TOP2A Antibodies
Background
The TOP2A gene encodes the topoisomerase IIα protein, which is mainly present in the cell nucleus of eukaryotic cells. This enzyme regulates the topological structure of DNA by temporarily cutting and rejoining the DNA double strands, thereby playing a crucial role in key cellular processes such as chromosome separation, transcriptional regulation, and maintenance of genomic stability. Rapidly proliferating cells (such as cancer cells) highly rely on TOP2A to support their frequent cell division. Therefore, this gene has become an important target for various chemotherapy drugs. The function and structure of this gene have been deeply analyzed since their discovery in the last century, and the research has greatly enhanced our understanding of DNA metabolism, cell cycle regulation, and the mechanism of action of anti-cancer drugs. It holds significant value in the field of molecular biology and tumor treatment.
Structure of TOP2A
The TOP2A gene encodes the topoisomerase IIα protein with a molecular weight of approximately 170-180 kDa. The molecular weight may vary among different species or under different cellular conditions, which is mainly related to post-translational modifications (such as phosphorylation) and alternative splicing variants.
| Species | Human (HeLa cells) | Mouse (embryonic stem cells) | Yeast (S. cerevisiae) | Common research models (recombinant proteins) |
| Molecular Weight (kDa) | ~170 | ~170 | ~160 (homologous substance) | Often use catalytic core structure domain (~ 110 kDa) |
| Primary Structural Differences | The expression level is the highest during the S/G2 phase of the cell cycle, and it is highly phosphorylated. | Highly homologous to human proteins and with conserved functions | They are homologous proteins with similar structure but different regulation | The core ATPase and DNA cleavage-rejoining domains are often utilized for biochemical research |
This protein consists of approximately 1,530 amino acids and forms a complex multi-domain three-dimensional structure. Its primary structure contains several key functional domains: the N-terminal ATPase domain, the central DNA binding and cleavage domain (containing a conserved tyrosine active site), and the C-terminal nuclear localization signal. The secondary structure is composed of alternating α helices and β sheets, jointly forming a "gateway" structure that accommodates DNA. Its key mechanism of action involves two main tyrosine residues (Tyr-804 and Tyr-820, taking the human sequence as an example), which work together to temporarily break the DNA double helix and catalyze the passage and reconnection of the strands, thereby unwinding the DNA supercoil. This process relies on ATP hydrolysis for energy supply.
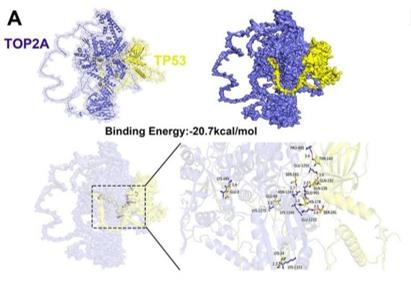 Fig. 1 Molecular docking analysis of TOP2A and TP53 Proteins.1
Fig. 1 Molecular docking analysis of TOP2A and TP53 Proteins.1
Key structural properties of TOP2A:
- Multi-domain homodimeric configuration, containing DNA binding and cleavage domains
- Conservative tyrosine active center (such as Tyr-804/820)
- ATP binds to the hydrolyzed domain, providing energy for conformational changes
- Open door structure of DNA, double-stranded DNA will be allowed to pass
Functions of TOP2A
The TOP2A gene encodes topoisomerase IIα, which primarily functions to regulate the DNA topological structure during cell division. However, it is also widely involved in various cellular processes, including transcriptional regulation and DNA damage repair.
| Function | Description |
| DNA Unwinding | By temporarily cutting and rejoining the double strands of DNA, it removes the supercoiling and knots of DNA, clearing obstacles for processes such as replication and transcription. |
| Chromosome Separation | During the later stage of cell mitosis, the physical connections (such as kinetochores) between sister chromatids are dissociated to ensure the correct separation of chromosomes. |
| Maintenance of genomic stability | By using precise cutting-rejoining reactions to handle DNA entanglement, it prevents the resulting DNA breaks and genomic rearrangements. |
| Anti-cancer Drug Targets | Their functions are crucial for rapidly proliferating cancer cells, making them the targets for various chemotherapy drugs (such as etoposide and doxorubicin). |
