signaling in T cells and limits humoral responses within lymph nodes." The Journal of Clinical Investigation 134.4 (2024). https://doi.org/10.1172/JCI174984
This study reveals that S1PR1 regulates the balance of the BCL2 family by inhibiting JNK activity, maintaining the survival of naive T cells. Its antagonists (such as ozanimod) affect T-cell homeostasis by interfering with this pathway, thereby limiting the immune response capacity within lymph nodes.
3. Geng, Xin, et al. "S1PR1 regulates lymphatic valve development and tertiary lymphoid organ formation in the ileum." Journal of Experimental Medicine 222.9 (2025): e20241799. https://doi.org/10.1084/jem.20241799
This study reveals that the absence of S1PR1 in lymphatic endothelial cells can lead to a reduction and dysfunction of lymphatic valves, causing the formation of tertiary lymphoid organs in the terminal ileum. This pathway affects lymphatic development by regulating key factors such as shear stress and FOXC2, revealing the novel role of S1PR1 in lymphatic structural abnormalities and subclinical inflammation.
4. Xiao, Lan, et al. "S1P-S1PR1 signaling: the "Sphinx" in osteoimmunology." Frontiers in immunology 10 (2019): 1409. https://doi.org/10.3389/fimmu.2019.01409
This study reveals that the S1P-S1PR1 signaling axis plays a core role in bone immunology, directly regulating bone formation and resorption, and indirectly influencing bone homeostasis by regulating immune responses such as T cells and macrophages. The inflammatory dysregulation of this pathway is closely related to chronic bone diseases such as rheumatoid arthritis.
5. Chen, Tianyi, et al. "Sphingosine-1-phosphate derived from PRP-Exos promotes angiogenesis in diabetic wound healing via the S1PR1/AKT/FN1 signalling pathway." Burns & Trauma 11 (2023): tkad003. https://doi.org/10.1093/burnst/tkad003
This study reveals that S1P in platelet-rich plasma exosomes significantly promotes angiogenesis at diabetic wounds and accelerates wound healing by activating the S1PR1/AKT/FN1 signaling pathway. This provides a new theoretical basis for the treatment of diabetic foot ulcers.
Creative Biolabs: S1pr1 Antibodies for Research
Creative Biolabs specializes in the production of high-quality S1pr1 antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom S1pr1 Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our S1pr1 antibodies, custom preparations, or technical support, contact us at email.
Reference
- Xiao, Lan, et al. "S1P-S1PR1 signaling: the "Sphinx" in osteoimmunology." Frontiers in immunology 10 (2019): 1409. https://doi.org/10.3389/fimmu.2019.01409
Anti-S1pr1 antibodies
Products List
 Loading...
Loading...
Hot products 
-
Mouse Anti-CCT6A/B Recombinant Antibody (CBXC-0168) (CBMAB-C5570-CQ)

-
Mouse Anti-ALB Recombinant Antibody (V2-55272) (CBMAB-H0819-FY)

-
Mouse Anti-ENO1 Recombinant Antibody (CBYC-A950) (CBMAB-A4388-YC)

-
Mouse Anti-DLG1 Monolconal Antibody (4F3) (CBMAB-0225-CN)

-
Human Anti-SARS-CoV-2 Spike Recombinant Antibody (CR3022) (CBMAB-CR014LY)

-
Mouse Anti-FOXL1 Recombinant Antibody (CBXF-0845) (CBMAB-F0462-CQ)

-
Mouse Anti-AFM Recombinant Antibody (V2-634159) (CBMAB-AP185LY)

-
Mouse Anti-CTNND1 Recombinant Antibody (CBFYC-2414) (CBMAB-C2487-FY)

-
Mouse Anti-DLC1 Recombinant Antibody (D1009) (CBMAB-D1009-YC)

-
Mouse Anti-BBS2 Recombinant Antibody (CBYY-0253) (CBMAB-0254-YY)

-
Mouse Anti-AMACR Recombinant Antibody (CB34A) (CBMAB-CA034LY)

-
Mouse Anti-AFDN Recombinant Antibody (V2-58751) (CBMAB-L0408-YJ)

-
Mouse Anti-ATP5F1A Recombinant Antibody (51) (CBMAB-A4043-YC)

-
Mouse Anti-CAT Recombinant Antibody (724810) (CBMAB-C8431-LY)

-
Mouse Anti-BRCA2 Recombinant Antibody (CBYY-1728) (CBMAB-2077-YY)

-
Mouse Anti-AK4 Recombinant Antibody (V2-180419) (CBMAB-A1891-YC)

-
Mouse Anti-CIITA Recombinant Antibody (CBLC160-LY) (CBMAB-C10987-LY)

-
Mouse Anti-ARHGDIA Recombinant Antibody (CBCNA-009) (CBMAB-R0415-CN)

-
Mouse Anti-ATP1B1 Recombinant Antibody (E4) (CBMAB-0463-LY)

-
Mouse Anti-AGK Recombinant Antibody (V2-258056) (CBMAB-M0989-FY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