| Transcriptional Regulation | By altering the DNA topological state in specific gene regions, it affects the binding of transcription factors and the expression level of genes. |
Unlike type I topoisomerases that can only unwind the negative supercoils of DNA, TOP2A can simultaneously handle both positive and negative supercoils as well as DNA knots, and it relies on ATP hydrolysis to provide energy. This makes it play a core and indispensable role in maintaining the overall topological balance of the genome.
Applications of TOP2A and TOP2A Antibody in Literature
1. Liu, Xinyue, et al. "Knockdown of TOP2A reverses cisplatin resistance in ovarian cancer by inhibiting EMT via ferroptosis mediated by the TP53/GPX4/SLC7A11 axis." Frontiers in Immunology 16 (2025): 1675373. https://doi.org/10.3389/fimmu.2025.1675373
Research has found that TOP2A regulates the GPX4/SLC7A11 axis by binding to TP53, influencing ferroptosis and EMT processes, and is a key mechanism for cisplatin resistance in ovarian cancer. High expression of TOP2A indicates a poor prognosis and can be used as a potential therapeutic target.
2. Thu, Vo Thuy Anh, et al. "Deciphering linezolid-induced hematologic toxicity: Targeting TOP2A and TOP2B via its primary metabolite PNU142586." Science advances 11.22 (2025): eadt5833. https://doi.org/10.1126/sciadv.adt5833
Research has found that the linezolid metabolite PNU142586 interferes with the function of DNA topoisomerase 2-α/2-β by binding to it, leading to cell proliferation inhibition and mitochondrial damage, which reveals a new mechanism of the antibiotic's hematological toxicity.
3. Zhang, Kaiwen, et al. "TOP2A modulates signaling via the AKT/mTOR pathway to promote ovarian cancer cell proliferation." Cancer Biology & Therapy 25.1 (2024): 2325126. https://doi.org/10.1080/15384047.2024.2325126
Research has found that TOP2A is highly expressed in ovarian cancer and is associated with a poor prognosis. It drives cell proliferation and cycle progression by activating the AKT/mTOR pathway, and its knockout can induce cell cycle arrest and apoptosis. Studies have shown that TOP2A is a potential prognostic marker and therapeutic target for ovarian cancer.
4. Zhang, Yufeng, et al. "TOP2A correlates with poor prognosis and affects radioresistance of medulloblastoma." Frontiers in oncology 12 (2022): 918959. https://doi.org/10.3389/fonc.2022.918959
Studies have found that TOP2A is highly expressed in medulloblastoma and suggests a poor prognosis. Its expression level positively regulates the malignant behavior and radiotherapy resistance of tumor cells, and the mechanism involves the Wnt/β-catenin signaling pathway. Targeting TOP2A can enhance radiotherapy sensitivity.
5. Wang, Ke, et al. "EZH2-H3K27me3-mediated silencing of mir-139-5p inhibits cellular senescence in hepatocellular carcinoma by activating TOP2A." Journal of Experimental & Clinical Cancer Research 42.1 (2023): 320. https://doi.org/10.1186/s13046-023-02855-2
Studies have found that EZH2 inhibits the upregulation of TOP2A expression by miR-139-5p, thereby suppressing the senescence of liver cancer cells and promoting their proliferation. Targeting this axis can induce cellular senescence, providing a new strategy for the treatment of liver cancer.
Creative Biolabs: TOP2A Antibodies for Research
Creative Biolabs specializes in the production of high-quality TOP2A antibodies for research and industrial applications. Our portfolio includes monoclonal antibodies tailored for ELISA, Flow Cytometry, Western blot, immunohistochemistry, and other diagnostic methodologies.
- Custom TOP2A Antibody Development: Tailor-made solutions to meet specific research requirements.
- Bulk Production: Large-scale antibody manufacturing for industry partners.
- Technical Support: Expert consultation for protocol optimization and troubleshooting.
- Aliquoting Services: Conveniently sized aliquots for long-term storage and consistent experimental outcomes.
For more details on our TOP2A antibodies, custom preparations, or technical support, contact us at email.
Reference
- Liu, Xinyue, et al. "Knockdown of TOP2A reverses cisplatin resistance in ovarian cancer by inhibiting EMT via ferroptosis mediated by the TP53/GPX4/SLC7A11 axis." Frontiers in Immunology 16 (2025): 1675373. https://doi.org/10.3389/fimmu.2025.1675373
Anti-TOP2A antibodies
 Loading...
Loading...
Hot products 
-
Mouse Anti-GDF5 Recombinant Antibody (1F4) (CBMAB-G2740-LY)

-
Mouse Anti-AQP2 Recombinant Antibody (G-3) (CBMAB-A3359-YC)

-
Mouse Anti-AAV9 Recombinant Antibody (V2-634029) (CBMAB-AP023LY)

-
Mouse Anti-CDK7 Recombinant Antibody (CBYY-C1783) (CBMAB-C3221-YY)

-
Mouse Anti-ACVR1C Recombinant Antibody (V2-179685) (CBMAB-A1041-YC)

-
Mouse Anti-C4B Recombinant Antibody (CBYY-C2996) (CBMAB-C4439-YY)

-
Mouse Anti-DDC Recombinant Antibody (8E8) (CBMAB-0992-YC)

-
Mouse Anti-FPR2 Recombinant Antibody (1D6) (CBMAB-F2628-CQ)

-
Mouse Anti-BIRC5 Recombinant Antibody (6E4) (CBMAB-CP2646-LY)

-
Mouse Anti-FTH1 Recombinant Antibody (CBXF-1896) (CBMAB-F3426-CQ)

-
Rabbit Anti-CAMK2A Recombinant Antibody (BA0032) (CBMAB-0137CQ)

-
Rat Anti-AChR Recombinant Antibody (V2-12500) (CBMAB-0990-CN)

-
Mouse Anti-NSUN6 Recombinant Antibody (D-5) (CBMAB-N3674-WJ)

-
Mouse Anti-DLL4 Recombinant Antibody (D1090) (CBMAB-D1090-YC)

-
Armenian hamster Anti-CD40 Recombinant Antibody (HM40-3) (CBMAB-C10365-LY)

-
Rabbit Anti-Acetyl-Histone H3 (Lys36) Recombinant Antibody (V2-623395) (CBMAB-CP0994-LY)

-
Mouse Anti-ADIPOR2 Recombinant Antibody (V2-179983) (CBMAB-A1369-YC)

-
Mouse Anti-CALR Recombinant Antibody (CBFYC-0763) (CBMAB-C0818-FY)

-
Rabbit Anti-ATF4 Recombinant Antibody (D4B8) (CBMAB-A3872-YC)

-
Mouse Anti-14-3-3 Pan Recombinant Antibody (V2-9272) (CBMAB-1181-LY)

- AActivation
- AGAgonist
- APApoptosis
- BBlocking
- BABioassay
- BIBioimaging
- CImmunohistochemistry-Frozen Sections
- CIChromatin Immunoprecipitation
- CTCytotoxicity
- CSCostimulation
- DDepletion
- DBDot Blot
- EELISA
- ECELISA(Cap)
- EDELISA(Det)
- ESELISpot
- EMElectron Microscopy
- FFlow Cytometry
- FNFunction Assay
- GSGel Supershift
- IInhibition
- IAEnzyme Immunoassay
- ICImmunocytochemistry
- IDImmunodiffusion
- IEImmunoelectrophoresis
- IFImmunofluorescence
- IGImmunochromatography
- IHImmunohistochemistry
- IMImmunomicroscopy
- IOImmunoassay
- IPImmunoprecipitation
- ISIntracellular Staining for Flow Cytometry
- LALuminex Assay
- LFLateral Flow Immunoassay
- MMicroarray
- MCMass Cytometry/CyTOF
- MDMeDIP
- MSElectrophoretic Mobility Shift Assay
- NNeutralization
- PImmunohistologyp-Paraffin Sections
- PAPeptide Array
- PEPeptide ELISA
- PLProximity Ligation Assay
- RRadioimmunoassay
- SStimulation
- SESandwich ELISA
- SHIn situ hybridization
- TCTissue Culture
- WBWestern Blot






